Abstract
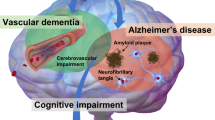
Cognitive impairment commonly accompanies clinical syndromes associated with vascular disease of the brain. Because of evolving definitional criteria, however, the frequency of cognitive impairment attributable to cerebrovascular disease is difficult to determine. Dementia occurs in up to one-third of elderly patients with stroke, a subset of whom have Alzheimer's disease (AD) rather than a pure vascular dementia syndrome. In fact, pure vascular dementia has been shown to be uncommon in most large autopsy series. A mixed etiology of AD and cerebrovascular disease is thought to become more common with increasing age, although no clinical criteria for the diagnosis of AD with cerebrovascular disease are currently available. Epidemiological studies have implicated subcortical small-vessel disease as a risk factor for cognitive impairment and dementia, but the cognitive expression and clinical significance of MRI white matter changes in individual patients is difficult to establish. The frequency of specific neuropathologic features of vascular cognitive impairment depends largely on study inclusion criteria. Cerebral meningocortical microangiopathies with distinctive clinicopathological profiles are associated with dementia in both sporadic cases and familial syndromes. In patients with AD, the contribution of amyloid-β protein to the degree of cognitive impairment has not been clearly defined.
Key Points
-
Several clinical subtypes of vascular cognitive impairment due to vascular disease are now recognized, ranging in severity from mild cognitive impairment to dementia
-
The risk of dementia after stroke is increased in older patients and in those with pre-existing cognitive impairment or cerebrovascular disease
-
The cognitive consequences of subcortical small-vessel disease is variable, suggesting that the subcortical white matter changes visualized on conventional MRI might be incomplete markers of the total burden of cerebrovascular disease
-
The cognitive profile of vascular cognitive impairment is predominantly subcortical, with prominent psychomotor slowing and executive deficits, but relatively preserved language and recognition memory
-
From a neuropathologic perspective, the morphologic substrates or correlates of vascular dementia are extremely heterogeneous
-
Cerebral microvascular diseases, both sporadic and genetically determined, including cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) and cerebral amyloid angiopathy, are instructive in terms of providing pathologic models of age-related arteriopathies and their consequences for brain parenchyma
-
The brains of aged individuals are likely to show impaired function as a consequence of combined 'Alzheimerization' and cerebrovascular disease
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chui H (2005) Neuropathology lessons in vascular dementia. Alzheimer Dis Assoc Disord 19: 45–52
Fernando MS and Ince PG (2004) Vascular pathologies and cognition in a population-based cohort of elderly people. J Neurol Sci 226: 13–17
Neuropathology Group of the Medical Research Council Cognitive Function and Ageing Study (2001) Pathological correlates of late-onset dementia in a multicentre, community-based population in England and Wales. Neuropathology Group of the Medical Research Council Cognitive Function and Ageing Study (MRC CFAS). Lancet 357: 169–175
O'Brien JT et al. (2003) Vascular cognitive impairment. Lancet Neurol 2: 89–98
Rockwood K et al. (2005) Clinical and radiographic subtypes of vascular cognitive impairment in a clinic-based cohort study. J Neurol Sci 240: 7–14
Vinters HV et al. (2000) Neuropathologic substrates of ischemic vascular dementia. J Neuropathol Exp Neurol 59: 931–945
Pohjasvaara T et al. (2000) Comparison of different clinical criteria (DSM-III, ADDTC, ICD-10, NINDS-AIREN, DSM-IV) for the diagnosis of vascular dementia. National Institute of Neurological Disorders and Stroke-Association Internationale pour la Recherche et l'Enseignement en Neurosciences. Stroke 31: 2952–2957
Bowler JV (2002) The concept of vascular cognitive impairment. J Neurol Sci 203–204: 11–15
Sachdev P (1999) Vascular cognitive disorder. Int J Geriatr Psychiatry 14: 402–403
Roman GC et al. (2004) Vascular cognitive disorder: a new diagnostic category updating vascular cognitive impairment and vascular dementia. J Neurol Sci 226: 81–87
Roman GC et al. (2002) Subcortical ischaemic vascular dementia. Lancet Neurol 1: 426–436
Leys D et al. (2005) Poststroke dementia. Lancet Neurol 4: 752–759
Linden T et al. (2004) Cognitive impairment and dementia 20 months after stroke. Neuroepidemiology 23: 45–52
Desmond DW et al. (2002) Incidence of dementia after ischemic stroke: results of a longitudinal study. Stroke 33: 2254–2260
Henon H et al. (2001) Poststroke dementia: incidence and relationship to prestroke cognitive decline. Neurology 57: 1216–1222
Luchsinger JA et al. (2005) Aggregation of vascular risk factors and risk of incident Alzheimer disease. Neurology 65: 545–551
Barba R et al. (2000) Poststroke dementia: clinical features and risk factors. Stroke 31: 1494–1501
Pohjasvaara T et al. (2000) How complex interactions of ischemic brain infarcts, white matter lesions, and atrophy relate to poststroke dementia. Arch Neurol 57: 1295–1300
Lin JH et al. (2003) Prediction of poststroke dementia. Neurology 61: 343–348
Rowan E et al. (2005) Impact of hypertension and apolipoprotein E4 on poststroke cognition in subjects >75 years of age. Stroke 36: 1864–1868
Vermeer SE et al. (2003) Silent brain infarcts and the risk of dementia and cognitive decline. N Engl J Med 348: 1215–1222
Perren F et al. (2005) The syndrome of combined polar and paramedian thalamic infarction. Arch Neurol 62: 1212–1216
Madureira S et al. (1999) A follow-up study of cognitive impairment due to inferior capsular genu infarction. J Neurol 246: 764–769
Longstreth WT Jr (2005) Brain vascular disease overt and covert. Stroke 36: 2062–2063
Gold G et al. (1998) Re-evaluating the role of vascular changes in the differential diagnosis of Alzheimer's disease and vascular dementia. Eur Neurol 40: 121–129
Ott A et al. (1999) Diabetes mellitus and the risk of dementia: The Rotterdam Study. Neurology 53: 1937–1942
Langa KM et al. (2004) Mixed dementia: emerging concepts and therapeutic implications. JAMA 292: 2901–2908
Kalimo H (Ed.; 2005) Pathology and Genetics: Cerebrovascular Diseases. Basel: ISN Neuropath Press
Jellinger KA (2002) The pathology of ischemic-vascular dementia: an update. J Neurol Sci 203–204: 153–157
Hansen LA and Crain BJ (1995) Making the diagnosis of mixed and non-Alzheimer's dementias. Arch Pathol Lab Med 119: 1023–1031
Nolan KA et al. (1998) Absence of vascular dementia in an autopsy series from a dementia clinic. J Am Geriatr Soc 46: 597–604
Longstreth WT Jr et al. (2005) Incidence, manifestations, and predictors of worsening white matter on serial cranial magnetic resonance imaging in the elderly: the Cardiovascular Health Study. Stroke 36: 56–61
Schmidtke K and Hull M (2005) Cerebral small vessel disease: how does it progress? J Neurol Sci 229–230: 13–20
Ringelstein EB and Nabavi DG (2005) Cerebral small vessel diseases: cerebral microangiopathies. Curr Opin Neurol 18: 179–188
Goto T et al. (2001) Magnetic resonance imaging findings and postoperative neurologic dysfunction in elderly patients undergoing coronary artery bypass grafting. Ann Thorac Surg 72: 137–142
Udaka F et al. (2002) White matter lesions and dementia: MRI–pathological correlation. Ann NY Acad Sci 977: 411–415
Kuller LH et al. (2005) Determinants of vascular dementia in the Cardiovascular Health Cognition Study. Neurology 64: 1548–1552
Prins ND et al. (2005) Cerebral small-vessel disease and decline in information processing speed, executive function and memory. Brain 128: 2034–2041
de Groot JC et al. (2000) Cerebral white matter lesions and cognitive function: the Rotterdam Scan Study. Ann Neurol 47: 145–151
Duning T et al. (2005) Excellent cognitive performance despite massive cerebral white matter changes. Neuroradiology 47: 749–752
de Leeuw FE et al. (2004) Alzheimer's disease—one clinical syndrome, two radiological expressions: a study on blood pressure. J Neurol Neurosurg Psychiatry 75: 1270–1274
Mosley TH Jr et al. (2005) Cerebral MRI findings and cognitive functioning: the Atherosclerosis Risk in Communities study. Neurology 64: 2056–2062
Roman GC (2004) Brain hypoperfusion: a critical factor in vascular dementia. Neurol Res 26: 454–458
Kalaria RN et al. (2001) Multiple substrates of late-onset dementia: implications for brain protection. Novartis Found Symp 235: 49–60
de Leeuw FE et al. (2000) Atrial fibrillation and the risk of cerebral white matter lesions. Neurology 54: 1795–1801
Markus HS et al. (2000) Reduced cerebral blood flow in white matter in ischaemic leukoaraiosis demonstrated using quantitative exogenous contrast based perfusion MRI. J Neurol Neurosurg Psychiatry 69: 48–53
Werring DJ et al. (2004) Cognitive dysfunction in patients with cerebral microbleeds on T2*-weighted gradient-echo MRI. Brain 127: 2265–2275
Kovari E et al. (2004) Cortical microinfarcts and demyelination significantly affect cognition in brain aging. Stroke 35: 410–414
Vinters HV (2001) Cerebral amyloid angiopathy: a microvascular link between parenchymal and vascular dementia? Ann Neurol 49: 691–693
Chabriat H and Bousser MG (2003) CADASIL. Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy. Adv Neurol 92: 147–150
Dichgans M (2002) CADASIL: a monogenic condition causing stroke and subcortical vascular dementia. Cerebrovasc Dis 13 (Suppl 2): 37–41
Miao Q et al. (2004) Fibrosis and stenosis of the long penetrating cerebral arteries: the cause of the white matter pathology in cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy. Brain Pathol 14: 358–364
Opherk C et al. (2004) Long-term prognosis and causes of death in CADASIL: a retrospective study in 411 patients. Brain 127: 2533–2539
Arango-Lasprilla JC et al. (2005) Cortical and subcortical diseases: do true neuropsychological differences exist? Arch Clin Neuropsychol 21: 29–40
Laukka EJ et al. (2004) Similar patterns of cognitive deficits in the preclinical phases of vascular dementia and Alzheimer's disease. J Int Neuropsychol Soc 10: 382–391
Frisoni GB et al. (2002) Mild cognitive impairment with subcortical vascular features: clinical characteristics and outcome. J Neurol 249: 1423–1432
Graham NL et al. (2004) Distinctive cognitive profiles in Alzheimer's disease and subcortical vascular dementia. J Neurol Neurosurg Psychiatry 75: 61–71
Tierney MC et al. (2001) Recognition memory and verbal fluency differentiate probable Alzheimer disease from subcortical ischemic vascular dementia. Arch Neurol 58: 1654–1659
Garrett KD et al. (2004) The neuropsychological profile of vascular cognitive impairment—no dementia: comparisons to patients at risk for cerebrovascular disease and vascular dementia. Arch Clin Neuropsychol 19: 745–757
Fahlander K et al. (2002) Cognitive functioning in Alzheimer's disease and vascular dementia: further evidence for similar patterns of deficits. J Clin Exp Neuropsychol 24: 734–744
Looi JC and Sachdev PS (1999) Differentiation of vascular dementia from AD on neuropsychological tests. Neurology 53: 670–678
Traykov L et al. (2002) Neuropsychological deficit in early subcortical vascular dementia: comparison to Alzheimer's disease. Dement Geriatr Cogn Disord 14: 26–32
Peters N et al. (2005) The pattern of cognitive performance in CADASIL: a monogenic condition leading to subcortical ischemic vascular dementia. Am J Psychiatry 162: 2078–2085
Buffon F et al. (2006) Cognitive profile in CADASIL. J Neurol Neurosurg Psychiatry 77: 175–180
O'Sullivan M et al. (2004) Diffusion tensor imaging of thalamus correlates with cognition in CADASIL without dementia. Neurology 62: 702–707
Reed BR et al. (2004) Clinical and neuropsychological features in autopsy-defined vascular dementia. Clin Neuropsychol 18: 63–74
Vinters HV et al. (1998) Diagnostic Neuropathology, 1–49. New York: Marcel Dekker Inc.
Medana IM and Esiri MM (2003) Axonal damage: a key predictor of outcome in human CNS diseases. Brain 126: 515–530
Brown WR et al. (2000) Apoptosis in leukoaraiosis. AJNR Am J Neuroradiol 21: 79–82
Hulette C et al. (1997) Clinical-neuropathologic findings in multi-infarct dementia: a report of six autopsied cases. Neurology 48: 668–672
Del ST et al. (1990) Vascular dementia: a clinicopathological study. J Neurol Sci 96: 1–17
Lammie GA (2002) Hypertensive cerebral small vessel disease and stroke. Brain Pathol 12: 358–370
Dickson DW et al. (1994) Hippocampal sclerosis: a common pathological feature of dementia in very old (> or = 80 years of age) humans. Acta Neuropathol (Berl) 88: 212–221
Verbeek MM et al. (2000) Cerebral Amyloid Angiopathy in Alzheimer's Disease and Related Disorders. Dordrecht: Kluwer Academic Publishers
Revesz T et al. (2002) Sporadic and familial cerebral amyloid angiopathies. Brain Pathol 12: 343–357
Rensink AA et al. (2003) Pathogenesis of cerebral amyloid angiopathy. Brain Res Brain Res Rev 43: 207–223
Vinters HV (1987) Cerebral amyloid angiopathy: a critical review. Stroke 18: 311–324
Vinters HV et al. (1996) Brain parenchymal and microvascular amyloid in Alzheimer's disease. Brain Pathol 6: 179–195
Greenberg SM et al. (1993) The clinical spectrum of cerebral amyloid angiopathy: presentations without lobar hemorrhage. Neurology 43: 2073–2079
Vinters HV (1992) Cerebral amyloid angiopathy and Alzheimer's disease: two entities or one? J Neurol Sci 112: 1–3
Anders KH et al. (1997) Giant cell arteritis in association with cerebral amyloid angiopathy: immunohistochemical and molecular studies. Hum Pathol 28: 1237–1246
Eng JA et al. (2004) Clinical manifestations of cerebral amyloid angiopathy-related inflammation. Ann Neurol 55: 250–256
Haglund M et al. (2004) Severe cerebral amyloid angiopathy characterizes an underestimated variant of vascular dementia. Dement Geriatr Cogn Disord 18: 132–137
Cadavid D et al. (2000) Cerebral beta amyloid angiopathy is a risk factor for cerebral ischemic infarction: a case control study in human brain biopsies. J Neuropathol Exp Neurol 59: 768–773
Vinters HV et al. (1998) Secondary microvascular degeneration in amyloid angiopathy of patients with hereditary cerebral hemorrhage with amyloidosis, Dutch type (HCHWA-D). Acta Neuropathol (Berl) 95: 235–244
Natte R et al. (2001) Dementia in hereditary cerebral hemorrhage with amyloidosis-Dutch type is associated with cerebral amyloid angiopathy but is independent of plaques and neurofibrillary tangles. Ann Neurol 50: 765–772
Obici L et al. (2005) A novel AβPP mutation exclusively associated with cerebral amyloid angiopathy. Ann Neurol 58: 639–644
Acknowledgements
Work in H Vinters' laboratory is supported by PHS grants P50 AG 16570 and P01 AG 12435. The research of O Selnes is supported by PHS grant 35610 (NINCDS). Technical assistance provided by Justine Pomakian and Negar Khanlou. We thank Pamela Talalay for her editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Selnes, O., Vinters, H. Vascular cognitive impairment. Nat Rev Neurol 2, 538–547 (2006). https://doi.org/10.1038/ncpneuro0294
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneuro0294
This article is cited by
-
Triptolide protects against white matter injury induced by chronic cerebral hypoperfusion in mice
Acta Pharmacologica Sinica (2022)
-
Direct AT2R Stimulation Slows Post-stroke Cognitive Decline in the 5XFAD Alzheimer’s Disease Mice
Molecular Neurobiology (2022)
-
Cognitive syndromes after the first stroke
Neurological Sciences (2018)
-
Temporal changes in blood–brain barrier permeability and cerebral perfusion in lacunar/subcortical ischemic stroke
BMC Neurology (2015)
-
Early treatment of minocycline alleviates white matter and cognitive impairments after chronic cerebral hypoperfusion
Scientific Reports (2015)