Abstract
The Epstein–Barr virus (EBV) is uniquely associated with a broad range of human malignancies. In spite of their diverse cellular origin, most of these malignancies share common features, including the expression of either some or all of the EBV latent proteins, which can be potentially exploited for immune-based therapies. Here we discuss new and emerging strategies to manipulate the immune response to specifically boost T-cell immunity towards viral proteins that are expressed in EBV-associated malignancies. These strategies are used either alone or as an adjuvant therapy in combination with chemotherapy and/or monoclonal antibodies. Overall, this strategy may serve as a new paradigm for the successful multi-modality treatment of malignancies.
Similar content being viewed by others
Introduction
Epstein–Barr virus (EBV)-associated malignancies arise in both immunosuppressed and immunocompetent individuals. These malignancies are of variable cellular origin. Some involve specific genetic lesions but all involve the expression of either some or all of the EBV latent proteins, EBV-associated nuclear antigens (EBNAs) 1, 2, 3A, 3B and 3C, and latent membrane proteins (LMPs) 1, 2 (Figure 1).1 In the immunosuppressed patient, these malignancies are of B-cell lymphocyte origin. These malignancies emerge as a result of a depressed immune response mediated by either graft-directed immunosuppressive therapy (post-transplant lymphoma [PTL], defined as early and late PTL) or HIV infection (Figure 1). The Asian population seems to be particularly susceptible to EBV-associated malignancies in immunocompetent individuals, suggesting a unique interaction of environmental and genetic factors that are not fully understood.2,3,4 These malignancies are of both epithelial and lymphoid origin and include nasopharyngeal carcinoma (NPC) and gastric carcinoma together with the lymphoid malignancies, Hodgkin's lymphoma (HL), and T-cell and natural killer cell lymphomas (Figure 1).
Each malignancy is characterized by a unique pattern of latent viral gene expression, cellular origin and genetic lesions.1 The viral gene expression is a crucial determinant for designing novel immunotherapeutic strategies for these malignancies. CNS, central nervous system; EBNA 1, 2, 3A, 3B, 3C, EBV-associated nuclear antigens 1, 2, 3A, 3B, 3C; EBV, Epstein–Barr virus; Ig, immunoglobin; LMP1 and 2, latent membrane proteins 1 and 2; LP, leader protein; NK, natural killer; RTA, viral immediate-early protein, also called BRLF1 or R;TR, terminal repeat; vIL-10, viral interleukin-10.
Our understanding of the immune control of EBV infection in healthy carriers5 has now reached the stage where this response can be considered for targeting towards viral proteins expressed in the EBV-associated malignancies. In healthy individuals, CYTOTOXIC T LYMPHOCYTES (CTLs) have a major role in the immune recovery phase stimulated in response to primary EBV infection, and in controlling the latent life-long EBV infection present in previously exposed individuals.6 Much of the immune recovery response is directed towards EBNA 3A, 3B and 3C proteins, whereas the response to LMPs is known to be relatively weak. This review considers new and emerging strategies to manipulate this immune response, by specifically boosting CTL numbers to determinants known to be present in EBV-associated malignancies, either alone or in conjunction with adjuvant therapies.
Current status of the treatment of EBV-associated malignancies
At present, treatment of EBV-associated diseases predominately exploits the malignancies' cellular properties, including cellular origin/level of differentiation, viral gene expression and chemo/radiosensitivity. EBV-associated malignancies are a heterogenous group of tumors that vary in their response to conventional therapies. They are in general relatively radiosensitive and chemosensitive in their early stages, but are less amenable to conventional modality therapy in late stage or relapse.7,8 This is the case particularly in early-stage disease, with 5-year survival rates as high as 90%.7 By contrast, in the case of late-stage/relapse disease, survival outcomes as low as 50–70% are more typical;8 but these might drop to below 20% in developing countries. These data have raised the possibility of combining immunotherapy with chemo/radiotherapy, particularly in advanced disease, to improve survival. The potential tolerability of this form of therapy in terms of its modest treatment-related toxicity compared with more conventional treatments is appealing. Histologic examination of most of these malignancies shows a heavy lymphoid infiltrate, which suggests that these malignant cells might be immunoactive. The commonly held view is, however, that this infiltrate might exert an immunosuppressive effect.9,10 To circumvent this, attempts have been made to expand EBV-specific T-cell immunity in vitro in the absence of an immunosuppressive environment, and to transfer the resulting expanded T cells for the treatment of high-risk patients11,12,13,14,15,16,17,18,19,20,21 (Table 1). Indeed, despite potential immunosuppression generated by the tumor, in vivo generation of EBV-specific CTLs has been achieved successfully in patients with advanced NPC, following immunization with autologous peptide-pulsed dendritic cells.22 This form of therapy has been particularly successful in the treatment of early PTL, which is characterized by the expression of a full complement of EBV latent genes, an activated B-cell phenotype with high levels of expression of antigen-processing genes, and a proven susceptibility to EBV-specific CTLs (Figures 1 and 2). The direct application of this technology to other EBV-associated malignancies will need modifications to overcome the significant barriers—as outlined below (Table 2).
BCL, B-cell lymphoma; CNS, central nervous system; CTL, cytotoxic T lymphocyte; EBV, Epstein–Barr virus; IL-10 and IL-13, interleukins 10 and 13; LMP, latent membrane protein; pSTAT3, phosphorylated signal transducer and activator of transcription 3; PTL, post-transplant lymphoma; slg, surface immunoglobulins;TAP1 and 2, transporters 1 and 2; TARC, thymus and activation-regulated chemokine; TGF-β, transforming growth factor-beta; Th2, T-helper cell 2; T/NK lymphomas,T-cell and natural killer lymphomas; VCAM, vascular cell adhesion molecule.
Each EBV-associated malignancy is characterized by a unique cellular phenotype, which is to some extent influenced by the pattern of EBV gene expression within the malignant cell.23 The full complement of latent EBV gene expression in early PTL is coincident with an activated cell phenotype and renders this malignancy an ideal target for T-cell-based immunotherapy. Reduced EBV gene expression (i.e. EBNA1 only), and/or a consistent loss of antigen-processing function through the MHC class I pathway, severely restricts the potential use of CTL-based immunotherapeutic strategies. Examples of such malignancies include Burkitt's lymphoma, HIV-associated lymphomas and late PTLs (Figure 2). Although loss of expression of immunodominant viral antigens (the EBNA3 proteins) in HL, NPC, T-cell and natural-killer-cell lymphoma and gastric carcinoma limits potential viral targets, recent studies have suggested that LMP1 and LMP2 can be efficiently targeted for immune-based therapies.16,22 Ex vivo profiling of EBV-specific T-cell responses, however, revealed that patients with HL and NPC might have impaired LMP1 and LMP2-specific T-cell function at the time of diagnosis, which rebounds to normal levels following tumor regression (M Gandhi and R Khanna, unpublished data). Although the precise mechanism for the loss of this T-cell function is unknown, there is some indication that the expansion of tumor cells is coincident with the emergence of immunoregulatory T cells that express high levels of CTLA4 (M Gandhi and R Khanna, unpublished data). Furthermore, in vivo analysis of tumor biopsies shows that malignant cells in HL and NPC express immunosuppressive cytokines, for example interleukins IL-10 and IL-13, transforming growth factor-beta (TGF-β) and thymus and activation-regulated chemokine, which might be induced by the activation of certain transcriptional factors, such as the signal transducer and activator of transcription STAT3.24
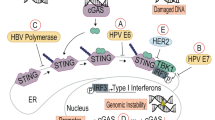
Apart from using restricted viral gene expression, immune-based therapies are also constrained by the immunomodulatory effects of certain EBV proteins expressed in malignant cells. These EBV proteins act at the level of antigen processing and presentation, cytokine regulation, regulation of adhesion molecule expression, and T-cell recognition25,26,27 (Figure 3). A classic example is the modulation of innate and adaptive immunity by LMP1. Previous studies have shown that this protein can activate various transcription factors including nuclear factor-kappa B and STAT3, which can induce expression of immunosuppressive cytokines or cause immune tolerance.28,29 Furthermore, LMP1 induces the expression of the DNA-binding zinc finger protein A20, which has recently been shown to inhibit inflammatory responses.30 Similarly, LMP2 is known to inhibit B-cell receptor signaling and to promote tumor cell survival.31,32 The glycine-alanine amino acid repeat within EBNA1 stabilizes the mature protein by preventing its degradation by the proteasome; this is in contrast to its previously assumed role as an immune evasion domain.33,34 This repeat also acts as a cis inhibitor of mRNA translation and thus limits the availability of EBNA1 for immune recognition.35
(A) The LMP1 transmembrane oncogenic protein contains three major domains: first, an amino-terminal cytoplasmic tail, which binds LMP1 to the plasma membrane; second, six hydrophobic transmembrane domains, which are involved in self-aggregation and oligomerization; third, a long carboxy-terminal cytoplasmic region, which contains the signaling activity of the protein. Two distinct functional domains referred to as C-terminal activation regions 1 and 2 (CTAR1 and CTAR2) have been identified on the basis of their ability to activate NF-κB, STAT3, PI3K and induce expression of the A20 protein. The activation of these transcription factors and other mediators has direct impact on the innate and adaptive immunity. (B) The Gly-Arg-rich domain has been shown to mediate homotypic interactions at a distance between DNA-bound EBNA1 molecules, as well as heterotypic interactions with cellular proteins. The Gly-Ala repeat domain is known to block mRNA transcription and proteasomal degradation of EBNA1. The core domain contains an eight-stranded antiparallel β-barrel, which forms the DNA binding and dimerization interface, and two α-helices per monomer. The extreme carboxyl terminus of EBNA1 is highly acidic. This acidic tail has been reported to have roles both in transactivation and in oriP plasmid maintenance. (C) The LMP2A protein includes 12 transmembrane domains, a 27-amino-acid cytoplasmic carboxyl terminus and a 119-amino-acid cytoplasmic amino-terminal domain that contains 8 tyrosine residues, 2 of which (Tyr74 and Tyr85) form an immunoreceptor tyrosine-based activation motif (ITAM). The LMP2A ITAM blocks signaling from the B-cell receptor. LMP2A also induces activation of various kinases including Ras, Akt and PI3K, which promote cell survival. CCL 17 and 22, chemokines 17 and 22; CTAR1 and CTAR2, C-terminal activation regions 1 and 2; EBNA 1, EBV-associated nuclear antigens 1; EBV, Epstein–Barr virus; Gly-Ala, glycine-alanine; Gly-Arg, glycine-arginine; LMP1 and 2A, latent membrane proteins 1 and 2A; mRNA, messenger RNA; NF-κB, nuclear factor-kappa B; NK, natural killer; NLS, nuclear localization signal; PI3K, phosphatidylinositol 3-kinase; STAT3, signal transducer and activator of transcription 3; TAP1 and 2, transporters 1 and 2; Th2, T-helper cell 2.
Modifying the tumor microenvironment to enhance immune-based therapies
It is now well established that tumor growth is critically dependent on a microenvironment that is resistant to immune control.36 There have been a number of attempts to modify the tumor microenvironment to promote immune recognition. These attempts include the use of pharmacological and/or gene therapy constructs designed to express either cytotoxic or inhibitory proteins selectively in tumor cells. Examples to date include treatment with the CD40 ligand to induce the expression of antigen-processing genes,37 drug-induced demethylation of the EBV genome in order to reverse expression of viral latent proteins,38 transduction of dominant negative forms of STAT3 and/or A20,28,29 and treatment with cytokines/chemokines, which induce differentiation of malignant cells (Table 2). Another approach is based on specific targeting of LMP1 and EBNA1 using N-end rule targeting (i.e. the in vivo half-life of a protein that relates to its N-terminal residue) or E3 ubiquitin-protein ligases to direct degradation of these proteins.39,40,41
Emerging cell-based therapies for treatment of ebv-associated malignancies
The effectiveness of T-cell-based immunotherapy for the prevention and treatment of a range of EBV-associated malignancies necessitates its wider application, perhaps in combination with chemotherapy and monoclonal antibody therapies. ADOPTIVE IMMUNOTHERAPY involves the ex vivo activation of EBV-specific T cells, thus circumventing in vivo host immune-suppressive mechanisms, to permit the expansion of large numbers of virus-specific CTLs for administration to the autologous host. A major constraint of the wider application of autologous CTL-based therapy is the delay between diagnosis and the preparation of a therapeutic dose of T cells (Table 2). To overcome this limitation, banks of cryopreserved, haploidentical EBV-specific CTLs derived from healthy seropositive subjects can be established.42 This strategy has proven clinical efficacy13 (Figure 4) and offers a distinct logistic advantage of speed of access and ease of generation that might permit its wide-scale use in treating both PTL and other malignancies, such as HL and NPC. This may be particularly relevant for developing countries where T cells could be transported to remote locations in which access to standard chemo/radiotherapy is restricted. Most importantly, infusion of haploidentical allogeneic CTL15,17 is associated with low rates of graft-versus-host disease, suggesting that prolonged in vitro culture diminishes alloreactivity.
Two broad strategies are currently being explored for the expansion of EBV-specific CTLs. (A) PBMC from the patient or from a healthy seropositive haploidentical donor are stimulated with autologous EBV-transformed LCLs for 3–4 weeks in the presence of recombinant interleukin-2 (IL-2). (B) Alternatively, PBMC from the patient or from a healthy seropositive haploidentical donor are stimulated with dendritic cells transduced with recombinant viral vectors encoding either full-length EBV protein or CTL epitopes linked together as a polyepitope. Following stimulation, these expanded T cells are assessed for antigen specificity and allospecific reactivity. CTLs generated from approach A or B can be either immediately transferred into the patient or stored as part of an EBV CTL bank for future adoptive immunotherapy. The establishment of T-cell banks from haploidentical healthy donors allows rapid access to the T cells for the treatment of EBV-associated malignancies. These CTLs can be used to treat malignancies with a full complement of EBV latent proteins (e.g. PTL and HIV-associated lymphomas). In the case of HL, NPC and T/NK lymphomas, however, the in vivo efficacy of these effectors cells can be enhanced significantly by transduction with dominant negative TGF-β receptor or depletion of regulatory T cells by treating patients with anti-CD25 and anti-CD45 mediated cytoreduction prior to adoptive immunotherapy. For in vivo tracking, the effector cells can be transduced with a neomycin gene marker. CTL, cytotoxic T lymphocyte; EBV, Epstein–Barr virus; HL, Hodgkin's lymphoma; LCL, lymphoblastoid cell lines; NPC, nasopharyngeal carcinoma; PBMC, peripheral blood mononuclear cells; PTL, post-transplant lymphoma; T/NK lymphomas, T-cell and natural killer lymphomas.
Another constraint for the widespread use of autologous or allogeneic CTLs, especially where latent EBV gene expression is limited, is the antigen specificity. Recent studies indicate the potential use of recombinant viral vectors expressing individual latent antigens,43,44 or T-cell epitopes linked as a POLYEPITOPE45 in combination with autologous dendritic cells, to expand T cells of the relevant specificity (Figure 5). Indeed, a clinical study aimed at expanding LMP2A-specific CTLs using autologous dendritic cells infected with a recombinant adenovirus encoding this antigen has been initiated for the treatment of relapsed HL (Table 1).
Three different vaccine formulation strategies based on recombinant viral vectors or plasmid DNA expressing full-length EBV antigens, CTL epitopes linked together as a polyepitope, or scrambled antigen vaccines, are currently being tested for the treatment of EBV-associated malignancies. Each of these vaccine strategies is designed to incorporate relevant EBV proteins in such a way that they induce a strong antigen-specific CTL response. (i) Preclinical testing and (ii) in vitro immunogenicity analysis has demonstrated the potential of these approaches to induce EBV-specific CTL responses. To counter tumor-induced immune evasion strategies such as the expression of immunosuppressive cytokines, the presence of regulatory T cells and/or loss of antigen processing function, co-delivery of immune-potentiating cytokines and specific targeting of recombinant antigens through the MHC class I/II pathways may be required to enhance the efficacy of these vaccines. These formulations could be delivered either indirectly (iii) following in vitro transduction of autologous DCs, either alone or as a component of combination therapy as discussed in the text, or directly (iv) into the patient. CTL, cytotoxic T lymphocyte; DC, dendritic cells; EBV, Epstein–Barr virus; IL-15, interleukin-15; PBMC, peripheral blood mononuclear cells.
Another significant challenge is presented by the in vivo efficacy of adoptively transferred CTLs in the remit of tumor-induced immune evasion strategies. This barrier might be reversed by the transduction of EBV-specific CTLs with a dominant negative TGF-β receptor46 or depletion of regulatory T cells by treating patients with anti-CD25 (Table 2 and Figure 4). Alternatively, the blockade of tumor and microenvironment-secreted cytokines such as IL-10, IL-13 and TGF-β might be an effective approach to the enhancement of anti-tumor immunity.47 It has also been proposed that anti-CD45-mediated cytoreduction prior to adoptive immunotherapy can facilitate rapid expansion of EBV-specific CTLs48 (Table 2 and Figure 5).
Much of the optimism about T-cell-based therapies is because of the measurement of surrogate markers, which are regulated in response to a reduction of viral load and/or reconstitution of EBV-specific T-cell immunity. An objective analysis of disease outcome is, however, less impressive, highlighting the need to explore alternative strategies for circumventing tumor-evasion mechanisms that preclude the development of a robust anti-tumor response (Table 2). Recent data from large patient cohort studies of EBV-associated malignancies provide ample evidence that nontargeted intensive chemotherapy is associated with short-term and long-term adverse effects7, and that responses to monoclonal antibody therapy are frequently not sustained,49 most likely owing to the selection of tumor escape mutants. Triple combination therapy comprising chemoradiotherapy, monoclonal antibodies and T-cell adoptive immunotherapy, aims to augment efficacy through the synergistic actions of multi-modality agents, while reducing treatment-related toxicities and the risk of tumor escape. Low-intensity chemotherapy not only debulks the tumor but potentially improves the host's-immune environment by eliminating regulatory T cells50 and providing space within the lymphocyte compartment to facilitate the reconstitution of virus-specific T cells.51 Triple combination therapy for EBV-associated malignancies could serve as a new paradigm for the successful multi-modality treatment of malignancies.
Emerging vaccine strategies for treatment of EBV-associated malignancies
One of the most important outcomes of adoptive immunotherapy has been the demonstration that CD8+ EBV-specific CTLs alone can provide therapeutic benefit by reversing the growth of EBV-associated malignancies (Table 1). As the success of these therapies becomes established there will be an increasing demand for their application in general clinical practice; however, the requirement for specialized facilities, as well as regulatory constraints for these therapies, is likely to preclude their widespread use, particularly in developing countries, unless new co-operative approaches be adopted. One option is national or international tissue banks of cryopreserved GMP grade T-cell lines, perhaps administered along the lines of current international registries such as those used in the field of bone marrow transplantation. Reconstitution of CD8+ T-cell immunity using low-cost prophylactic/therapeutic vaccine formulations incorporating immunogenic determinants are likely to overcome these limitations. Indeed, preclinical studies based on recombinant viral vectors or plasmid DNA expressing either full-length EBV antigens or CTL epitopes linked together as a polyepitope, have shown encouraging results44,45,52 (Figure 5). Another technology currently under development is scrambled antigen vaccines (SAVINE). This technology is designed to incorporate each of the relevant EBV proteins encoded as overlapping peptides, rearranged and rejoined in such a way that potential CTL epitopes are retained53 for safer vaccine delivery. As in the case of adoptive immunotherapy, this vaccination strategy will need to counter tumor-induced immune evasion strategies, such as the expression of immunosuppressive cytokines, the presence of regulatory T cells and/or loss of antigen-processing function. Co-delivery of immune-potentiating cytokines and specific targeting of recombinant antigens through the MHC class I/II pathways is likely to enhance the efficacy of these vaccines.54 These formulations could be delivered either directly or following in vitro transduction of autologous dendritic cells (Figure 5), alone or as a component of combination therapy.
Conclusion
Despite major clinical and scientific advances within the discipline, the aim of routinely supplementing pharmacologic strategies with immune cell-based therapies remains an elusive goal. As scientific advances are made, so new challenges for laboratories developing novel therapies emerge. EBV-associated malignancies could serve as a new prototype for the successful multi-modality treatment of malignancies (T-cell immunotherapy in combination with monoclonal antibodies and chemotherapy). Among the key challenges facing researchers attempting to pioneer such strategies are the regulatory issues involved in immunotherapy. Too often, regulatory authorities apply restrictive criteria based on a model of product development that is better suited to the drug industry than to the academic research institute (i.e. a patented commercial product to be manufactured by a company). For the future benefit of patients with EBV-associated malignancies, it is vital that there is regulatory understanding so that there can be continued academic-led interventional research into this evolving field.
Review criteria
Data for this review were identified by searching the PubMed database and references from relevant articles. Numerous articles were identified through searches of the authors' files. Search terms were “Epstein Barr virus”, “renal transplant”, “lung transplant”, “heart transplant”, “liver transplant”, “bone marrow transplant”, “immunotherapy”, “CTL” and “immunity”. Only English language papers were reviewed. No time limits for these searches were applied.
References
Young LS and Rickinson AB (2004) Epstein-Barr virus: 40 years on. Nat Rev Cancer 4: 757–768
Feng BJ et al. (2002) Genome-wide scan for familial nasopharyngeal carcinoma reveals evidence of linkage to chromosome 4. Nat Genet 31: 395–399
Burrows JM et al. (2004) Selection pressure-driven evolution of the Epstein-Barr virus-encoded oncogene LMP1 in virus isolates from Southeast Asia. J Virol 78: 7131–7137
Ho JH et al. (1978) Salted fish and nasopharyngeal carcinoma in southern Chinese. Lancet 2: 626
Rickinson AB and Moss DJ (1997) Human cytotoxic T lymphocyte responses to Epstein-Barr virus infection. Annu Rev Immunol 15: 405–431
Khanna R and Burrows SR (2000) Role of cytotoxic T lymphocytes in Epstein-Barr virus-associated diseases. Annu Rev Microbiol 54: 19–48
Gandhi MK et al. (2004) Epstein-Barr virus-associated Hodgkin's lymphoma. Br J Haematol 125: 267–281
Lin JC et al. (2003) Phase III study of concurrent chemoradiotherapy versus radiotherapy alone for advanced nasopharyngeal carcinoma: positive effect on overall and progression-free survival. J Clin Oncol 21: 631–637
Poppema S et al. (1999) Immune reactions in classical Hodgkin's lymphoma. Semin Hematol 36: 253–259
Maggio E et al. (2002) Chemokines, cytokines and their receptors in Hodgkin's lymphoma cell lines and tissues. Ann Oncol 13 (Suppl 1): 52–56
Rooney CM et al. (1998) Infusion of cytotoxic T cells for the prevention and treatment of Epstein-Barr virus-induced lymphoma in allogeneic transplant recipients. Blood 92: 1549–1555
Comoli P et al. (2002) Infusion of autologous Epstein-Barr virus (EBV)-specific cytotoxic T cells for prevention of EBV-related lymphoproliferative disorder in solid organ transplant recipients with evidence of active virus replication. Blood 99: 2592–2598
Papadopoulos EB et al. (1994) Infusions of donor leukocytes to treat Epstein-Barr virus-associated lymphoproliferative disorders after allogeneic bone marrow transplantation. N Engl J Med 330: 1185–1191
Khanna R et al. (1999) Activation and adoptive transfer of Epstein-Barr virus-specific cytotoxic T cells in solid organ transplant patients with posttransplant lymphoproliferative disease. Proc Natl Acad Sci USA 96: 10391–10396
Haque T et al. (2002) Treatment of Epstein-Barr-virus-positive post-transplantation lymphoproliferative disease with partly HLA-matched allogeneic cytotoxic T cells. Lancet 360: 436–442
Bollard CM et al. (2004) Cytotoxic T lymphocyte therapy for Epstein-Barr virus+ Hodgkin's Disease. J Exp Med 200:1623–1633
Lucas KG et al. (2004) Adoptive immunotherapy with allogeneic Epstein-Barr virus (EBV)-specific cytotoxic T-lymphocytes for recurrent, EBV-positive Hodgkin disease. Cancer 100: 1892–1901
Roskrow MA et al. (1998) Epstein-Barr virus (EBV)-specific cytotoxic T lymphocytes for the treatment of patients with EBV-positive relapsed Hodgkin's disease. Blood 91: 2925–2934
Straathof KC et al. (2004) Treatment of Nasopharyngeal Carcinoma with Epstein-Barr Virus-specific T Lymphocytes. Blood [10.1182/fj.07-2975fje]
Comoli P et al. (2004) Adoptive transfer of allogeneic Epstein-Barr virus (EBV)-specific cytotoxic T cells with in vitro antitumor activity boosts LMP2-specific immune response in a patient with EBV-related nasopharyngeal carcinoma. Ann Oncol 15: 113–117
Chua D et al. (2001) Adoptive transfer of autologous Epstein-Barr virus-specific cytotoxic T cells for nasopharyngeal carcinoma. Int J Cancer 94: 73–80
Lin CL et al. (2002) Immunization with Epstein-Barr Virus (EBV) peptide-pulsed dendritic cells induces functional CD8+ T-cell immunity and may lead to tumor regression in patients with EBV-positive nasopharyngeal carcinoma. Cancer Res 62: 6952–6958
Kuppers R (2003) B cells under influence: transformation of B cells by Epstein-Barr virus. Nat Rev Immunol 3: 801–812
Chen H et al. (2001) Linkage between STAT regulation and Epstein-Barr virus gene expression in tumors. J Virol 75: 2929–2937
Spriggs MK (1996) One step ahead of the game: viral immunomodulatory molecules. Annu Rev Immunol 14: 101–130
Spriggs MK et al. (1996) The extracellular domain of the Epstein-Barr virus BZLF2 protein binds the HLA-DR beta chain and inhibits antigen presentation. J Virol 70: 5557–5563
Khanna R et al. (1995) Immune regulation in Epstein-Barr virus-associated diseases. Microbiol Rev 59: 387–405
Wang T et al. (2004) Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat Med 10: 48–54
Cheng F et al. (2003) A critical role for Stat3 signaling in immune tolerance. Immunity 19: 425–436
Eliopoulos AG et al. (1997) Epstein-Barr virus-encoded LMP1 and CD40 mediate IL-6 production in epithelial cells via an NF-κB pathway involving TNF receptor-associated factors. Oncogene 14: 2899–2916
Portis T and Longnecker R (2004) Epstein-Barr virus (EBV) LMP2A mediates B-lymphocyte survival through constitutive activation of the Ras/PI3K/Akt pathway. Oncogene 23: 8619–8628
Portis T et al. (2002) The LMP2A signalosome—a therapeutic target for Epstein-Barr virus latency and associated disease. Front Biosci 7: d414–426
Lee SP et al. (2004) CD8 T cell recognition of endogenously expressed epstein-barr virus nuclear antigen 1. J Exp Med 199: 1409–1420
Tellam J et al. (2004) Endogenous presentation of CD8+ T Cell epitopes from Epstein-Barr virus-encoded nuclear antigen 1. J Exp Med 199: 1421–1431
Yin Y et al. (2003) Self-inhibition of synthesis and antigen presentation by Epstein-Barr virus-encoded EBNA1. Science 301: 1371–1374
Mapara MY and Sykes M (2004) Tolerance and cancer: mechanisms of tumor evasion and strategies for breaking tolerance. J Clin Oncol 22: 1136–1151
Khanna R et al. (1997) Engagement of CD40 antigen with soluble CD40 ligand up-regulates peptide transporter expression and restores endogenous processing function in Burkitt's lymphoma cells. J Immunol 159: 5782–5785
Chan AT et al. (2004) Azacitidine induces demethylation of the Epstein-Barr virus genome in tumors. J Clin Oncol 22: 1373–1381
Zhou P et al. (2000) Harnessing the ubiquitination machinery to target the degradation of specific cellular proteins. Mol Cell 6: 751–756
Tellam J et al. (2003) Proteasomal targeting of a viral oncogene abrogates oncogenic phenotype and enhances immunogenicity. Blood 102: 4535–4540
Tellam J et al. (2001) Targeting of EBNA1 for rapid intracellular degradation overrides the inhibitory effects of the Gly-Ala repeat domain and restores CD8+ T cell recognition. J Biol Chem 276: 33353–33360
Wilkie GM et al. (2004) Establishment and characterization of a bank of cytotoxic T lymphocytes for immunotherapy of Epstein-Barr virus-associated diseases. J Immunother 27: 309–316
Wagner HJ et al. (2003) Expansion of EBV latent membrane protein 2a specific cytotoxic T cells for the adoptive immunotherapy of EBV latency type 2 malignancies: influence of recombinant IL12 and IL15. Cytotherapy 5: 231–240
Taylor GS et al. (2004) Dual stimulation of Epstein-Barr Virus (EBV)-specific CD4+- and CD8+-T-cell responses by a chimeric antigen construct: potential therapeutic vaccine for EBV-positive nasopharyngeal carcinoma. J Virol 78: 768–778
Duraiswamy J et al. (2004) Induction of therapeutic T-cell responses to subdominant tumor-associated viral oncogene after immunization with replication-incompetent polyepitope adenovirus vaccine. Cancer Res 64: 1483–1489
Bollard CM et al. (2002) Adapting a transforming growth factor beta-related tumor protection strategy to enhance antitumor immunity. Blood 99: 3179–3187
Zagury D and Gallo RC (2004) Anti-cytokine Ab immune therapy: present status and perspectives. Drug Discov Today 9: 72–81
Wulf GG et al. (2003) Anti-CD45-mediated cytoreduction to facilitate allogeneic stem cell transplantation. Blood 101: 2434–2439
Milpied N et al. (2000) Humanized anti-CD20 monoclonal antibody (Rituximab) in post transplant B-lymphoproliferative disorder: a retrospective analysis on 32 patients. Ann Oncol 11 (Suppl 1): S113–S116
Shimizu J et al. (1999) Induction of tumor immunity by removing CD25+CD4+ T cells: a common basis between tumor immunity and autoimmunity. J Immunol 163: 5211–5218
Maine GN and Mule JJ (2002) Making room for T cells. J Clin Invest 110: 157–159
Duraiswamy J et al. (2003) Therapeutic LMP1 polyepitope vaccine for EBV-associated Hodgkin disease and nasopharyngeal carcinoma. Blood 101: 3150–3156
Thomson SA and Ramshaw IA (2004) Synthetic peptides and uses therefore. Canberra: Australian National University. US Patent No: 20040054137AI
Thomson SA et al. (1998) Targeting a polyepitope protein incorporating multiple class II-restricted viral epitopes to the secretory/endocytic pathway facilitates immune recognition by CD4+ cytotoxic T lymphocytes: a novel approach to vaccine design. J Virol 72: 2246–2252
Acknowledgements
The authors would like to thank Dr Cliona Rooney and Dr Catherine Bollard for sharing their unpublished data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- CYTOTOXIC T LYMPHOCYTES (CTLs)
-
T lymphocytes that have a cytolytic function on target-cell binding; they generally recognize antigenic peptides presented by HLA class I molecules
- EBNA1
-
A protein that regulates the replication and segregation of the viral episomes in latently-infected cells and transactivates the expression of other EBV latent proteins
- ADOPTIVE IMMUNOTHERAPY
-
Ex vivo activation of antigen-specific T cells that circumvent host immune-suppressive mechanisms, allowing expansion of antigen-specific cytotoxic T lymphocytes
- POLYEPITOPE
-
Multiple DNA-encoded epitopes linked without their flanking regions in a synthetic construct, which retain broad-spectrum immunogenicity
Rights and permissions
About this article
Cite this article
Khanna, R., Moss, D. & Gandhi, M. Technology Insight: applications of emerging immunotherapeutic strategies for Epstein–Barr virus-associated malignancies. Nat Rev Clin Oncol 2, 138–149 (2005). https://doi.org/10.1038/ncponc0107
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncponc0107
This article is cited by
-
A practical method to screen and identify functioning biomarkers in nasopharyngeal carcinoma
Scientific Reports (2021)
-
Therapeutic vaccine targeting Epstein-Barr virus latent protein, LMP1, suppresses LMP1-expressing tumor growth and metastasis in vivo
BMC Cancer (2017)
-
Identifying novel genes and chemicals related to nasopharyngeal cancer in a heterogeneous network
Scientific Reports (2016)
-
T-cell therapy in the treatment of post-transplant lymphoproliferative disease
Nature Reviews Clinical Oncology (2012)
-
Methotrexate-associated mantle-cell lymphoma in an elderly man with myasthenia gravis
Nature Clinical Practice Oncology (2008)