Abstract
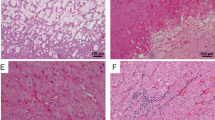
Hereditary papillary renal carcinoma (HPRC) is a recently recognized form of inherited kidney cancer characterized by a predisposition to develop multiple, bilateral papillary renal tumours1–4. The pattern of inheritance of HPRC is consistent with autosomal dominant transmission with reduced penetrance. HPRC is histologically and genetically distinct from two other causes of inherited renal carcinoma, von Hippel-Lindau disease (VHL) and the chromosome translocation (3;8)5–6 Malignant papillary renal carcinomas are characterized by trisomy of chromosomes 7,16 and 17, and in men, by loss of the Y chromosome7. Inherited and sporadic clear cell renal carcinomas are characterized by inactivation of both copies of the VHL gene by mutation, and/or by hypermethylation8–11. We found that the HPRC gene was located at chromosome 7q31.1-34 in a 27-centimorgan (cM) interval between D7S496 and D757537. We identified missense mutations located in the tyrosine kinase domain of the MET gene in the germline of affected members of HPRC families and in a subset of sporadic papillary renal carcinomas. Three mutations in the MET gene are located in codons that are homologous to those in c-kit and RET, proto-oncogenes that are targets of naturally-occurring mutations. The results suggest that missense mutations located in the MET proto-oncogene lead to constitutive activation of the MET protein and papillary renal carcinomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Zbar, B. et al. Hereditary papillary renal carcinoma. J. Urol. 151, 561–566, (1994).
Zbar, B. et al. Hereditary papillary renal cell carcinoma: clinical studies in 10 families. J. Urol. 153, 907–912 (1995).
Bernades, P. et al. Cancer du rein gauche chez trois membres d'une meme fratrie. Sem.Hop. Paris 48, 2813–2818 (1972).
Bernues, M. et al. Cytogenetic characterization of a familial papillary renal cell carcinoma. Cancer Genet, and Cytogenet. 84, 123–127 (1995).
Melman, K.L. & Rosen, S.W. Lindau's disease: review of the literature and study of a large kindred. Am. J. Med. 36, 595–617 (1964).
Cohen, A.J. et al. Hereditary renal-cell carcinoma associated with a chromosomal translocation. NewEngl. J. Med. 301, 592–595 (1979).
Kovacs, G. Molecular cytogenetics of renal tumours. Adv. Cancer Res. 62, 89–124 (1993).
Latif, F. et al. Identification of the von Hippel-Lindau disease tumour suppressor gene. Science 260, 1317–1320 (1993).
Schmidt, L. et al. Mechanism of tumourigenesis of renal carcinomas associated with the constitutional chromosome 3;8 translocation. Cancer J. from Sci. Am. 1, 191–196 (1995).
Gnarra, J.R. et al. Mutations of the VHL tumour suppressor gene in renal carcinoma. Nature Genet. 7, 85–90 (1994).
Herman, J.G. et al. Silencing of the VHL tumour suppressor gene by DNA methylation in renal carcinoma. Proc. Nat. Acad. Sci. USA 91, 9700–9704 (1994).
Bergerheim, U.S.R. et al. Chromosomal gains and losses in sporadic and hereditary papillary renal carcinomas by comparative genomic hybridization. J. Urol. 155, 543A(1996).
Cooper, C.S. et al. Molecular cloning of a new transforming gene from a chemically transformed human cell line. Nature 311, 29–33 (1984).
Park, M. et al. Mechanism of met oncogene activation. Cell 45, 895–904 (1986).
Wainwright, B.J. et al. Isolation of a human gene with protein sequence similarity to human and murine int-1 and the Drosophila segment polarity mutant wingless. EMBO J. 7, 1743–1748 (1988).
Nusse, R., SVarmus, H.R. Wnt genes. Cell 69, 1073–1087 (1992).
Stuart, E.T. & Gruss, P. PAX: Developmental control genes in cell growth and differentiation. Cell Growth Differ. 7, 405–412 (1996).
LM.et al. Germ-line mutations of the RET proto-oncogene in multiple endocrine neoplasia type 2A. Nature 363, 458–460 (1993).
Donis-Keller, H. et al. Mutations in the RET proto-oncogene associated with MEN 2A and FMTC. Hum. Mol. Genet 2, 851–856 (1993).
Hofstra, R.M. et al. A mutation in the RET proto-oncogene associated with multiple endocrine neoplasia type 2B and sporadic medullary thyroid carcinoma. Nature 367, 375–376 (1994).
Piao, S. & Bernstein, A. A point mutation in the catalytic domain of c-kit induces growth factor independence, tumourigenicity and differentiation of mast cells. Blood 87, 3117–3123 (1996).
Nagata, H. et al. Identification of a point mutation in the catalytic domain of the protooncogene c-kit in peripheral blood mononuclear cells of patients who have mastocytosis with an associated hematologic disorder. Proc. Natl. Acad. Sci. USA 92, 10560–10564 (1995).
Bolino, A. et al. RET mutations in exons 13 and 14 of FMTC patients. Oncogene 10, 2415–2419 (1995).
Eng, C. et al. A novel point mutation in the tyrosine kinase domain of the RET proto-oncogene in sporadic medulllary thyroid carcinoma and in a family with FMTC. Oncogene 10, 509–513 (1995).
Santoro, M. et al. Activation of RET as a dominant transforming gene by germline mutations of MEN2A and MEN2B. Science 267, 381–383 (1995).
Park, M. et al. Sequence of MET protooncogene cONA has features characteristic of the tyrosine kinase family of growth-factor receptors. Proc. Natl. Acad. Sci. USA 84, 6379–6383 (1987).
Weidner, K.M., Sachs, M. & Birchmeier, W., Met receptor tyrosine kinase transduces motility, proliferation and morphogenic signals of scatter factor/hepatocyte growth factor in epithelial cells. J. Cell Biol. 121, 145–154, (1993).
Bladt, F., Riethmacher, D., Isenmann, S., Sguzzi, A. & Birchmeier, C. Essential role for the c-met receptor in the migration of myogenic precursor cells into the limb bud. Nature 376, 768–771 (1995).
Ponzetto, C. et al. c-met is amplified but not mutated in a cell line with an activated met tyrosine kinase. Oncogene 6, 553–559 (1991).
Dl Renzo, M. et al. Overexpression of the c-MET/HGF receptor gene in human thyroid carcinomas. Oncogene 7, 2549–2553 (1992).
Bremner, R. & Balmain, A. Genetic changes in skin tumour progression: correlation between presence of a mutant ras gene and loss of heterozygosity on mouse chromosome 7. Cell 61, 407–417 (1990).
Wirchubsky, Z., Wiener, F., Spira, J., Sumegi, J. & Klein, G. Triplication of one chromosome no 15 with an altered c-myc homologue in a T-cell lymphoma line of AKR origin (TIKAUT). Int. J. Cancer 33, 477–481 (1984).
Bianchi, A.B., Alkaz, C.M. & Conti, C.J. Nonrandom duplication of the chromosome bearing a mutated Ha-ras-1 allele in mouse skin tumours. Proc. Natl. Acad. Sci. USA 87, 6902–6906 (1990).
Meloni, A.M., Dobbs, R.M., Pontes, J.E. & Sandberg, A.A. Translocation [X;1] in papillary renal adenocarcinomaA new Cytogenetic subtype. Cancer Genet. Cytogenet. 65, 1–6 (1993).
Sidhar, S.K. et al. The t(X;1)(p11.2;q21.2) translocation in papillary renal cell carcinoma fuses a novel gene PRCC to the TFE3 transcription factor gene. Hum Mol. Genet. 5, 1333–1338 (1996).
Weterman, M.A.J., Wilbrink, M. & van Kessel, A.G. Fusion of the transcription factor TFE3 gene to a novel gene, PRCC, in t(X;1)(p11;q21)-positive papillary renal cell carcinoma. Proc. Natl. Acad. Sci. USA 93, 15294–15298 (1996).
Choyke, P.L. et al. Imaging features of hereditary papillary renal carcinoma. J. Computer Assist. Tomography (in the press).
Cottingham, R.W. Jr., Idury, R.M. & Schaffer, A.A. Faster sequential genetic linkage computations. Amer. J. Human Genet. 53, 252–263 (1993).
O'Connell, J.R.D.E. The VITESSE algorithm for rapid exact multilocus linkage analysis via genotype set-recording and fuzzy inheritance. Nature Genet. 11, 402–408 (1995).
Weier, H.-U.G., Kleine, H.-O. & Gray, J.W. Labeling of the centromeric region on human chromosome 8 by in situ hybridization. Hum. Genet. 87, 489–494 (1991).
Takahashi, M., Buma, Y. & Hiai, H. Isolation of ret proto-oncogene cDNA with an amino-terminal signal sequence. Oncogene 4, 805–806 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schmidt, L., Duh, FM., Chen, F. et al. Germline and somatic mutations in the tyrosine kinase domain of the MET proto-oncogene in papillary renal carcinomas. Nat Genet 16, 68–73 (1997). https://doi.org/10.1038/ng0597-68
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0597-68
This article is cited by
-
Oncogenic alterations in advanced NSCLC: a molecular super-highway
Biomarker Research (2024)
-
Metabolic alterations in hereditary and sporadic renal cell carcinoma
Nature Reviews Nephrology (2024)
-
Inhibition of HSP 90 is associated with potent anti-tumor activity in Papillary Renal Cell Carcinoma
Journal of Experimental & Clinical Cancer Research (2022)
-
Anti-cancer therapeutic strategies based on HGF/MET, EpCAM, and tumor-stromal cross talk
Cancer Cell International (2022)
-
hOA-DN30: a highly effective humanized single-arm MET antibody inducing remission of ‘MET-addicted’ cancers
Journal of Experimental & Clinical Cancer Research (2022)