Abstract
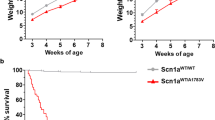
Succinate semialdehyde dehydrogenase (ALDH5A1, encoding SSADH deficiency is a defect of 4-aminobutyric acid (GABA) degradation that manifests in humans as 4-hydroxybutyric (gamma-hydroxybutyric, GHB) aciduria. It is characterized by a non-specific neurological disorder including psychomotor retardation, language delay, seizures, hypotonia and ataxia. The current therapy, vigabatrin (VGB), is not uniformly successful1. Here we report the development of Aldh5a1-deficient mice. At postnatal day 16–22 Aldh5a1−/− mice display ataxia and develop generalized seizures leading to rapid death. We observed increased amounts of GHB and total GABA in urine, brain and liver homogenates and detected significant gliosis in the hippocampus of Aldh5a1−/− mice. We found therapeutic intervention with phenobarbital or phenytoin ineffective, whereas intervention with vigabatrin or the GABAB receptor antagonist CGP 35348 (ref. 2) prevented tonic-clonic convulsions and significantly enhanced survival of the mutant mice. Because neurologic deterioration coincided with weaning, we hypothesized the presence of a protective compound in breast milk. Indeed, treatment of mutant mice with the amino acid taurine rescued Aldh5a1−/− mice. These findings provide insight into pathomechanisms and may have therapeutic relevance for the human SSADH deficiency disease and GHB overdose and toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gibson, K.M. & Jakobs, C. Disorders of beta- and gamma amino acids in free and peptide-linked forms. in The metabolic and molecular bases of inherited disease 8th edn (eds Scriver, C.R., Beaudet, A.L., Sly, W.S. & Valle, D.) 2079–2105 (McGraw-Hill, New York, 2001).
Olpe, H.R. et al. CGP 35348: a centrally active blocker of GABAB receptors. Eur. J. Pharmacol. 187, 27–38 (1990).
Eng, L.F., Yu, A.C.H. & Lee, Y.L. Astrocytic response to injury. Prog. Brain Res. 94, 353–365 (1992).
Landis, D.M.D. The early reactions of non-neuronal cells to brain injury. Annu. Rev. Neurosci. 17, 133–151 (1994).
Rizvi, T.A. et al. Region-specific astrogliosis in brains of mice heterozygous for mutations in the neurofibromatosis type 1 (Nf1) tumor suppressor. Brain Res. 816, 111–123 (1999).
Miller, D.B., Bartke, A. & O'Callaghan, J.P. Increased glial fibrillary acidic protein (GFAP) levels in the brains of transgenic mice expressing the bovine growth hormone (bGH) gene. Exp. Gerontol. 30, 383–400 (1995).
Minn, A., Schubert, M., Neiss, W.F. & Muller-Hill, B. Enhanced GFAP expression in astrocytes of transgenic mice expressing the human brain-specific trypsinogen IV. Glia 22, 338–347 (1998).
Nitecka, L. et al. Maturation of kainic acid seizure-brain damage syndrome in the rat. II. Histopathological sequelae. Neuroscience 13, 1073–1094 (1984).
Fernandes, M.J., Dube, C., Boyet, S., Marescaux, C. & Nehlig, A. Correlation between hypermetabolism and neuronal damage during status epilepticus induced by lithium and pilocarpine in immature and adult rats. J. Cereb. Blood Flow Metab. 19, 195–209. (1999).
Gibson, K.M. et al. Vigabatrin therapy in six patients with succinic semialdehyde dehydrogenase deficiency. J. Inherit. Metab. Dis. 18, 143–146 (1995).
Hardus, P. et al. Visual field loss associated with vigabatrin: quantification and relation to dosage. Epilepsia 42, 262–267. (2001).
Mathivet, P., Bernasconi, R., De Barry, J., Marescaux, C. & Bittiger, H. Binding characteristics of gamma-hydroxybutyric acid as a weak but selective GABAB receptor agonist. Eur. J. Pharmacol. 321, 67–75 (1997).
Erhardt, S., Andersson, B., Nissbrandt, H. & Engberg, G. Inhibition of firing rate and changes in the firing pattern of nigral dopamine neurons by gamma-hydroxybutyric acid (GHBA) are specifically induced by activation of GABAB receptors. Naunyn Schmiedebergs Arch. Pharmacol. 357, 611–619 (1998).
Motalli, R. et al. GABAB receptor activation promotes seizure activity in the juvenile rat hippocampus. J. Neurophysiol. 82, 638–647 (1999).
Benavides, J. et al. High affinity binding sites for gamma-hydroxybutyric acid in rat brain. Life Sci. 30, 953–961 (1982).
Snead, O.C. & Liu, C.C. Gamma-hydroxybutyric acid binding sites in rat and human brain synaptosomal membranes. Biochem. Pharmacol. 33, 2587–2590 (1984).
Kaupmann, K. et al. GABAB-receptor subtypes assemble into functional heteromeric complexes. Nature 396, 683–687 (1998).
Jones, K.A. et al. GABAB receptors function as a heteromeric assembly of the subunits GABABR1 and GABABR2. Nature 396, 674–679 (1998).
Lingenhoehl, K. et al. Gamma-hydroxybutyrate is a weak agonist at recombinant GABAB receptors. Neuropharmacology 38, 1667–1673 (1999).
Diana, M. et al. Low doses of gamma-hydroxybutyric acid stimulate the firing rate of dopaminergic neurons in unanesthetized rats. Brain Res. 566, 208–211 (1991).
Snead, O.C. The ontogeny of [3H]gamma-hydroxybutyrate and [3H]GABAB binding sites: relation to the development of experimental absence seizures. Brain Res. 659, 147–156 (1994).
Sarwar, G., Botting, H.G., Davis, T.A., Darling, P. & Pencharz, P.B. Free amino acids in milks of human subjects, other primates and non-primates. Br. J. Nutr. 79, 129–131 (1998).
Saransaari, P. & Oja, S.S. Taurine release modified by GABAergic agents in hippocampal slices from adult and developing mice. Amino Acids 18, 17–30 (2000).
Oja, S.S. & Saransaaria, P. Modulation of taurine release by glutamate receptors and nitric oxide. Prog. Neurobiol. 62, 407–425 (2000).
Flint, A.C., Liu, X. & Kriegstein, A.R. Nonsynaptic glycine receptor activation during early neocortical development. Neuron 20, 43–53 (1998).
Smith, S.S. & Li, J. GABAB receptor stimulation by baclofen and taurine enhances excitatory amino acid induced phosphatidylinositol turnover in neonatal rat cerebellum. Neurosci. Lett. 132, 59–64 (1991).
del Olmo, N., Bustamante, J., del Rio, R.M. & Solis, J.M. Taurine activates GABAA but not GABAB receptors in rat hippocampal CA1 area. Brain Res. 864, 298–307 (2000).
Clements, J.R., Magnusson, K.R. & Beitz, A.J. Ultrastructural description of taurine- like immunoreactive cells and processes in rat hippocampus. Synapse 4, 70–79 (1989).
Timby, N., Eriksson, A. & Bostrom, K. Gamma-hydroxybutyrate associated deaths. Am. J. Med. 108, 518–519 (2000).
Thomas, K.R. & Capecchi, M.R. Site-directed mutagenesis by gene targeting in mouse embryo-derived stem cells. Cell 51, 503–512 (1987).
Gibson, K.M. et al. Stable isotope dilution analysis of 4-hydroxybutyric acid: an accurate method for quantification in physiological fluids and the prenatal diagnosis of 4-hydroxybutyric aciduria. Biomed. Environ. Mass Spectrom. 19, 89–93 (1990).
Acknowledgements
The authors acknowledge the contribution of M. Baetscher and J. Dekoning from the OHSU transgenic core facility in the development of the animal model, M. Al-Dhalimy and M. Noll for assistance with genotyping and animal studies, S. Akaboshi for assistance in characterization of murine seizures, H. Bartels for assistance with behavioral studies and amino acid analysis and C. Fernandez-Canon for providing the pgk-TK vector. The expert technical assistance of W. Guerand, V. Pereira and D. Schor in determination of metabolite levels in tissues and physiologic fluids is gratefully acknowledged. This work was supported in part by Research Grants No. 9813 from the Oregon Health Sciences Foundation (K.M.G.), #1-FY00-352 from the March of Dimes Birth Defects Foundation (K.M.G.), and NS 40270 from the National Institute of Neurological Disorders and Stroke, National Institutes of Health (K.M.G.). R.D.-A. was supported by P30 AG12300 and RO3 AG16450, from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hogema, B., Gupta, M., Senephansiri, H. et al. Pharmacologic rescue of lethal seizures in mice deficient in succinate semialdehyde dehydrogenase. Nat Genet 29, 212–216 (2001). https://doi.org/10.1038/ng727
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng727
This article is cited by
-
Clinical and molecular outcomes from the 5-Year natural history study of SSADH Deficiency, a model metabolic neurodevelopmental disorder
Journal of Neurodevelopmental Disorders (2024)
-
Phenotypic correlates of structural and functional protein impairments resultant from ALDH5A1 variants
Human Genetics (2023)
-
Post-mortem tissue analyses in a patient with succinic semialdehyde dehydrogenase deficiency (SSADHD). I. Metabolomic outcomes
Metabolic Brain Disease (2020)
-
Succinic semialdehyde dehydrogenase deficiency, a disorder of GABA metabolism: an update on pharmacological and enzyme‐replacement therapeutic strategies
Journal of Inherited Metabolic Disease (2018)
-
Metabolism of amino acid neurotransmitters: the synaptic disorder underlying inherited metabolic diseases
Journal of Inherited Metabolic Disease (2018)