Abstract
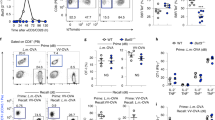
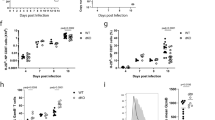
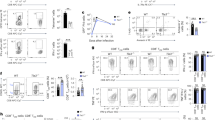
B and T lymphocyte attenuator (BTLA) is a negative regulator of T cell activation, but its function in vivo is not well characterized. Here we show that mice deficient in full-length BTLA or its ligand, herpesvirus entry mediator, had increased number of memory CD8+ T cells. The memory CD8+ T cell phenotype resulted from a T cell–intrinsic perturbation of the CD8+ T cell pool. Naive BTLA-deficient CD8+ T cells were more efficient than wild-type cells at generating memory in a competitive antigen-specific system. This effect was independent of the initial expansion of the responding antigen-specific T cell population. In addition, BTLA negatively regulated antigen-independent homeostatic expansion of CD4+ and CD8+ T cells. These results emphasize two central functions of BTLA in limiting T cell activity in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Min, B. et al. Neonates support lymphopenia-induced proliferation. Immunity 18, 131–140 (2003).
Margolick, J.B. & Donnenberg, A.D. T-cell homeostasis in HIV-1 infection. Semin. Immunol. 9, 381–388 (1997).
Dudley, M.E. et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298, 850–854 (2002).
Goldrath, A.W. & Bevan, M.J. Selecting and maintaining a diverse T-cell repertoire. Nature 402, 255–262 (1999).
Ernst, B., Lee, D.S., Chang, J.M., Sprent, J. & Surh, C.D. The peptide ligands mediating positive selection in the thymus control T cell survival and homeostatic proliferation in the periphery. Immunity 11, 173–181 (1999).
Ge, Q., Rao, V.P., Cho, B.K., Eisen, H.N. & Chen, J. Dependence of lymphopenia-induced T cell proliferation on the abundance of peptide/MHC epitopes and strength of their interaction with T cell receptors. Proc. Natl. Acad. Sci. USA 98, 1728–1733 (2001).
Kennedy, M.K. et al. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15–deficient mice. J. Exp. Med. 191, 771–780 (2000).
Prlic, M., Blazar, B.R., Khoruts, A., Zell, T. & Jameson, S.C. Homeostatic expansion occurs independently of costimulatory signals. J. Immunol. 167, 5664–5668 (2001).
Carreno, B.M. & Collins, M. The B7 family of ligands and its receptors: new pathways for costimulation and inhibition of immune responses. Annu. Rev. Immunol. 20, 29–53 (2002).
Li, O., Zheng, P. & Liu, Y. CD24 expression on T cells is required for optimal T cell proliferation in lymphopenic host. J. Exp. Med. 200, 1083–1089 (2004).
Greenwald, R.J., Freeman, G.J. & Sharpe, A.H. The B7 family revisited. Annu. Rev. Immunol. 23, 515–548 (2005).
Watanabe, N. et al. BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat. Immunol. 4, 670–679 (2003).
Han, P., Goularte, O.D., Rufner, K., Wilkinson, B. & Kaye, J. An inhibitory Ig superfamily protein expressed by lymphocytes and APCs is also an early marker of thymocyte positive selection. J. Immunol. 172, 5931–5939 (2004).
Sedy, J.R. et al. B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator. Nat. Immunol. 6, 90–98 (2005).
Gonzalez, L.C. et al. A coreceptor interaction between the CD28 and TNF receptor family members B and T lymphocyte attenuator and herpesvirus entry mediator. Proc. Natl. Acad. Sci. USA 102, 1116–1121 (2005).
Compaan, D.M. et al. Attenuating lymphocyte activity: the crystal structure of the BTLA-HVEM complex. J. Biol. Chem. 280, 39553–39561 (2005).
Hurchla, M.A. et al. B and T lymphocyte attenuator exhibits structural and expression polymorphisms and is highly Induced in anergic CD4+ T cells. J. Immunol. 174, 3377–3385 (2005).
Otsuki, N., Kamimura, Y., Hashiguchi, M. & Azuma, M. Expression and function of the B and T lymphocyte attenuator (BTLA/CD272) on human T cells. Biochem. Biophys. Res. Commun. 344, 1121–1127 (2006).
Tamada, K. et al. Modulation of T-cell-mediated immunity in tumor and graft-versus-host disease models through the LIGHT co-stimulatory pathway. Nat. Med. 6, 283–289 (2000).
Zhai, Y. et al. LIGHT, a novel ligand for lymphotoxin β receptor and TR2/HVEM induces apoptosis and suppresses in vivo tumor formation via gene transfer. J. Clin. Invest. 102, 1142–1151 (1998).
Mauri, D.N. et al. LIGHT, a new member of the TNF superfamily, and lymphotoxin α are ligands for herpesvirus entry mediator. Immunity 8, 21–30 (1998).
Wang, Y. et al. The role of herpesvirus entry mediator as a negative regulator of T cell-mediated responses. J. Clin. Invest. 115, 711–717 (2005).
Morel, Y. et al. Reciprocal expression of the TNF family receptor herpes virus entry mediator and its ligand LIGHT on activated T cells: LIGHT down-regulates its own receptor. J. Immunol. 165, 4397–4404 (2000).
Cheung, T.C. et al. Evolutionarily divergent herpesviruses modulate T cell activation by targeting the herpesvirus entry mediator cosignaling pathway. Proc. Natl. Acad. Sci. USA 102, 13218–13223 (2005).
Krieg, C., Han, P., Stone, R., Goularte, O.D. & Kaye, J. Functional analysis of B and T lymphocyte attenuator engagement on CD4+ and CD8+ T cells. J. Immunol. 175, 6420–6427 (2005).
Chemnitz, J.M., Lanfranco, A.R., Braunstein, I. & Riley, J.L. B and T lymphocyte attenuator–mediated signal transduction provides a potent inhibitory signal to primary human CD4 T cells that can be initiated by multiple phosphotyrosine motifs. J. Immunol. 176, 6603–6614 (2006).
Deppong, C. et al. Cutting edge: B and T lymphocyte attenuator and programmed death receptor-1 inhibitory receptors are required for termination of acute allergic airway inflammation. J. Immunol. 176, 3909–3913 (2006).
Tao, R. et al. Differential effects of B and T lymphocyte attenuator and programmed death-1 on acceptance of partially versus fully MHC-mismatched cardiac allografts. J. Immunol. 175, 5774–5782 (2005).
Tan, K.B. et al. Characterization of a novel TNF-like ligand and recently described TNF ligand and TNF receptor superfamily genes and their constitutive and inducible expression in hematopoietic and non-hematopoietic cells. Gene 204, 35–46 (1997).
Curtsinger, J.M., Lins, D.C. & Mescher, M.F. CD8+ memory T cells (CD44high, Ly-6C+) are more sensitive than naive cells to (CD44low, Ly-6C−) to TCR/CD8 signaling in response to antigen. J. Immunol. 160, 3236–3243 (1998).
Sprent, J. & Surh, C.D. T cell memory. Annu. Rev. Immunol. 20, 551–579 (2002).
Judge, A.D., Zhang, X., Fujii, H., Surh, C.D. & Sprent, J. Interleukin 15 controls both proliferation and survival of a subset of memory-phenotype CD8+ T cells. J Exp Med 196, 935–946 (2002).
Boyman, O., Cho, J.H., Tan, J.T., Surh, C.D. & Sprent, J. A major histocompatibility complex class I–dependent subset of memory phenotype CD8+ cells. J. Exp. Med. 203, 1817–1825 (2006).
Sallusto, F., Geginat, J. & Lanzavecchia, A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu. Rev. Immunol. 22, 745–763 (2004).
Agarwal, S. & Rao, A. Modulation of chromatin structure regulates cytokine gene expression during T cell differentiation. Immunity 9, 765–775 (1998).
Veiga-Fernandes, H., Walter, U., Bourgeois, C., McLean, A. & Rocha, B. Response of naive and memory CD8+ T cells to antigen stimulation in vivo. Nat. Immunol. 1, 47–53 (2000).
Surh, C.D. & Sprent, J. Regulation of mature T cell homeostasis. Semin. Immunol 17, 183–191 (2005).
Badovinac, V.P., Messingham, K.A., Jabbari, A., Haring, J.S. & Harty, J.T. Accelerated CD8+ T-cell memory and prime-boost response after dendritic-cell vaccination. Nat. Med. 11, 748–756 (2005).
Harty, J.T., Tvinnereim, A.R. & White, D.W. CD8+ T cell effector mechanisms in resistance to infection. Annu Rev Immunol 18, 275–308 (2000).
Shen, H. et al. Compartmentalization of bacterial antigens: differential effects on priming of CD8 T cells and protective immunity. Cell 92, 535–545 (1998).
Tamada, K. et al. Cutting edge: selective impairment of CD8+ T cell function in mice lacking the TNF superfamily member LIGHT. J. Immunol. 168, 4832–4835 (2002).
Sedgmen, B.J., Dawicki, W., Gommerman, J.L., Pfeffer, K. & Watts, T.H. LIGHT is dispensable for CD4+ and CD8+ T cell and antibody responses to influenza A virus in mice. Int. Immunol. 18, 797–806 (2006).
Hou, S., Hyland, L., Ryan, K.W., Portner, A. & Doherty, P.C. Virus-specific CD8+ T-cell memory determined by clonal burst size. Nature 369, 652–654 (1994).
Badovinac, V.P., Porter, B.B. & Harty, J.T. Programmed contraction of CD8+ T cells after infection. Nat. Immunol. 3, 619–626 (2002).
Boyman, O., Kovar, M., Rubinstein, M.P., Surh, C.D. & Sprent, J. Selective stimulation of T cell subsets with antibody-cytokine immune complexes. Science 311, 1924–1927 (2006).
Tan, J.T. et al. IL-7 is critical for homeostatic proliferation and survival of naive T cells. Proc. Natl. Acad. Sci. USA 98, 8732–8737 (2001).
Zhang, X., Sun, S., Hwang, I., Tough, D.F. & Sprent, J. Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity 8, 591–599 (1998).
Vella, A.T., Dow, S., Potter, T.A., Kappler, J. & Marrack, P. Cytokine-induced survival of activated T cells in vitro and in vivo. Proc. Natl. Acad. Sci. USA 95, 3810–3815 (1998).
Kuroda, K. et al. Implantation of IL-2-containing osmotic pump prolongs the survival of superantigen-reactive T cells expanded in mice injected with bacterial superantigen. J. Immunol. 157, 1422–1431 (1996).
Shimizu, K., Fields, R.C., Giedlin, M. & Mule, J.J. Systemic administration of interleukin 2 enhances the therapeutic efficacy of dendritic cell-based tumor vaccines. Proc. Natl. Acad. Sci. USA 96, 2268–2273 (1999).
Blattman, J.N. et al. Therapeutic use of IL-2 to enhance antiviral T-cell responses in vivo. Nat. Med. 9, 540–547 (2003).
Williams, M.A., Tyznik, A.J. & Bevan, M.J. Interleukin-2 signals during priming are required for secondary expansion of CD8+ memory T cells. Nature 441, 890–893 (2006).
Reed, A.J. et al. The frequency of double-positive thymocytes expressing an αβ TCR clonotype regulates peripheral CD4 T cell compartment homeostasis. Immunology 116, 400–407 (2005).
Hamilton, S.E., Wolkers, M.C., Schoenberger, S.P. & Jameson, S.C. The generation of protective memory-like CD8+ T cells during homeostatic proliferation requires CD4+ T cells. Nat. Immunol. 7, 475–481 (2006).
Seddon, B., Tomlinson, P. & Zamoyska, R. Interleukin 7 and T cell receptor signals regulate homeostasis of CD4 memory cells. Nat. Immunol. 4, 680–686 (2003).
Lefrancois, L., Marzo, A.L., Masopust, D., Schluns, K.S. & Vezy, V. Migration of primary and memory CD8 T cells. Adv. Exp. Med. Biol. 512, 141–146 (2002).
Murali-Krishna, K. et al. Persistence of memory CD8 T cells in MHC class I–deficient mice. Science 286, 1377–1381 (1999).
Swain, S.L., Hu, H. & Huston, G. Class II–independent generation of CD4 memory T cells from effectors. Science 286, 1381–1383 (1999).
Wang, J. et al. The critical role of LIGHT in promoting intestinal inflammation and Crohn's disease. J. Immunol. 174, 8173–8182 (2005).
Hogquist, K.A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Acknowledgements
We thank P. Aliahmad and M. Fung for critically reviewing this manuscript, S. Freigang and J.T. Tan for discussion; N. Sanathara and O. Goularte for technical assistance; and the Department of Animal Resources and the Flow Cytometry Core Facility at The Scripps Research Institute for support. Supported by the National Institutes of Health (AI-31231 to J.K.), and the Swiss National Science Foundation and the Novartis Foundation (to O.B.). This is manuscript 18376 from the Scripps Research Institute.
Author information
Authors and Affiliations
Contributions
C.K. and O.B. did the experiments; C.K., O.B. and J.K. designed the experiments, interpreted the results and wrote the manuscript; and Y.-X.F. contributed reagents and helped with the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
BTLA and HVEM expression on CD8+ T cell subsets. (PDF 59 kb)
Supplementary Fig. 2
Increased CD4 to CD8 T cell ratio in BTLA-KO and HVEM-KO mice. (PDF 63 kb)
Supplementary Fig. 3
BTLA expression on homeostatically expanded WT CD8+ OT-I Tg T cells. (PDF 67 kb)
Rights and permissions
About this article
Cite this article
Krieg, C., Boyman, O., Fu, YX. et al. B and T lymphocyte attenuator regulates CD8+ T cell–intrinsic homeostasis and memory cell generation. Nat Immunol 8, 162–171 (2007). https://doi.org/10.1038/ni1418
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1418
This article is cited by
-
BTLA biology in cancer: from bench discoveries to clinical potentials
Biomarker Research (2024)
-
Cross-talk between disulfidptosis and immune check point genes defines the tumor microenvironment for the prediction of prognosis and immunotherapies in glioblastoma
Scientific Reports (2024)
-
Beyond the anti-PD-1/PD-L1 era: promising role of the BTLA/HVEM axis as a future target for cancer immunotherapy
Molecular Cancer (2023)
-
Memory T cells: strategies for optimizing tumor immunotherapy
Protein & Cell (2020)
-
Therapeutic Potential of B and T Lymphocyte Attenuator Expressed on CD8+ T Cells for Contact Hypersensitivity
Journal of Investigative Dermatology (2013)