Abstract
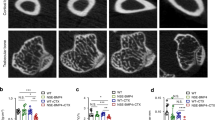
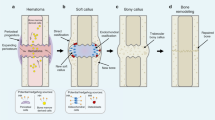
Heterotopic ossification consists of ectopic bone formation within soft tissues after surgery or trauma. It can have debilitating consequences, but there is no definitive cure. Here we show that heterotopic ossification was essentially prevented in mice receiving a nuclear retinoic acid receptor-γ (RAR-γ) agonist. Side effects were minimal, and there was no significant rebound effect. To uncover the mechanisms of these responses, we treated mouse mesenchymal stem cells with an RAR-γ agonist and transplanted them into nude mice. Whereas control cells formed ectopic bone masses, cells that had been pretreated with the RAR-γ agonist did not, suggesting that they had lost their skeletogenic potential. The cells became unresponsive to rBMP-2 treatment in vitro and showed decreases in phosphorylation of Smad1, Smad5 and Smad8 and in overall levels of Smad proteins. In addition, an RAR-γ agonist blocked heterotopic ossification in transgenic mice expressing activin receptor-like kinase-2 (ALK2) Q207D, a constitutively active form of the receptor that is related to ALK2 R206H found in individuals with fibrodysplasia ossificans progressiva. The data indicate that RAR-γ agonists are potent inhibitors of heterotopic ossification in mouse models and, thus, may also be effective against injury-induced and congenital heterotopic ossification in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
05 October 2012
In the version of this article initially published, the authors inadvertently used α-tubulin normalization data in the western blots shown in Figure 4b and Figure 4e from separate experiments used to generate the other bands shown. The error did not affect the main conclusions of the paper. Nonetheless, to verify the data, the authors have repeated the experiments, and the data from one such experiment are now included for both these figure panels. The authors regret the occurrence and apologize for it. The error has been corrected in the HTML and PDF versions of the article.
References
Chalmers, J., Gray, D.H. & Rush, J. Observation on the induction of bone in soft tissues. J. Bone Joint Surg. Br. 57, 36–45 (1975).
Garland, D.E. A clinical prospective on common forms of acquired heterotopic ossification. Clin. Orthop. Relat. Res. 263, 13–29 (1991).
Nilsson, O.S. & Persson, P.E. Heterotopic bone formation after joint replacement. Curr. Opin. Rheumatol. 11, 127–131 (1999).
Forsberg, J.A. et al. Heterotopic ossification in high-energy wartime extremity injuries: prevalence and risk factors. J. Bone Joint Surg. Am. 91, 1084–1091 (2009).
McCarthy, E.F. & Sundaram, M. Heterotopic ossification: a review. Skeletal Radiol. 34, 609–619 (2005).
Vanden Bossche, L. & Vanderstraeten, G. Heterotopic ossification: a review. J. Rehabil. Med. 37, 129–136 (2005).
Pignolo, R.J., Suda, R.K. & Kaplan, F.S. The fibrodysplasia ossificans progressiva lesion. Clin. Rev. Bone Min. Metab. 3, 195–200 (2005).
Shore, E.M. et al. A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat. Genet. 38, 525–527 (2006).
Chao, S.T., Joyce, M.J. & Suh, J.H. Treatment of heterotopic ossification. Orthopedics 30, 457–464 (2007).
Kaplan, F.S. The medical management of fibrodysplasia ossificans progressiva: current treatment considerations. Clin. Proc. Intl. Clin. Consort. FOP 3, 1082 (2008).
Fukuda, T. et al. Generation of a mouse with conditionally activated signaling through the BMP receptor, ALK2. Genesis 44, 159–167 (2006).
Zhang, D. et al. ALK2 functions as a BMP type I receptor and induces Indian hedgehog in chondrocytes during skeletal development. J. Bone Miner. Res. 18, 1593–1604 (2003).
Yu, P.B. et al. BMP type I receptor inhibition reduces heterotopic ossification. Nat. Med. 14, 1363–1369 (2008).
Shimono, K. et al. Inhibition of ectopic bone formation by a selective retinoic acid receptor α-agonist: a new therapy for heterotopic ossification? J. Orthop. Res. 28, 271–277 (2010).
Pacifici, M., Cossu, G., Molinaro, M. & Tato, F. Vitamin A inhibits chondrogenesis but not myogenesis. Exp. Cell Res. 129, 469–474 (1980).
Weston, A.D., Chandraratna, R.A.S., Torchia, J. & Underhill, T.M. Requirement for RAR-mediated gene repression in skeletal progenitor differentiation. J. Cell Biol. 158, 39–51 (2002).
Weston, A.D., Hoffman, L.M. & Underhill, T.M. Revisiting the role of retinoid signaling in skeletal development. Birth Defects Res. C. Embryo Today 69, 156–173 (2003).
Rochette-Egly, C. & Germain, P. Dynamic and combinatorial control of gene expression by nuclear retinoic acid receptors (RARs). Nucl. Recept. Signal. 7, e005 (2009).
Koyama, E. et al. Retinoid signaling is required for chondrocyte maturation and endochondral bone formation during limb skeletogenesis. Dev. Biol. 208, 375–391 (1999).
Williams, J.A. et al. Retinoic acid receptors are required for skeletal growth, matrix homeostasis and growth plate function in postnatal mouse. Dev. Biol. 328, 315–327 (2009).
Thacher, S.M., Vasudevan, J. & Chandraratna, R.A.S. Therapeutic applications for ligands of retinoic receptors. Curr. Pharm. Des. 6, 25–58 (2000).
O'Connor, J.P. Animal models of heterotopic ossification. Clin. Orthop. Relat. Res. 346, 71–80 (1998).
Géhin, M. et al. Structural basis for engineering of retinoic acid receptor isotype-selective agonists and antagonists. Chem. Biol. 6, 519–529 (1999).
Keidel, S., LeMotte, P. & Apfel, C. Different agonist- and antagonist-induced conformational changes in retinoic acid receptors analyzed by protease mapping. Mol. Cell Biol. 14, 287–298 (1994).
Klein, E.S. et al. Identification and functional separation of retinoic acid receptor neutral antagonists and inverse agonists. J. Biol. Chem. 271, 22692–22696 (1996).
Bernard, B.A. et al. Identification of synthetic retinoids for selectivity for human nuclear retinoic acid receptor γ. Biochem. Biophys. Res. Commun. 186, 977–983 (1992).
Hind, M. & Stinchcombe, S. Palovarotene, a novel retinoic acid receptor gamma agonist for the treatment of emphysema. Curr. Opin. Investig. Drugs 10, 1243–1250 (2009).
Blaner, W.S. & Olson, J.A. in The Retinoids: Biology, Chemistry, and Medicine (eds. Sporn, M.B., Roberts, A.B. & Goodman, D.S.) 229–255 (Raven, 1994).
Zasloff, M.A., Rocke, D.M., Crofford, L.J., Hahn, G.V. & Kaplan, F.S. Treatment of patients who have fibrodysplasia ossificans progressiva with isotretinoin. Clin. Orthop. Relat. Res. 346, 121–129 (1998).
DeSimone, D.P. & Reddi, A.H. Influence of vitamin A on matrix-induced endochondral bone formation. Calcif. Tissue Int. 35, 732–739 (1983).
Shen, Q. et al. The fibrodysplasia ossificans progressiva R206H ACVRa mutation activates BMP-independent chondrogenesis and zebrafish embryo ventralization. J. Clin. Invest. 119, 3462–3472 (2009).
Korchynskyi, O. & ten Dijke, P. Identification and functional characterization of distinct critically important bone morphogenetic protein-specific response elements in the Id1 promoter. J. Biol. Chem. 277, 4883–4891 (2002).
Massagué, J. TGF-β signal transduction. Annu. Rev. Biochem. 67, 753–791 (1998).
Derynck, R. & Zhang, Y.E. Smad-dependent and Smad-independent pathways in the TGF-β family signaling. Nature 425, 577–584 (2003).
Otsuru, S. et al. Osteopoietic engraftment after bone marrow transplantation: effect of imbred strain of mice. Exp. Hematol. 38, 836–844 (2010).
Hoffman, L.M. et al. BMP action in skeletogenesis involves attenuation of retinoid signaling. J. Cell Biol. 174, 101–113 (2006).
Pendaries, V., Verrecchia, F., Michel, S. & Mauviel, A. Retinoic acid receptors interfere with the TGF-β/Smad signaling pathway in a ligand-specific manner. Oncogene 22, 8212–8220 (2003).
Yasuhara, R. et al. Wnt/β-catenin and retinoic acid receptor signaling pathways interact to regulate chondrocyte function and matrix turnover. J. Biol. Chem. 285, 317–327 (2010).
Day, T.F., Guo, X., Garrett-Beal, L. & Yang, Y. Wnt/β-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev. Cell 8, 739–750 (2005).
Tamamura, Y. et al. Developmental regulation of Wnt/β-catenin signals is required for growth plate assembly, cartilage integrity, and endochondral ossification. J. Biol. Chem. 280, 19185–19195 (2005).
Nozaki, Y. et al. Anti-inflammatory effect of all-trans-retinoic acid in inflammatory arthritis. Clin. Immunol. 119, 272–279 (2006).
Benglis, D., Wang, M.Y. & Levi, A.D. A comprehensive review of the safety profile of bone morphogenetic protein in spine surgery. Neurosurgery 62, ONS423–ONS431 (2008).
Shao, J.-S., Cai, J. & Towler, D.A. Molecular mechanisms of vascular calcification: lessons learned from the aorta. Arterioscler. Thromb. Vasc. Biol. 26, 1423–1430 (2006).
Bone, L.B., Johnson, K.D., Weigelt, J. & Scheinberg, R. Early versus delayed stabilization of femural fractures. A prospective randomized study. J. Bone Joint Surg. Am. 71, 336–340 (1989).
Kakudo, N., Kausumoto, K., Wang, Y.B., Iguchi, Y. & Ogawa, Y. Immunolocalization of vascular endothelial growth factor on intramuscular ectopic osteoinduction by bone morphogenetic protein-2. Life Sci. 79, 1847–1855 (2006).
Acknowledgements
We thank T. Katagiri (Saitama) for the Id1-luc reporter plasmid; S. Otsuru and E. Horwitz (Children's Hospital of Philadelphia) for the GFP-expressing MSCs and culture methods; and N. Ghyselinck and P. Chambon (Institut National de la Santé et de la Recherche Médicale, Illkirch Cedex) for the RAR floxed and null mouse lines. This work was supported by contract no. W81XWH-07-1-0212 from the Department of the Army, United States Army Medical Research Acquisition Activity (M.P.) and US National Institutes of Health RO1 grant AR056837 (M.I. and M.P.).
Author information
Authors and Affiliations
Contributions
M.I. and M.P. directed the project. K.S., W.T., C.M., A.H.-T.C., J.H.D., C.M. and M.E.-I. performed experiments, analyzed data and participated in experimental design. R.A.C. provided expertise on retinoid biology. Y.M. provided expertise in mouse genetics. M.P., K.S. and M.I. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Methods (PDF 795 kb)
Rights and permissions
About this article
Cite this article
Shimono, K., Tung, We., Macolino, C. et al. Potent inhibition of heterotopic ossification by nuclear retinoic acid receptor-γ agonists. Nat Med 17, 454–460 (2011). https://doi.org/10.1038/nm.2334
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2334
This article is cited by
-
Palovarotene approved as first treatment for fibrodysplasia ossificans progressiva (FOP)
Journal of Rare Diseases (2024)
-
Single-cell RNA sequencing in orthopedic research
Bone Research (2023)
-
A Pharmacokinetic, Safety, and Tolerability Trial of Palovarotene in Healthy Japanese and Non-Japanese Participants
European Journal of Drug Metabolism and Pharmacokinetics (2023)
-
The Pharmacokinetic Profile of Palovarotene: An Open-Label Phase I Trial Investigating the Effect of Food and Potential for Drug–Drug Interaction in Healthy Participants
European Journal of Drug Metabolism and Pharmacokinetics (2023)
-
The role of miRNA and lncRNA in heterotopic ossification pathogenesis
Stem Cell Research & Therapy (2022)