Abstract
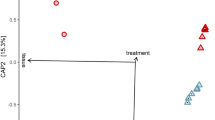
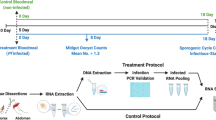
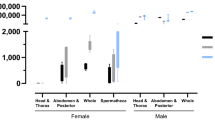
The use of transcriptional profiles for predicting mosquito age is a novel solution for the longstanding problem of determining the age of field-caught mosquitoes. Female mosquito age is of central importance to the transmission of a range of human pathogens. The transcriptional age-grading protocol we present here was developed in Aedes aegypti, principally as a research tool. Age predictions are made on the basis of transcriptional data collected from mosquitoes of known age. The abundance of eight candidate gene transcripts is quantified relative to a reference gene using quantitative reverse transcriptase-PCR (RT-PCR). Normalized gene expression (GE) measures are analyzed using canonical redundancy analysis to obtain a multivariate predictor of mosquito age. The relationship between the first redundancy variate and known age is used as the calibration model. Normalized GE measures are quantified for wild-caught mosquitoes, and ages are then predicted using this calibration model. Rearing of mosquitoes to specific ages for calibration data can take up to 40 d. Molecular analysis of transcript abundance, and subsequent age predictions, should take ∼3–5 d for 100 individuals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cook, P.E., McMeniman, C.J. & O'Neill, S.L. Modifying insect population age structure to control vector-borne disease. In Transgenesis and the Management of Vector-borne Disease (ed. Askoy, S.) (Landes Bioscience, Austin, TX, 2007).
Cook, P.E. et al. The use of transcriptional profiles to predict adult mosquito age under field conditions. Proc. Natl. Acad. Sci. USA 103, 18060–18065 (2006).
Gerade, B.B. et al. Field validation of Aedes aegypti (Diptera: Culicidae) age estimation by analysis of cuticular hydrocarbons. J. Med. Entomol. 41, 231–238 (2004).
Detinova, T.S. Age-grouping methods in Diptera of medical importance with special reference to some vectors of malaria. Monogr. Ser. World Health Organ. 47, 13–191 (1962).
Hayes, E.J. & Wall, R. Age-grading adult insects: a review of techniques. Physiol. Entomol. 24, 1 (1999).
Lehane, M.J. Determining the age of an insect. Parasitol. Today 1, 81–85 (1985).
Robson, S.K., Vickers, M., Blows, M.W. & Crozier, R.H. Age determination in individual wild-caught Drosophila serrata using pteridine concentration. J. Exp. Biol. 209, 3155–3163 (2006).
Wu, D. & Lehane, M.J. Pteridine fluorescence for age determination of Anopheles mosquitoes. Med. Vet. Entomol. 13, 48–52 (1999).
Penilla, R.P., Rodríguez, M.H., López, A.D., Viader-Salvadó, J.M. & Sánchez, C.N. Pteridine concentrations differ between insectary-reared and field-collected Anopheles albimanus mosquitoes of the same physiological age. Med. Vet. Entomol. 16, 225–234 (2002).
Holt, R.A. et al. The genome sequence of the malaria mosquito Anopheles gambiae. Science 298, 129–149 (2002).
Sanders, H.R., Evans, A.M., Ross, L.S. & Gill, S.S. Blood meal induces global changes in midgut gene expression in the disease vector, Aedes aegypti. Insect Biochem. Mol. Biol. 33, 1105–1122 (2003).
Nolan, T., Hands, R.E. & Bustin, S.A. Quantification of mRNA using real-time RT-PCR. Nat. Protoc. 1, 1559–1582 (2006).
Ståhlberg, A., Håkansson, J., Xian, X., Semb, H. & Kubista, M. Properties of the reverse transcription reaction in mRNA quantification. Clin. Chem. 50, 509–515 (2004).
Ståhlberg, A., Kubista, M. & Pfaffl, M. Comparison of reverse transcriptases in gene expression analysis. Clin. Chem. 50, 1678–1680 (2004).
Bustin, S.A. & Nolan, T. Pitfalls of quantitative real-time reverse-transcription polymerase chain reaction. J. Biomol. Tech. 15, 155–166 (2004).
Luu-The, V., Paquet, N., Calvo, E. & Cumps, J. Improved real-time RT-PCR method for high-throughput measurements using second derivative calculation and double correction. BioTechniques 38, 287–293 (2005).
Rasmussen, R. Quantification on the Lightcycler. In Rapid Cycle Real-time PCR: Methods and Applications (eds. Meuer, S.C., Wittwer, C. & Nakagawara, K.) pp. 21–34 (Springer, Heidelberg, Germany, 2001).
Williams, C.R., Long, S.A., Russell, R.C. & Ritchie, S.A. Field efficacy of the BG-sentinel compared with CDC backpack aspirators and CO2-baited EVS traps for collection of adult Aedes aegypti in Cairns, Queensland, Australia. J. Am. Mosq. Control Assoc. 22, 296–300 (2006).
Sambrook, J. & Russell, D.W. Molecular Cloning: A Laboratory Manual. 3rd ed. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2001).
Wilfinger, W.W., Mackey, K. & Chomczynski, P. Effect of pH and ionic strength on the spectrophotometric assessment of nucleic acid purity. BioTechniques 22, 474–476 (1997).
Aitchison, J. The statistical analysis of compositional data Reprint ed. (Blackburn Press, Caldwell, NJ, 2003).
Chomczynski, P. & Sacchi, N. The single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction: twenty-something years on. Nat. Protoc. 1, 581–585 (2006).
Acknowledgements
We are grateful to Dr. Jeremy C. Brownlie, Conor J. McMeniman and Grant Hughes for constructive reviews of earlier drafts of this manuscript. This research was funded by grants from the Australian Research Council (LP0455732), the Mosquito and Arbovirus Research Committee, and the Foundation for the National Institutes of Health through the Grand Challenges in Global Health Initiative.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Note
SAS syntax required to implement statistical analyses (PDF 31 kb)
Rights and permissions
About this article
Cite this article
Cook, P., Hugo, L., Iturbe-Ormaetxe, I. et al. Predicting the age of mosquitoes using transcriptional profiles. Nat Protoc 2, 2796–2806 (2007). https://doi.org/10.1038/nprot.2007.396
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.396
This article is cited by
-
Evaluation of diffuse reflectance spectroscopy for predicting age, species, and cuticular resistance of Anopheles gambiae s.l under laboratory conditions
Scientific Reports (2023)
-
Rapid age-grading and species identification of natural mosquitoes for malaria surveillance
Nature Communications (2022)
-
Use of transcriptional age grading technique to determine the chronological age of Sri Lankan Aedes aegypti and Aedes albopictus females
Parasites & Vectors (2021)
-
Ability of near-infrared spectroscopy and chemometrics to predict the age of mosquitoes reared under different conditions
Parasites & Vectors (2020)
-
Evaluation of reference genes at different developmental stages for quantitative real-time PCR in Aedes aegypti
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.