Abstract
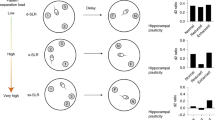
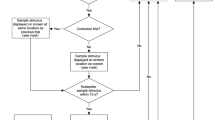
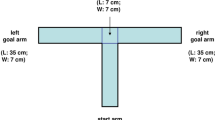
The automated touchscreen operant chamber for rats and mice allows for the assessment of multiple cognitive domains within the same testing environment. This protocol presents the location discrimination (LD) task and the trial-unique delayed nonmatching-to-location (TUNL) task, which both assess memory for location. During these tasks, animals are trained to a predefined criterion during ∼20–40 daily sessions. In LD sessions, touching the same location on the screen is rewarded on consecutive trials, followed by a reversal of location-reward contingencies. TUNL, a working memory task, requires animals to 'nonmatch' to a sample location after a delay. In both the LD and TUNL tasks, spatial similarity can be varied, allowing assessment of pattern separation ability, a function that is thought to be performed by the dentate gyrus (DG). These tasks are therefore particularly useful in animal models of hippocampal, and specifically DG, function, but they additionally permit discernment of changes in pattern separation from those in working memory.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
McTighe, S.M., Mar, A.C., Romberg, C., Bussey, T.J. & Saksida, L.M. A new touchscreen test of pattern separation: effect of hippocampal lesions. Neuroreport 20, 881–885 (2009).
Talpos, J.C., Dias, R., Bussey, T.J. & Saksida, L.M. Hippocampal lesions in rats impair learning and memory for locations on a touch-sensitive computer screen: the 'ASAT' task. Behav. Brain Res. 192, 216–225 (2008).
Talpos, J.C., McTighe, S.M., Dias, R., Saksida, L.M. & Bussey, T.J. Trial-unique, delayed nonmatching-to-location (TUNL): a novel, highly hippocampus-dependent automated touchscreen test of location memory and pattern separation. Neurobiol. Learn. Mem. 94, 341–352 (2010).
Talpos, J.C., Winters, B.D., Dias, R., Saksida, L.M. & Bussey, T.J. A novel touchscreen-automated paired-associate learning (PAL) task sensitive to pharmacological manipulation of the hippocampus: a translational rodent model of cognitive impairments in neurodegenerative disease. Psychopharmacology 205, 157–168 (2009).
Morris, R. Developments of a water-maze procedure for studying spatial learning in the rat. J. Neurosci. Methods 11, 47–60 (1984).
Barnes, C.A. Memory deficits associated with senescence: a neurophysiological and behavioral study in the rat. J. Comp. Physiol. Psychol. 93, 74–104 (1979).
Olton, D.S. The radial arm maze as a tool in behavioral pharmacology. Physiol. Behav. 40, 793–797 (1987).
Olton, D.S. Mazes, maps, and memory. Am. Psychol. 34, 583–596 (1979).
Dunnett, S.B. Comparative effects of cholinergic drugs and lesions of nucleus basalis or fimbria-fornix on delayed matching in rats. Psychopharmacology 87, 357–363 (1985).
Bierley, R.A. & Kesner, R.P. Short-term memory: the role of the midbrain reticular formation. J. Comp. Physiol. Psychol. 94, 519–529 (1980).
Aggleton, J.P., Keith, A.B., Rawlins, J.N., Hunt, P.R. & Sahgal, A. Removal of the hippocampus and transection of the fornix produce comparable deficits on delayed non-matching to position by rats. Behav. Brain Res. 52, 61–71 (1992).
Deadwyler, S.A., Bunn, T. & Hampson, R.E. Hippocampal ensemble activity during spatial delayed-nonmatch-to-sample performance in rats. J. Neurosci. 16, 354–372 (1996).
Hampson, R.E. & Deadwyler, S.A. Ensemble codes involving hippocampal neurons are at risk during delayed performance tests. Proc. Natl. Acad. Sci. USA 93, 13487–13493 (1996).
Bussey, T.J. et al. New translational assays for preclinical modelling of cognition in schizophrenia: the touchscreen testing method for mice and rats. Neuropharmacology 62, 1191–1203 (2012).
Bussey, T.J. et al. The touchscreen cognitive testing method for rodents: how to get the best out of your rat. Learn. Mem. 15, 516–523 (2008).
Horner, A.E. et al. The touchscreen operant platform for testing learning and memory in rats and mice. Nat. Protoc. 8, 1961–1984 (2013).
Nithianantharajah, J. et al. Synaptic scaffold evolution generated components of vertebrate cognitive complexity. Nat. Neurosci. 16, 16–24 (2013).
Mar, A. et al. The touchscreen operant platform for testing executive function in rats and mice. Nat. Protoc. 8, 1985–2005 (2013).
Wong, A.A. & Brown, R.E. Visual detection, pattern discrimination and visual acuity in 14 strains of mice. Genes Brain Behav. 5, 389–403 (2006).
Prusky, G.T., Harker, K.T., Douglas, R.M. & Whishaw, I.Q. Variation in visual acuity within pigmented, and between pigmented and albino rat strains. Behav. Brain Res. 136, 339–348 (2002).
Treves, A. & Rolls, E.T. Computational constraints suggest the need for two distinct input systems to the hippocampal CA3 network. Hippocampus 2, 189–199 (1992).
Treves, A., Tashiro, A., Witter, M.P. & Moser, E.I. What is the mammalian dentate gyrus good for? Neuroscience 154, 1155–1172 (2008).
Leutgeb, J.K., Leutgeb, S., Moser, M.B. & Moser, E.I. Pattern separation in the dentate gyrus and CA3 of the hippocampus. Science 315, 961–966 (2007).
Gilbert, P.E., Kesner, R.P. & Lee, I. Dissociating hippocampal subregions: double dissociation between dentate gyrus and CA1. Hippocampus 11, 626–636 (2001).
Yassa, M.A. & Stark, C.E. Pattern separation in the hippocampus. Trends Neurosci. 34, 515–525 (2011).
Gilbert, P.E., Kesner, R.P. & DeCoteau, W.E. Memory for spatial location: role of the hippocampus in mediating spatial pattern separation. J. Neurosci. 18, 804–810 (1998).
McHugh, T.J. et al. Dentate gyrus NMDA receptors mediate rapid pattern separation in the hippocampal network. Science 317, 94–99 (2007).
Clelland, C.D. et al. A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science 325, 210–213 (2009).
DeCarolis, N.A. & Eisch, A.J. Hippocampal neurogenesis as a target for the treatment of mental illness: a critical evaluation. Neuropharmacology 58, 884–893 (2010).
Eisch, A.J. et al. Adult neurogenesis, mental health, and mental illness: hope or hype? J. Neurosci. 28, 11785–11791 (2008).
Sahay, A. & Hen, R. Adult hippocampal neurogenesis in depression. Nat. Neurosci. 10, 1110–1115 (2007).
Lucassen, P.J. et al. Regulation of adult neurogenesis by stress, sleep disruption, exercise and inflammation: implications for depression and antidepressant action. Eur. Neuropsychopharmacol. 20, 1–17 (2010).
Reif, A. et al. Neural stem cell proliferation is decreased in schizophrenia, but not in depression. Mol. Psychiatry 11, 514–522 (2006).
Toro, C.T. & Deakin, J.F. Adult neurogenesis and schizophrenia: a window on abnormal early brain development? Schizophr. Res. 90, 1–14 (2007).
Jin, K. et al. Increased hippocampal neurogenesis in Alzheimer's disease. Proc. Natl. Acad. Sci. USA 101, 343–347 (2004).
Donovan, M.H. et al. Decreased adult hippocampal neurogenesis in the PDAPP mouse model of Alzheimer's disease. J. Comp. Neurol. 495, 70–83 (2006).
Wen, P.H. et al. The presenilin-1 familial Alzheimer disease mutant P117L impairs neurogenesis in the hippocampus of adult mice. Exp. Neurol. 188, 224–237 (2004).
Thompson, A., Boekhoorn, K., Van Dam, A.M. & Lucassen, P.J. Changes in adult neurogenesis in neurodegenerative diseases: cause or consequence? Genes Brain Behav. 7 (suppl. 1), 28–42 (2008).
Cameron, H.A. & Gould, E. Adult neurogenesis is regulated by adrenal steroids in the dentate gyrus. Neuroscience 61, 203–209 (1994).
Heine, V.M., Maslam, S., Zareno, J., Joels, M. & Lucassen, P.J. Suppressed proliferation and apoptotic changes in the rat dentate gyrus after acute and chronic stress are reversible. Eur. J. Neurosci. 19, 131–144 (2004).
Mirescu, C., Peters, J.D. & Gould, E. Early life experience alters response of adult neurogenesis to stress. Nat. Neurosci. 7, 841–846 (2004).
Oomen, C.A. et al. Severe early life stress hampers spatial learning and neurogenesis, but improves hippocampal synaptic plasticity and emotional learning under high-stress conditions in adulthood. J. Neurosci. 30, 6635–6645 (2010).
Snyder, J.S., Soumier, A., Brewer, M., Pickel, J. & Cameron, H.A. Adult hippocampal neurogenesis buffers stress responses and depressive behaviour. Nature 476, 458–461 (2011).
Seki, T. & Arai, Y. Age-related production of new granule cells in the adult dentate gyrus. Neuroreport 6, 2479–2482 (1995).
Kuhn, H.G., Dickinson-Anson, H. & Gage, F.H. Neurogenesis in the dentate gyrus of the adult rat: age-related decrease of neuronal progenitor proliferation. J. Neurosci. 16, 2027–2033 (1996).
van Praag, H., Christie, B.R., Sejnowski, T.J. & Gage, F.H. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc. Natl. Acad. Sci. USA 96, 13427–13431 (1999).
Galea, L.A. Gonadal hormone modulation of neurogenesis in the dentate gyrus of adult male and female rodents. Brain Res. Rev. 57, 332–341 (2008).
Tanapat, P., Hastings, N.B., Reeves, A.J. & Gould, E. Estrogen stimulates a transient increase in the number of new neurons in the dentate gyrus of the adult female rat. J. Neurosci. 19, 5792–5801 (1999).
Shors, T.J. et al. Neurogenesis in the adult is involved in the formation of trace memories. Nature 410, 372–376 (2001).
Leuner, B., Gould, E. & Shors, T.J. Is there a link between adult neurogenesis and learning? Hippocampus 16, 216–224 (2006).
Deng, W., Aimone, J.B. & Gage, F.H. New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? Nat. Rev. Neurosci. 11, 339–350 (2010).
Bekinschtein, P., Oomen, C.A., Saksida, L.M. & Bussey, T.J. Effects of environmental enrichment and voluntary exercise on neurogenesis, learning and memory, and pattern separation: BDNF as a critical variable? Semin. Cell Dev. Biol. 22, 536–542 (2011).
Dunnett, S.B., Wareham, A.T. & Torres, E.M. Cholinergic blockade in prefrontal cortex and hippocampus disrupts short-term memory in rats. Neuroreport 1, 61–64 (1990).
Granon, S., Vidal, C., Thinus-Blanc, C., Changeux, J.P. & Poucet, B. Working memory, response selection, and effortful processing in rats with medial prefrontal lesions. Behav. Neurosci. 108, 883–891 (1994).
Sloan, H.L., Good, M. & Dunnett, S.B. Double dissociation between hippocampal and prefrontal lesions on an operant delayed matching task and a water maze reference memory task. Behav. Brain Res. 171, 116–126 (2006).
Chudasama, Y. & Muir, J.L. A behavioural analysis of the delayed non-matching to position task: the effects of scopolamine, lesions of the fornix and of the prelimbic region on mediating behaviours by rats. Psychopharmacology 134, 73–82 (1997).
Dudchenko, P. & Sarter, M. Behavioral microanalysis of spatial delayed alternation performance: rehearsal through overt behavior, and effects of scopolamine and chlordiazepoxide. Psychopharmacology 107, 263–270 (1992).
Hearst, E. Delayed alternation in the pigeon. J. Exp. Anal. Behav. 5, 225–228 (1962).
Herremans, A.H., Hijzen, T.H., Welborn, P.F., Olivier, B. & Slangen, J.L. Effects of infusion of cholinergic drugs into the prefrontal cortex area on delayed matching to position performance in the rat. Brain Res. 711, 102–111 (1996).
McAllister, K.A.L., Saksida, L.M. & Bussey, T.J. Dissociation between memory retention across a delay and pattern separation following medial prefrontal cortex lesions in the touchscreen TUNL task. Neurobiol. Learn. Mem. 101, 120–126 (2013).
Postman, L. & Underwood, B. Critical issues in interference theory. Mem. Cogn. 1, 19–40 (1973).
Dale, R.H.I. & Roberts, W.A. Variations in radial maze performance under different levels of food and water-deprivation. Animal Learn. Behav. 14, 60–64 (1986).
Roberts, W.A. & Dale, R.H.I. Remembrance of places lasts-proactive-inhibition and patterns of choice in rat spatial memory. Learn. Motiv. 12, 261–281 (1981).
Grant, D.S. Intertrial interference in rat short-term memory. J. Exp. Psychol. Animal Behav. Processes 7, 217–227 (1981).
Nuechterlein, K.H. et al. The MATRICS consensus cognitive battery, part 1: test selection, reliability, and validity. Am. J. Psychiatry 165, 203–213 (2008).
Barch, D.M. et al. CNTRICS final task selection: working memory. Schizophr. Bull. 35, 136–152 (2009).
Harrison, P.J. & Weinberger, D.R. Schizophrenia genes, gene expression, and neuropathology: on the matter of their convergence. Mol. Psychiatry 10, 40–68 (2005).
Harrison, P.J. The hippocampus in schizophrenia: a review of the neuropathological evidence and its pathophysiological implications. Psychopharmacology 174, 151–162 (2004).
Meyer-Lindenberg, A.S. et al. Regionally specific disturbance of dorsolateral prefrontal-hippocampal functional connectivity in schizophrenia. Arch. Gen. Psychiatry 62, 379–386 (2005).
Carter, C.S. et al. Functional hypofrontality and working memory dysfunction in schizophrenia. Am. J. Psychiatry 155, 1285–1287 (1998).
Creer, D.J., Romberg, C., Saksida, L.M., van Praag, H. & Bussey, T.J. Running enhances spatial pattern separation in mice. Proc. Natl. Acad. Sci. USA 107, 2367–2372 (2010).
Coba, M.P. et al. TNiK is required for postsynaptic and nuclear signaling pathways and cognitive function. J. Neurosci. 32, 13987–13999 (2012).
Chaudhury, D. & Colwell, C.S. Circadian modulation of learning and memory in fear-conditioned mice. Behav. Brain Res. 133, 95–108 (2002).
Beeler, J.A., Prendergast, B. & Zhuang, X. Low amplitude entrainment of mice and the impact of circadian phase on behavior tests. Physiol. Behav 87, 870–880 (2006).
Roedel, A., Storch, C., Holsboer, F. & Ohl, F. Effects of light or dark phase testing on behavioural and cognitive performance in DBA mice. Lab. Anim. 40, 371–381 (2006).
Satoh, Y., Kawai, H., Kudo, N., Kawashima, Y. & Mitsumoto, A. Temperature rhythm reentrains faster than locomotor rhythm after a light phase shift. Physiol. Behav. 88, 404–410 (2006).
Bussey, T.J., Muir, J.L. & Robbins, T.W. A novel automated touchscreen procedure for assessing learning in the rat using computer graphic stimuli. Neurosci. Res. Commun. 15, 103–110 (1994).
Cardinal, R.N. & Aitken, M.R. Whisker: a client-server high-performance multimedia research control system. Behav. Res. Methods 42, 1059–1071 (2010).
Acknowledgements
The protocols described here are those used in our laboratory at present, and were written by current members of the group. However, many researchers have contributed to the development of touchscreen tasks and we would like to gratefully acknowledge their contribution. They include S. Bartko, J. Brigman, S. Forwood, C. Graybeal, A. Izquierdo, L. Lyon, A. Marti, K. McAllister, S. McTighe, J. Nithianantharajah, C. Romberg, J. Talpos and B. Winters. The research leading to these results has received support from the Innovative Medicine Initiative Joint Undertaking under grant agreement no. 115008, of which resources are composed of a European Federation of Pharmaceutical Industries and Associations in-kind contribution and financial contribution from the European Union's Seventh Framework Programme (FP7/2007-2013); and the Wellcome Trust/Medical Research Council (089703/Z/09/Z) and Alzheimer's Research UK (ART/PG2006/5). A.E.H. receives funding from the European Union Seventh Framework Programme under grant agreement nos. 241995 (Project 'GENCODYS') and 242167 (Project 'SYNSYS').
Author information
Authors and Affiliations
Contributions
All authors contributed to the writing of this manuscript. C.A.O. coordinated this effort.
Corresponding author
Ethics declarations
Competing interests
L.M.S. and T.J.B. consult for Campden Instruments, Ltd. A.E.H. is an employee of Synome, Ltd.
Rights and permissions
About this article
Cite this article
Oomen, C., Hvoslef-Eide, M., Heath, C. et al. The touchscreen operant platform for testing working memory and pattern separation in rats and mice. Nat Protoc 8, 2006–2021 (2013). https://doi.org/10.1038/nprot.2013.124
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.124
This article is cited by
-
Working memory and pattern separation in founder strains of the BXD recombinant inbred mouse panel
Scientific Reports (2022)
-
Using touchscreen-delivered cognitive assessments to address the principles of the 3Rs in behavioral sciences
Lab Animal (2021)
-
The spontaneous location recognition task for assessing spatial pattern separation and memory across a delay in rats and mice
Nature Protocols (2021)
-
MicroRNA-210 Regulates Dendritic Morphology and Behavioural Flexibility in Mice
Molecular Neurobiology (2021)
-
GLP-1R activation alters performance in cognitive tasks in a sex-dependent manner
Neurological Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.