Abstract
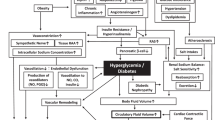
Hypertension and type 2 diabetes mellitus (T2DM) are powerful risk factors for cardiovascular disease (CVD) and chronic kidney disease (CKD), both of which are leading causes of morbidity and mortality worldwide. Research into the pathophysiology of CVD and CKD risk factors has identified salt sensitivity and insulin resistance as key elements underlying the relationship between hypertension and T2DM. Excess dietary salt and caloric intake, as commonly found in westernized diets, is linked not only to increased blood pressure, but also to defective insulin sensitivity and impaired glucose homeostasis. In this setting, activation of the sympathetic nervous system and the renin–angiotensin–aldosterone system (RAAS), as well as increased signaling through the mineralocorticoid receptor (MR), result in increased production of reactive oxygen species and oxidative stress, which in turn contribute to insulin resistance and impaired vascular function. In addition, insulin resistance is not limited to classic insulin-sensitive tissues such as skeletal muscle, but it also affects the cardiovascular system, where it participates in the development of CVD and CKD. Current clinical knowledge points towards an impact of salt restriction, RAAS blockade, and MR antagonism on cardiovascular and renal protection, but also on improved insulin sensitivity and glucose homeostasis.
Key Points
-
A pathophysiological link exists between hypertension and insulin resistance
-
Excess dietary salt intake has been linked to activation of the sympathetic nervous system (SNS), decreased insulin sensitivity, and activation of the renin–angiotensin–aldosterone system (RAAS)
-
SNS and RAAS activation and decreased insulin sensitivity participate in the pathogenesis of vascular dysfunction, which seems to be mediated by increased inflammation and oxidative stress
-
Insulin resistance affects classic insulin target tissues such as skeletal muscle, but also cardiovascular and renal tissue where it contributes to the development of cardiovascular and chronic kidney disease
-
Current studies suggest a beneficial role of mineralocorticoid receptor blockade, RAAS modulation, and decreased sodium intake on hypertension, insulin resistance, and cardiovascular and renal diseases
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Lastra, G., Manrique, C. M. & Sowers, J. R. Obesity, cardiometabolic syndrome, and chronic kidney disease: the weight of the evidence. Adv. Chronic Kidney Dis. 13, 365–373 (2006).
Lastra-Lastra, G., Sowers, J. R., Restrepo-Erazo, K., Manrique-Acevedo, C. & Lastra-González, G. Role of aldosterone and angiotensin II in insulin resistance: an update. Clin. Endocrinol. (Oxf.) 71, 1–6 (2009).
Strazzullo, P., Galletti, F. & Barba, G. Altered renal handling of sodium in human hypertension: short review of the evidence. Hypertension 41, 1000–1005 (2003).
De Wardener, H. E. & MacGregor, G. A. The natriuretic hormone and essential hypertension. Lancet 1, 1450–1454 (1982).
Houston, M. C. Sodium and hypertension. A review. Arch. Intern. Med. 146, 179–185 (1986).
Orlov, S. N. & Mongin, A. A. Salt-sensing mechanisms in blood pressure regulation and hypertension. Am. J. Physiol. Heart Circ. Physiol. 293, H2039–H2053 (2007).
Grim, C. E. et al. Racial differences in blood pressure in Evans County, Georgia: relationship to sodium and potassium intake and plasma renin activity. J. Chron. Dis. 33, 155–162 (1980).
Sacks, F. M. et al. Rationale and design of the Dietary Approaches to Stop Hypertension trial (DASH): A multicenter controlled-feeding study of dietary patterns to lower blood pressure. Ann. Epidemiol. 5, 108–118 (1995).
Sacks, F. M. et al. for the DASH-Sodium Collaborative Research Group. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. N. Engl. J. Med. 344, 3–10 (2001).
Weinberger, M. H., Miller, J. Z., Luft, F. C., Grim, C. E. & Fineberg, N. S. Definitions and characteristics of sodium sensitivity and blood pressure resistance. Hypertension 8, 127–134 (1996).
Weinberger, M. H. Salt sensitivity of blood pressure in humans. Hypertension 27, 481–490 (1996).
Franco, V. & Oparil, S. Salt sensitivity, a determinant of blood pressure, cardiovascular disease and survival. J. Am. Coll. Nutr. 25 (Suppl. 3), 247S–255S (2006).
Skrabal, F., Hamberger, L. & Ledochowski, M. Inherited salt sensitivity in normotensive humans as a cause of essential hypertension: a new concept. J. Cardiovasc. Pharmacol. 6 (Suppl. 1), S215–S223 (1984).
Weinberger, M. H., Fineberg, N. S., Fineberg, E. & Weinberger, M. Salt sensitivity, pulse pressure, and death in normal and hypertensive humans. Hypertension 27, 429–432 (2001).
Rocchini, A. P. et al. The effect of weight loss on the sensitivity of blood pressure to sodium in obese adolescents. N. Engl. J. Med. 321, 580–585 (1989).
Zavaroni, I. et al. Association between salt sensitivity and insulin concentrations in patients with hypertension. Am. J. Hypertens. 8, 855–858 (1995).
Sharma, A. M., Ruland, K., Spies, K. P. & Distler, A. Salt sensitivity in young normotensive subjects is associated with a hyperinsulinemic response to oral glucose. J. Hypertens. 9, 329–335 (1991).
Galletti, F. et al. NaCl sensitivity of essential hypertensive patients is related to insulin resistance. J. Hypertens. 15, 1485–1491 (1997).
Melander, O., Groop, L. & Hulthén, U. L. Effect of salt on insulin sensitivity differs according to gender and degree of salt sensitivity. Hypertension 35, 827–831 (2000).
Agarwal, M. K., Mirshahi, F., Mirshahi, M. & Rostene, W. Immunochemical detection of the mineralocorticoid receptor in rat brain. Neuroendocrinology 5, 575–580 (1993).
Geerling, J. C., Kawata, M. & Loewy, A. D. Aldosterone-sensitive neurons in the rat central nervous system. J. Comp. Neurol. 494, 515–527 (2006).
Connell, J. M. & Davies, E. The new biology of aldosterone. J. Endocrinol. 186, 1–20 (2005).
Gomez-Sanchez, E. P. Intracerebroventricular infusion of aldosterone induces hypertension in rats. Endocrinology 118, 819–823 (1986).
DiBona, G. F. & Sawin, L. L. Effect of arterial baroreceptor denervation on sodium balance. Hypertension 40, 547–551 (2002).
Sakai, R. R., McEwen, B. S., Fluharty, S. J. & Ma, L. Y. The amygdala: site of genomic and nongenomic arousal of aldosterone-induced sodium intake. Kidney Int. 57, 1337–1345 (2000).
Huang, B. S., Van Vliet, B. N. & Leenen, F. H. Increases in CSF [Na+] precede the increases in blood pressure in Dahl S rats and SHR on a high-salt diet. Am. J. Physiol. Heart Circ. Physiol. 287, H1160–H2116 (2004).
Huang, B. S., Amin, M. S. & Leenen, F. H. The central role of the brain in salt-sensitive hypertension. Curr. Opin. Cardiol. 21, 295–304 (2006).
Huang, B. S. et al. Activation of brain renin–angiotensin–aldosterone system by central sodium in Wistar rats. Am. J. Physiol. Heart Circ. Physiol. 291, H1109–H1117 (2006).
O'Donaughy, T. L., Qi, Y. & Brooks, V. L. Central action of increased osmolality to support blood pressure in deoxycorticosterone acetate-salt rats. Hypertension 48, 658–663 (2006).
Huang, B. S., Zheng, H., Patel, K. P. & Leenen, F. H. Central infusion of aldosterone decreases nNOS and increases AT1R and components of NADPH oxidase within the PVN [abstract P015]. Hypertension 52, e59 (2008).
Zhang, Z. H., Yu, Y., Kang, Y. M., Wei, S. G. & Felder, R. B. Aldosterone acts centrally to increase brain renin–angiotensin system activity and oxidative stress in normal rats. Am. J. Physiol. Heart Circ. Physiol. 294, H1067–H1074 (2008).
Yu, Y. et al. Does aldosterone upregulate the brain renin–angiotensin system in rats with heart failure? Hypertension 51, 727–733 (2008).
Wescott, K. V., Huang, B. S. & Leenen, F. H. Brain renin–angiotensin–aldosterone system and ventricular remodeling after myocardial infarct: a review. Can. J. Physiol. Pharmacol. 87, 979–988 (2009).
Felder, R. B. Mineralocorticoid receptors, inflammation and sympathetic drive in a rat model of systolic heart failure. Exp. Physiol. 95, 19–25 (2010).
Shi, P., Raizada, M. K. & Sumners, C. Brain cytokines as neuromodulators in cardiovascular control. Clin. Exp. Pharmacol. Physiol. 37, e52–e57 (2010).
Dunn, A. J. Cytokine activation of the HPA axis. Ann. NY Acad. Sci. 917, 608–617 (2000).
Francis, J., Weiss, R. M., Johnson, A. K. & Felder, R. B. Central mineralocorticoid receptor blockade decreases plasma TNF-alpha after coronary artery ligation in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 284, R328–R335 (2003).
Francis, J., Beltz, T., Johnson, A. K. & Felder, R. B. Mineralocorticoids act centrally to regulate blood-borne tumor necrosis factor-alpha in normal rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 285, R1402–R1409 (2003).
Lastra, G., Manrique, C., McFarlane, S. I. & Sowers, J. R. Cardiometabolic syndrome and chronic kidney disease. Curr. Diab. Rep. 6, 207–212 (2006).
Modan, M. et al. Hyperinsulinemia. A link between hypertension obesity and glucose intolerance. J. Clin. Invest. 75, 809–817 (1985).
Després, J. P. et al. Hyperinsulinemia as an independent risk factor for ischemic heart disease. N. Engl. J. Med. 334, 952–958 (1996).
Spät, A. & Hunyady, L. Control of aldosterone secretion: a model for convergence in cellular signaling pathways. Physiol. Rev. 84, 489–539 (2004).
Brilla, C. G. & Weber, K. T. Mineralocorticoid excess, dietary sodium, and myocardial fibrosis. J. Lab. Clin. Med. 120, 893–901 (1992).
Stas, S. et al. Mineralocorticoid receptor blockade attenuates chronic overexpression of the renin–angiotensin–aldosterone system stimulation of reduced nicotinamide adenine dinucleotide phosphate oxidase and cardiac remodeling. Endocrinology 148, 3773–3780 (2007).
Lastra, G. et al. Low-dose spironolactone reduces reactive oxygen species generation and improves insulin-stimulated glucose transport in skeletal muscle in the TG(mRen2)27 rat. Am. J. Physiol. Endocrinol. Metab. 295, E110–E116 (2008).
Endemann, D. H., Touyz, R. M., Iglarz, M., Savoia, C. & Schiffrin, E. L. Eplerenone prevents salt-induced vascular remodeling and cardiac fibrosis in stroke-prone spontaneously hypertensive rats. Hypertension 43, 1252–1257 (2004).
Bochud, M. et al. Plasma aldosterone is independently associated with the metabolic syndrome. Hypertension 48, 239–245 (2006).
Verhave, J. C. et al. for the PREVEND Study Group. Sodium intake affects urinary albumin excretion especially in overweight subjects. J. Intern. Med. 256, 324–330 (2004).
Fujita, T. Mineralocorticoid receptors, salt-sensitive hypertension, and metabolic syndrome. Hypertension 55, 813–818 (2010).
Montezano, A. C. & Touyz, R. M. Networking between systemic angiotensin II and cardiac mineralocorticoid receptors. Hypertension 52, 1016–1018 (2008).
Yamamuro, M. et al. Aldosterone, but not angiotensin II, reduces angiotensin converting enzyme 2 gene expression levels in cultured neonatal rat cardiomyocytes. Circ. J. 72, 1346–1350 (2008).
Zhang, A. D. et al. Cross-talk between mineralocorticoid and angiotensin II signaling for cardiac remodeling. Hypertension 52, 1060–1067 (2008).
Lastra, G., Manrique, C. M. & Sowers, J. R. Obesity, cardiometabolic syndrome, and chronic kidney disease: the weight of the evidence. Adv. Chronic Kidney Dis. 13, 365–373 (2006).
Rossi, G. P. et al. for the Primary Aldosteronism Prevalence in hypertension Study Investigators. Body mass index predicts plasma aldosterone concentrations in overweight-obese primary hypertensive patients. J. Clin. Endocrinol. Metab. 93, 2566–2571 (2008).
Jeon, J. H. et al. A novel adipokine CTRP1 stimulates aldosterone production. FASEB J. 22, 1502–1511 (2008).
Caprio, M. et al. Pivotal role of the mineralocorticoid receptor in corticosteroid-induced adipogenesis. FASEB J. 21, 2185–2194 (2007).
Wang, H. et al. Paradoxical mineralocorticoid receptor activation and left ventricular diastolic dysfunction under high oxidative stress conditions. J. Hypertension 26, 1453–1462 (2008).
Kawarazaki, H. et al. Mineralocorticoid receptor activation contributes to salt-induced hypertension and renal injury in prepubertal Dahl salt-sensitive rats. Nephrol. Dial. Transplant. doi: 10.1093/ndt/gfq197.
Nagase, M., Matsui, H., Shibata, S., Gotoda, T. & Fujita, T. Salt-induced nephropathy in obese spontaneously hypertensive rats via paradoxical activation of the mineralocorticoid receptor: role of oxidative stress. Hypertension 50, 877–883 (2007).
Griendling, K. K., Minieri, C. A., Ollerenshaw, J. D. & Alexander, R. W. Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells. Circ. Res. 74, 1141–1148 (1994).
Copper, S. A. et al. Renin–angiotensin–aldosterone system and oxidative stress in cardiovascular insulin resistance. Am. J. Physiol. Heart Circ. Physiol. 293, H2009–H2023 (2007).
Henquin, J. C. Triggering and amplifying pathways of regulation of insulin secretion by glucose. Diabetes 49, 1751–1760 (2000).
Shimamoto, K. et al. Does insulin resistance participate in an impaired glucose tolerance in primary aldosteronism? J. Hum. Hypertens. 8, 755–759 (1994).
Brown, N. J. Aldosterone and vascular inflammation. Hypertension 51, 161–167 (2008).
Manrique, C., Lastra, G., Whaley-Connel, A. & Sowers, J. R. Hypertension and the cardiometabolic syndrome. J. Clin. Hypertens. (Greenwich) 7, 471–476 (2005).
Yusuf, S. et al. for the Heart Outcomes Prevention Evaluation Study Investigators. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. N. Engl. J. Med. 342, 145–153 (2000).
Hansson, L. et al. Effect of angiotensin-converting-enzyme inhibition compared with conventional therapy on cardiovascular morbidity and mortality in hypertension: the Captopril Prevention Project (CAPPP) randomised trial. Lancet 353, 611–616 (1999).
Pfeffer, M. A. et al. for the CHARM Investigators and Committees. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM-Overall programme. Lancet 362, 759–766 (2003).
Barzilay, J. I. et al. for the ALLHAT Collaborative Research Group. Fasting glucose levels and incident diabetes mellitus in older nondiabetic adults randomized to receive 3 different classes of hypertensive treatment: a report from the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). Arch. Intern. Med. 166, 2191–2201 (2006).
Shindler, D. M. et al. Diabetes mellitus, a predictor of morbidity and mortality in the Studies of Left Ventricular Dysfunction (SOLVD) Trials and Registry. Am. J. Cardiol. 77, 1017–1020 (1996).
Devereux, R. B. et al. Regression of hypertensive left ventricular hypertrophy by losartan compared with atenolol: the Losartan Intervention for Endpoint Reduction in Hypertension (LIFE) trial. Circulation 110, 1456–1462 (2004).
Pitt, B. et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 341, 709–717 (1999).
Pitt, B. et al. for the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study Investigators. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N. Engl. J. Med. 348, 1309–1321 (2003).
Bosch, J. et al. for the DREAM Trial Investigators. Effect of ramipril on the incidence of diabetes. N. Engl. J. Med. 355, 1551–1562 (2006).
Sowers, J. R., Whaley-Connell, A. & Epstein, M. Narrative review: the emerging clinical implications of the role of aldosterone in the metabolic syndrome and resistant hypertension. Ann. Intern. Med. 150, 776–783 (2009).
McMurray, J. J. et al. for the NAVIGATOR Study Group. Effect of valsartan on the incidence of diabetes and cardiovascular events. N. Engl. J. Med. 362, 1477–1490 (2010).
Author information
Authors and Affiliations
Contributions
G. Lastra, S. Dhuper, M. S. Johnson, and J. R. Sowers contributed to discussion of content for the article, researched data to include in the manuscript, reviewed and edited the manuscript before submission, and revised the manuscript in response to the peer-reviewers' comments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lastra, G., Dhuper, S., Johnson, M. et al. Salt, aldosterone, and insulin resistance: impact on the cardiovascular system. Nat Rev Cardiol 7, 577–584 (2010). https://doi.org/10.1038/nrcardio.2010.123
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2010.123
This article is cited by
-
Perimenopausal syndrome and hypertension during perimenopause in South China: prevalence, relationships and risk factors
BMC Women's Health (2024)
-
Association between triglyceride glucose-body mass and one-year all-cause mortality of patients with heart failure: a retrospective study utilizing the MIMIC-IV database
Cardiovascular Diabetology (2023)
-
Triglyceride-glucose index, renal function and cardiovascular disease: a national cohort study
Cardiovascular Diabetology (2023)
-
Dietary salt intake predicts future development of metabolic syndrome in the general population
Hypertension Research (2023)
-
Inverse Salt Sensitivity of Blood Pressure: Mechanisms and Potential Relevance for Prevention of Cardiovascular Disease
Current Hypertension Reports (2022)