Abstract
The incidence of Clostridium difficile infection (CDI) has increased since 2000, with greater numbers of severe cases reported, in part due to the emergence of a hypervirulent strain. Initial therapy with metronidazole is still recommended for mild to moderate CDI, but vancomycin is recommended for first-line therapy of severe CDI. Colectomy could be life-saving for some patients with severe disease that does not respond to maximal medical therapy. Recurrent CDI is a challenge to treat; no single effective therapy currently exists. Treatments include antibiotics, adjunct probiotics, fecal microbiota transplant and immune approaches. This Review discusses the various therapeutic approaches used for the treatment of refractory and recurrent CDI.
Key Points
-
The incidence and prevalence of Clostridium difficile infection (CDI) have been increasing since 2000, with many severe cases occurring due to emergence of a hypervirulent strain
-
Metronidazole remains the mainstay of therapy for mild to moderate CDI but vancomycin should be used in patients with severe disease
-
Early surgical intervention can be life-saving in very ill patients with severe or fulminant disease who do not respond to maximal medical therapy, especially those ≥65 years
-
Patients with IBD have an increased risk of CDI, which can be more severe in these individuals
-
The treatment of recurrent CDI presents a clinical challenge; multiple therapeutic approaches have been tried, but none has been uniformly effective
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Archibald, L. K., Banerjee, S. N. & Jarvis, W. R. Secular trends in hospital-acquired Clostridium difficile disease in the United States, 1987–2001. J. Infect. Dis. 189, 1585–1589 (2004).
McDonald, L. C., Owings, M. & Jernigan, D. B. Clostridium difficile infection in patients discharged from US short-stay hospitals, 1996–2003. Emerg. Infect. Dis. 12, 409–415 (2006).
Warny, M., Vaerman, J. P., Avesani, V. & Delmée, M. Human antibody response to Clostridium difficile toxin A in relation to clinical course of infection. Infect. Immun. 62, 384–389 (1994).
Privitera, G. et al. Prospective study of Clostridium difficile intestinal colonization and disease following single-dose antibiotic prophylaxis in surgery. Antimicrob. Agents Chemother. 35, 208–210 (1991).
Ticehurst, J. R. et al. Effective detection of toxigenic Clostridium difficile by a two-step algorithm including tests for antigen and cytotoxin. J. Clin. Microbiol. 44, 1145–1149 (2006).
Larson, A. M., Fung, A. M. & Fang, F. C. Evaluation of tcbB real-time PCR in a three-step diagnostic algorithm for detection of toxigenic Clostridium difficile. J. Clin. Microbiol. 48, 124–130 (2010).
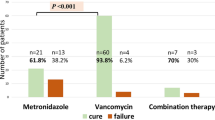
Zar, F. A., Bakkanagari, S. R., Moorthi, K. M. & Davis, M. B. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin. Infect. Dis. 45, 302–307 (2007).
Cohen, S. H. et al. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA). Infect. Control Hosp. Epidemiol. 31, 431–455 (2010).
Nair, S., Yadav, D., Corpuz, M. & Pitchumoni, C. S. Clostridium difficile colitis: factors influencing treatment failure and relapse—a prospective evaluation. Am. J. Gastroenterol. 93, 1873–1876 (1998).
Burdon, D. W. et al. Antibiotic susceptibility of Clostridium difficile. J. Antimicrob. Chemother. 5, 307–310 (1979).
Shuttleworth, R., Taylor, M. & Jones, D. M. Antimicrobial susceptibilities of Clostridium difficile. J. Clin. Pathol. 33, 1002–1005 (1980).
Nelson, R. Antibiotic treatment for Clostridium difficile-associated diarrhea in adults. Cochrane Database of Systematic Reviews, Issue 3, Art. No.: CD004610. doi:10.1002/14651858.CD004610.pub3 (2007).
Baines, S. D. et al. Emergence of reduced susceptibility to metronidazole in Clostridium difficile. J. Antimicrob. Chemother. 62, 1046–1052 (2008).
Peláez, T. et al. Metronidazole resistance in Clostridium difficile is heterogeneous. J. Clin. Microbiol. 46, 3028–3032 (2008).
Musher, D. M. et al. Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole. Clin. Infect. Dis. 40, 1586–1590 (2005).
Pepin, J. et al. Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin. Infect. Dis. 40, 1591–1597 (2005).
Aslam, S., Hamill, R. J. & Musher, D. M. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Lancet Infect. Dis. 5, 549–557 (2005).
Wong, S. S., Woo, P. C., Luk, W. K. & Yuen, K. Y. Susceptibility testing of Clostridium difficile against metronidazole and vancomycin by disk diffusion and Etest. Diagn. Microbiol. Infect. Dis. 34, 1–6 (1999).
Marchese, A., Salerno, A., Pesce, A., Debbia, E. A. & Schito, G. C. In vitro activity of rifaximin, metronidazole and vancomycin against Clostridium difficile and the rate of selection of spontaneously resistant mutants against representative anaerobic and aerobic bacteria, including ammonia-producing species. Chemotherapy 46, 253–266 (2000).
Jamal, W. Y., Mokaddas, E. M., Verghese, T. L. & Rotimi, V. O. In vitro activity of 15 antimicrobial agents against clinical isolates of Clostridium difficile in Kuwait. Int. J. Antimicrob. Agents 20, 270–274 (2002).
Peláez, T. et al. Reassessment of Clostridium difficile susceptibility to metronidazole and vancomycin. Antimicrob. Agents Chemother. 46, 1647–1650 (2002).
Tedesco, F., Markham, R., Gurwith, M., Christie, D. & Bartlett, J. G. Oral vancomycin for antibiotic-associated pseudomembranous colitis. Lancet 2, 226–228 (1978).
Al-Nassir, W. N. et al. Comparison of clinical and microbiological response to treatment of Clostridium difficile-associated disease with metronidazole and vancomycin. Clin. Infect. Dis. 47, 56–62 (2008).
Louie, T. J. et al. Tolevamer, a novel nonantibiotic polymer, compared with vancomycin in the treatment of mild to moderately severe Clostridium difficile-associated diarrhea. Clin. Infect. Dis. 43, 411–420 (2006).
Louie, T. J. et al. Results of a phase III study comparing tolevamer, vancomycin and metronidazole in Clostridium difficile-associated diarrhea (CDAD) [abstract K-4259]. In Program and Abstracts of the 47th Interscience Conference on Antimicrobial Agents and Chemotherapy (ASM Press, Washington, D. C., 2007).
Apisarnthanarak, A., Razavi, B. & Mundy, L. M. Adjunctive intracolonic vancomycin for severe Clostridium difficile colitis: case series and review of the literature. Clin. Infect. Dis. 35, 690–696 (2002).
Young, G. P. et al. Antibiotic-associated colitis due to Clostridium difficile: double-blind comparison of vancomycin with bacitracin. Gastroenterology 89, 1038–1045 (1985).
Dudley, M. N. et al. Oral bacitracin vs vancomycin therapy for Clostridium difficile-induced diarrhea. A randomized double-blind trial. Arch. Intern. Med. 146, 1101–1104 (1986).
de Lalla, F. et al. Prospective study of oral teicoplanin versus oral vancomycin for therapy of pseudomembranous colitis and Clostridium difficile-associated diarrhea. Antimicrob. Agents Chemother. 36, 2192–2196 (1992).
de Lalla, F. et al. Treatment of Clostridium difficile-associated disease with teicoplanin. Antimicrob. Agents Chemother. 33, 1125–1127 (1989).
Wullt, M. & Odenholt, I. A double-blind randomized controlled trial of fusidic acid and metronidazole for treatment of an initial episode of Clostridium difficile-associated diarrhoea. J. Antimicrob. Chemother. 54, 211–216 (2004).
Wenisch, C., Parschalk, B., Hasenhündl, M., Hirschl, A. M. & Graninger, W. Comparison of vancomycin, teicoplanin, metronidazole, and fusidic acid for the treatment of Clostridium difficile-associated diarrhea. Clin. Infect. Dis. 22, 813–818 (1996).
Musher, D. M., Logan, N., Bressler, A. M., Johnson, D. P. & Rossignol, J. F. Nitazoxanide versus vancomycin in Clostridium difficile infection: a randomized, double-blind study. Clin. Infect. Dis. 48, e41–e46 (2009).
Musher, D. M. et al. Clostridium difficile colitis that fails conventional metronidazole therapy: response to nitazoxanide. J. Antimicrob. Chemother. 59, 705–710 (2007).
Garey, K. W., Salazar, M., Shah, D., Rodrigue, R. & DuPont, H. I. Rifamycin antibiotics for treatment of Clostridium difficile-associated diarrhea. Ann. Pharmacother. 42, 827–835 (2008).
Basu, P. P. et al. Rifaximin therapy for metronidazole-unresponsive Clostridium difficile infection: a prospective pilot trial. Therap. Adv. Gastroenterol. 414, 221–225 (2010).
Herpers, B. L. et al. Intravenous tigecycline as adjunctive or alternative therapy for severe refractory Clostridium difficile infection. Clin. Infect. Dis. 48, 1732–1735 (2009).
Lu, C. L., Liu, C. Y., Huang, Y. T., Wang, H. P. & Hsueh, P. R. Severe and refractory Clostridium difficile infection successfully treated with tigecycline and metronidazole. Int. J. Antimicrob. Agents 35, 311–312 (2010).
Kopterides, P. et al. Failure of tigecycline to treat severe Clostridium difficile. Anaesth. Intensive Care 38, 755–758 (2010).
Salcedo, J. et al. Intravenous immunoglobulin therapy for severe Clostridium difficile colitis. Gut 41, 366–370 (1997).
McPherson, S., Rees, C. J., Ellis, R., Soo, S. & Panter, S. J. Intravenous immunoglobulin for the treatment of severe, refractory, and recurrent Clostridium difficile diarrhea. Dis. Colon Rectum 49, 640–645 (2006).
Juang, P. et al. Clinical outcomes of intravenous immune globulin in severe Clostridium difficile-associated diarrhea. Am. J. Infect. Control 35, 131–137 (2007).
Abougergi, M. S., Broor, A., Cui, W. & Jaar, B. G. Intravenous immunoglobulin for the treatment of severe Clostridium difficile colitis: an observational study and review of the literature. J. Hosp. Med. 5, E1–E9 (2010).
O'Horo, J. & Safdar, N. The role of immunoglobulin for the treatment of Clostridium difficile infection: a systematic review. Int. J. Infect. Dis. 13, 663–667 (2009).
Abougergi, M. S. & Kwon, J. H. Intravenous immunoglobulin for the treatment of Clostridium difficile infection: a review. Dig. Dis. Sci. 56, 19–26 (2011).
Khoruts, A., Dicksved, J., Jansson, J. K. & Sadowsky, M. J. Changes in the composition of the human fecal microbiome after bacteriotherapy for recurrent Clostridium difficile-associated diarrhea. J. Clin. Gastroenterol. 44, 354–360 (2010).
Eiseman, B., Silen, W., Bascom, G. S. & Kauvar, A. J. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery 44, 854–859 (1958).
Bowden, T. A. Jr, Mansberger, A. R. Jr & Lykins, L. E. Pseudomembranous enterocolitis: mechanism for restoring floral homeostasis. Am. Surg. 47, 178–183 (1981).
You, D. M., Franzos, M. A. & Holman, R. P. Successful treatment of fulminant Clostridium difficile infection with fecal bacteriotherapy. Ann. Intern. Med. 148, 632–633 (2008).
Koo, H. L., Koo, D. C., Musher, D. M. & DuPont, H. L. Antimotility agents for treatment of Clostridium difficile diarrhea and colitis. Clin. Infect. Dis. 48, 598–605 (2009).
Dallal, R. M. et al. Fulminant Clostridium difficile: an underappreciated and increasing cause of death and complication. Ann. Surg. 235, 363–372 (2002).
Koss, K. et al. The outcome of surgery in fulminant Clostridium difficile colitis. Colorectal Dis. 8, 149–154 (2006).
Lamontagne, F. et al. Impact of emergency colectomy on survival of patients with fulminant Clostridium difficile colitis during an epidemic caused by a hypervirulent strain. Ann. Surg. 245, 267–272 (2007).
Dudukgian, H., Sie, E., Gonzalez-Ruiz, C., Etzioni, D. A. & Kaiser, A. M. C. difficile colitis–predictors of fatal outcome. J. Gastrointest. Surg. 14, 315–322 (2010).
Seder, C. W. et al. Early colectomy may be associated with improved survival in fulminant Clostridium difficile colitis: an 8-year experience. Am. J. Surg. 197, 302–307 (2009).
Al-Abed, Y. A., Gray, E. A. & Rothnie, N. D. Outcomes of emergency colectomy for fulminant Clostridium difficile colitis. Surgeon 8, 330–333 (2010).
Butala, P. & Divino, C. M. Surgical aspects of fulminant Clostridium difficile colitis. Am. J. Surg. 200, 131–135 (2010).
Ananthakrishnan, A. N. & Binion, D. G. Impact of Clostridium difficile on inflammatory bowel disease. Expert Rev. Gastroenterol. Hepatol. 4, 589–600 (2010).
Issa, M. et al. Impact of Clostridium difficile on inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 5, 345–351 (2007).
Rodemann, J. F., Dubberke, E. R., Reske, K. A., Seo da, H. & Stone, C. D. Incidence of Clostridium difficile infection in inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 5, 339–344 (2007).
Ananthakrishnan, A. N. & Binion, D. G. Clostridium difficile and inflammatory bowel disease: more questions than answers? Inflamm. Bowel Dis. 16, 1445–1446 (2010).
Das, R., Feuerstadt, P. F. & Brandt, L. J. Glucocorticoids are associated with increased risk of short-term mortality in hospitalized patients with Clostridium difficile-associated disease. Am. J. Gastroenterol. 105, 2040–2049 (2010).
McFarland, L. V. et al. Recurrent Clostridium difficile disease: epidemiology and clinical characteristics. Infect. Control Hosp. Epidemiol. 20, 43–50 (1999).
McFarland, L. V. Update on the changing epidemiology of Clostridium difficile-associated disease. Nat. Clin. Pract. Gastroenterol. Hepatol. 5, 40–48 (2008).
McFarland, L. V. et al. Possible role of cross transmission between neonates and mothers with recurrent Clostridium difficile infections. Am. J. Infect. Control 27, 301–303 (1999).
Johnson, S. Recurrent Clostridium difficile infection: a review of risk factors, treatments, and outcomes. J. Infect. 58, 403–410 (2009).
Hu, M. Y. et al. Prospective derivation and validation of a clinical prediction rule for recurrent Clostridium difficile infection. Gastroenterology 136, 1206–1214 (2009).
Cadena, J. et al. Clinical predictors and risk factors for relapsing Clostridium difficile infection. Am. J. Med. Sci. 339, 350–355 (2010).
Kyne, L., Warny, M., Qumar, A. & Kelly, C. P. Association between antibody response in toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet 357, 189–193 (2001).
Surawicz, C. M. Treatment of recurrent Clostridium difficile infection-associated disease. Nat. Clin. Pract. Gastroenterol. Hepatol. 1, 32–38 (2004).
McFarland, L. V., Elmer, G. W. & Surawicz, C. M. Breaking the cycle: treatment strategies for 163 cases of recurrent C. difficile disease. Am. J. Gastroenterol. 97, 1769–1775 (2002).
Johnson, S., Schriever, C., Galang, M., Kelly, C. P. & Gerding, D. N. Interruption of recurrent Clostridium difficile-associated diarrhea episodes by serial therapy with vancomycin and rifaximin. Clin. Infect. Dis. 44, 846–848 (2007).
Johnson, S. et al. Rifaximin redux: treatment of recurrent Clostridium difficile infections with rifaximin immediately post-vancomycin treatment. Anaerobe 15, 290–291 (2009).
Neff, G., Zacharias, V., Kaiser, T. E., Gaddis, A. & Kemmer, N. Rifaximin for the treatment of recurrent Clostridium difficile infection after liver transplantation: a case series. Liver Transpl. 16, 960–963 (2010).
Garey, K. W., Jiang, Z. D., Bellard, A. & DuPont, H. L. Rifaximin in treatment of recurrent Clostridium difficile-associated diarrhea: an uncontrolled pilot study. J. Clin. Gastroenterol. 43, 91–93 (2009).
Louie, T. J., Emery, J., Krulicki, W., Byrne, B. & Mah, M. OPT-80 eliminates Clostridium difficile and is sparing of Bacteroides species during treatment of C. difficile infection. Antimicrob. Agents Chemother. 53, 261–263 (2009).
Sullivan, K. M. & Spooner, L. M. Fidaxomicin: a macrocyclic antibiotic for the management of Clostridium difficile infection. Ann. Pharmacother. 44, 352–359 (2010).
Tannock, G. W. et al. A new macrocyclic antibiotic, fidaxomicin (OPT-80), causes less alteration to the bowel microbiota of Clostridium difficile-infected patients than does vancomycin. Microbiology 156, 3354–3359 (2010).
McFarland, L. V. et al. A randomized placebo-controlled trial of Saccharomyces boulardii in combination with standard antibiotics for Clostridium difficile disease. JAMA 271, 1913–1918 (1994).
Surawicz, C. M. et al. The search for a better treatment for recurrent Clostridium difficile disease: use of high-dose vancomycin combined with Saccharomyces boulardii. Clin. Infect. Dis. 31, 1012–1017 (2000).
Wullt, M., Gagslatt, M. L. & Odenhold, I. Lactobacillus plantarum 299v for the treatment of recurrent Clostridium difficile-associated diarrhea: a double-blind placebo-controlled trial. Scand. J. Infect. Dis. 35, 365–367 (2003).
Pochapin, M. The effect of probiotics on Clostridium difficile diarrhea. Am. J. Gastroenterol. 95 (Suppl. 1), S11–S13 (2000).
Lawrence, S. J., Korzenik, J. R. & Mundy, L. M. Probiotics for recurrent Clostridium difficile disease. J. Med. Microbiol. 54, 905–906 (2005).
Bakken, J. S. Resolution of Clostridium difficile-associated diarrhea using staggered antibiotic withdrawal and kefir. Minn. Med. 92, 38–40 (2009).
Liong, M. T. Safety of probiotics: translocation and infection. Nutr. Rev. 66, 192–202 (2008).
Seal, D. et al. Treatment of relapsing Clostridium difficile diarrhea by administration of a non-toxigenic strain. Eur. J. Clin. Microbiol. 6, 51–53 (1987).
Schwan, A., Sjolin, S., Trottestam, U. & Aronsson, B. Relapsing Clostridium difficile enterocolitis cured by rectal infusion of homologous faeces. Lancet 2, 845 (1983).
Tvede, M. & Rask-Madsen, J. Bacteriotherapy for chronic relapsing Clostridium difficile diarrhea in six patients. Lancet 1, 1156–1160 (1989).
Silverman, M. S., Davis, I. & Pillai, D. R. Success of self-administered home fecal transplantation for chronic Clostridium difficile infection. Clin. Gastroenterol. Hepatol. 8, 471–473 (2010).
Rohlke, F., Surawicz, C. M. & Stollman, N. Fecal flora reconstitution for recurrent Clostridium difficile infection: results and methodology. J. Clin. Gastroenterol. 44, 567–570 (2010).
Yoon, S. S. & Brandt, L. J. Treatment of refractory/recurrent C. difficile-associated disease by donated stool transplanted via colonoscopy: a case series of 12 patients. J. Clin. Gastroenterol. 44, 562–566 (2010).
Aas, J., Gessert, C. E. & Bakken, J. S. Recurrent Clostridium difficile colitis: case series involving 18 patients treated with donor stool administered via a nasogastric tube. Clin. Infect. Dis. 36, 580–586 (2003).
MacConnachie, A. A., Fox, R., Kennedy, D. R. & Seaton, R. A. Faecal transplant for recurrent Clostridium difficile-associated diarrhoea: a UK case series. QJM 102, 781–784 (2009).
Bakken, J. S. Fecal bacteriotherapy for recurrent Clostridium difficile infection. Anaerobe 15, 285–289 (2009).
Khoruts, A. & Sadowsky, M. J. Therapeutic transplantation of the distal gut microbiota. Mucosal Immunol. 4, 4–7 (2011).
van Nood, E., Speelman, P., Kuijper, E. J. & Keller, J. J. Struggling with recurrent Clostridium difficile infections: is donor faeces the solution? Eur. Surveill. 14, 19316 (2009).
Van Dissel, J. T. et al. Bovine antibody-enriched whey to aid in the prevention of a relapse of Clostridium difficile-associated diarrhoea preclinical and preliminary clinical data. J. Med. Microbiol. 54, 197–205 (2005).
Mattila, E. et al. A randomized, double-blind study comparing Clostridium difficile immune whey and metronidazole for recurrent Clostridium difficile-associated diarrhoea: efficacy and safety data of a prematurely interrupted trial. Scand. J. Infect. Dis. 40, 702–708 (2008).
Sougioultzis, S. et al. Clostridium difficile toxoid vaccine in recurrent C. difficile-associated diarrhea. Gastroenterology 128, 764–770 (2005).
Wilcox, W. H. Descriptive study of intravenous immunoglobulin for the treatment of recurrent Clostridium difficile diarrhoea. J. Antimicrob. Chemother. 53, 882–884 (2004).
Beales, I. L. Intravenous immunoglobulin for recurrent Clostridium difficile diarrhoea. Gut 51, 456 (2002).
Lowy, I. et al. Treatment with monoclonal antibodies against Clostridium difficile toxins. N. Engl. J. Med. 362, 197–205 (2010).
Gerding, D. N. & Johnson, S. Management of Clostridium difficile infection: thinking inside and outside the box. Clin. Infect. Dis. 51, 1306–1313 (2010).
Pépin, J. Vancomycin for the treatment of Clostridium difficile infection: for whom is this expensive bullet really magic? Clin. Infect. Dis. 46, 1493–1498 (2008).
Pépin, J. et al. Clostridium difficile-associated diarrhea in a region of Quebec from 1991 to 2003: a changing pattern of disease severity. CMAJ 171, 466–472 (2004).
Acknowledgements
Thanks to Drs Thomas Bendy, Lawrence Brandt, Colleen Kelly and Alexander Khoruts who participated in discussions of proper terminology of the fecal microbiota transplant procedure.
Author information
Authors and Affiliations
Contributions
C. M. Surawicz and J. Alexander contributed equally to researching data for the article and discussing the content. The contribution of C. M. Surawicz in writing and reviewing/editing the final manuscript was substantially more.
Corresponding author
Ethics declarations
Competing interests
C. M. Surawicz has been on the Speaker's bureau for Biocodex. J. Alexander declares no competing interests.
Rights and permissions
About this article
Cite this article
Surawicz, C., Alexander, J. Treatment of refractory and recurrent Clostridium difficile infection. Nat Rev Gastroenterol Hepatol 8, 330–339 (2011). https://doi.org/10.1038/nrgastro.2011.59
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2011.59
This article is cited by
-
Biofilms harbour Clostridioides difficile, serving as a reservoir for recurrent infection
npj Biofilms and Microbiomes (2021)
-
A Systematic Literature Review of Economic Evaluations of Antibiotic Treatments for Clostridium difficile Infection
PharmacoEconomics (2017)
-
The incidence and drug resistance of Clostridium difficile infection in Mainland China: a systematic review and meta-analysis
Scientific Reports (2016)
-
A cost-effectiveness and budget impact analysis of first-line fidaxomicin for patients with Clostridium difficile infection (CDI) in Germany
Infection (2016)
-
Epidemiology and Recurrence Rates of Clostridium difficile Infections in Germany: A Secondary Data Analysis
Infectious Diseases and Therapy (2016)