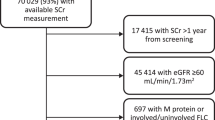
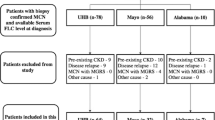
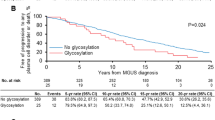
Abstract
Abnormalities of immunoglobulin free light chains (FLCs) are frequently present in patients with monoclonal gammopathies and can cause kidney disease. The recent introduction of highly sensitive immunoassays that measure FLCs to levels below those present in normal individuals has provided a new tool for diagnosis and management in this setting. Here, we review the biology of FLC production in health and disease, and the utility of FLC immunoassays in the assessment of monoclonal gammopathies in kidney disease.
Key Points
-
Serum free light chain (FLC) immunoassays can identify monoclonal FLCs with high sensitivity and specificity
-
The screening of serum alone using serum protein electrophoresis and measuring serum κ and λ FLC levels will identify all patients with multiple myeloma and 99% of patients with AL amyloidosis
-
Concentrations of serum polyclonal FLCs are GFR dependent and increase with increasing renal impairment
-
The monitoring of serum FLCs provides an insight into the response to treatment of patients with acute kidney injury and myeloma
-
Renal recovery from cast nephropathy is associated with a significant reduction in serum FLC levels
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bradwell, A. R. et al. Highly sensitive automated immunoassay for immunoglobulin FLCs in serum and urine. Clin. Chem. 47, 673–680 (2001).
Katzmann, J. A. et al. Serum reference intervals and diagnostic ranges for free κ and free λ immunoglobulin light chains: relative sensitivity for detection of monoclonal light chains. Clin. Chem. 48, 1437–1444 (2002).
Doyle, A., Soutar, R. & Geddes, C. C. Multiple myeloma in chronic kidney disease. Nephron Clin. Pract. 111, c7–c11 (2009).
Jones, H. B. Papers on chemical pathology, Lecture III. Lancet II, 88–92 (1847).
Solomon, A., Waldmann, T. A., Fahey, J. L. & McFarlane, A. S. Metabolism of Bence Jones proteins. J. Clin. Invest. 43, 103–117 (1964).
Lindstedt, G. & Lungberg, P. Loss of tubular proteinuria pattern during urine concentration with a commerical membrane filter cell. Clin. Chem. Acta 56, 125–126 (1974).
Harrison, H. H. The “Ladder Light Chain” or “Pseudo-Oligoclonal” pattern in urinary immunofixation electrophoresis (IFE) studies: a distinct IFE pattern and an explanation hypothesis relating it to free polyclonal light chains. Clin. Chem. 37, 1559–1564 (1991).
Hess, P. P., Mastropaolo, W., Thompson, G. D. & Levinson, S. S. Interference of polyclonal free light chains with identification of Bence-Jones proteins. Clin. Chem. 39, 1734–1738 (1993).
Bridgen, M. L., Neal, E. D., McNeely, N. D. & Hoag, G. N. The optimal urine collection for detection and monitoring of Bence Jones proteinuria. Am. J. Clin. Path. 93, 689–693 (1990).
Hill, P. G., Forsyth, J. M., Rai, B. & Mayne, S. Serum free light chains: an alternative to the urine Bence Jones proteins screening test for monoclonal gammopathies. Clin. Chem. 52, 1743–1748 (2006).
Hutchison, C. A. et al. Serum free light chain measurement aids the diagnosis of myeloma in patients with severe renal failure. BMC Nephrol. 22, 11 (2008).
Pratt, G. The evolving use of serum free light chain assays in haematology. Br. J. Haematol. 141, 413–422 (2008).
Dispenzieri, A. et al. International Myeloma Working Group guidelines for serum-free light chain analysis in multiple myeloma and related disorders. Leukemia 23, 215–224 (2009).
Hutchison, C. A. et al. Quantitative assessment of serum and urinary polyclonal free light chains in patients with chronic kidney disease. Clin. J. Am. Soc. Nephrol. 3, 1684–1690 (2008).
Hutchison, C. A. et al. Efficient removal of immunoglobulin free light chains by hemodialysis for multiple myeloma: in vitro and in vivo studies. J. Am. Soc. Nephrol. 18, 886–895 (2007).
Cserti, C., Haspel, R., Stowell, C. & Dzik, W. Light-chain removal by plasmapheresis in myeloma-associated renal failure. Transfusion 47, 511–514 (2007).
Pillon, L. et al. Approach to acute renal failure in biopsy proven myeloma cast nephropathy: is there still a role for plasmapharesis? Kidney Int. 74, 956–961 (2008).
Leung, N. et al. Improvement of cast nephropathy with plasma exchange depends on the diagnosis and on reduction of serum free light chains. Kidney Int. 73, 1282–1288 (2008).
Ritz, E. Plasma exchange for acute renal failure of myeloma—logical, yet ineffective. J. Am. Soc. Nephrol. 17, 914–916 (2006).
Brockhurst, I., Harris, K. P. & Chapman, C. S. Diagnosis and monitoring a case of light-chain deposition disease in the kidney using a new, sensitive immunoassay. Nephrol. Dial. Transplant. 20, 1251–1253 (2005).
Miettinen, T. A. & Kekki, M. Effect of impaired hepatic and renal function on Bence Jones protein catabolism in human subjects. Clin. Chim. Acta 18, 395–407 (1967).
Wochner, R. D., Strober, W. & Waldmann, T. A. The role of the kidney in the catabolism of Bence Jones proteins and immunoglobulin fragments. J. Exp. Med. 126, 207–221 (1967).
Klassen, R. et al. Light chains are a ligand for megalin. J. Appl. Physiol. 98, 257–263 (2005).
Batuman, V. et al. Myeloma light chains are ligands for cubilin (gp280). Am. J. Physiol. Renal Physiol. 275, F246–F254 (1998).
Hutchison, C. A. et al. Quantitative assessment of serum and urinary polyclonal free light chains in patients with type II diabetes: an early marker of diabetic kidney disease? Expert Opin. Ther. Targets 12, 667–676 (2008).
Hoffman, U. et al. Free immunoglobulin light chains in patients with rheumatic diseases. Z. Rheumatol. 62 (Suppl. 1), 1051 (2003).
Vermeersch, P., Van Hoovels, L., Delforge, M., Godelieve, M. & Bossuyt, X. Diagnostic performance of serum free light chain measurement in patients suspected of a monoclonal B-cell disorder. Br. J. Haematol. 143, 496–502 (2008).
Katzmann, J. A. et al. Elimination of the need for urine studies in the screening algorithm for monoclonal gammopathies by using serum immunofixation and free light chain assays. Mayo Clin. Proc. 81, 1575–1578 (2006).
Hofman, W., Garbrecht, M., Bradwell, A. R. & Guder, W. G. A new concept for detection of Bence Jones proteinuria in patients with monoclonal gammopathy. Clin. Lab. 50, 181–185 (2004).
Beetham, R., Wassell, J., Wallage, M. J., Whiteway, A. J. & James, J. A. Can serum free light chains replace urine electrophoresis in the detection of monoclonal gammopathies? Ann. Clin. Biochem. 44, 516–522 (2007).
Palladini, G. et al. Identification of amyloidogenic light chains requires the combination of serum free light chain assay with immunofixation of serum and urine. Clin. Chem. 55, 499–504 (2009).
Katzmann, J. A. et al. Screening panels for detection of monoclonal gammopathies. Clin. Chem. 55, 1517–1522 (2009).
Holding, S. et al. Combination of serum free light chain analysis with capillary zone electrophoresis improves screening for monoclonal gammopathies [abstract]. Blood 110, 1497 (2007).
Smith, A., Wisloff, F. & Samson, D. On behalf of the UK Myeloma Forum, Nordic Myeloma Study Group and British Committee for Standards in Haematology. Br. J. Haematol. 132, 410–451 (2005).
Kyle, R. A. et al. Prevalence of monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 354, 1362–1369 (2006).
Kyle, R. A. & Rajkumar, S. V. Monoclonal gammopathy of undetermined significance. Br. J. Haematol. 134, 573–589 (2006).
Kyle, R. A. et al. A long term study of prognosis in monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 346, 564–569 (2002).
Hutchison, C. A. et al. Free light chain abnormalities in patients with chronic kidney disease [abstract]. J. Am. Soc. Nephrol. 17, 899a (2006).
Rajkumar, S. V. et al. Serum free light chain ratio is an independent risk factor for progression in monoclonal gammopathy of undetermined significance. Blood 106, 812–817 (2005).
Blade, J. Monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 355, 2765–2770 (2006).
Blade, J. et al. Renal failure in multiple myeloma: presenting features and predictors of outcome in 94 patients from a single institution. Arch. Intern. Med. 158, 1889–1893 (1998).
Sanders, P. W. Pathogenesis and treatment of myeloma kidney. J. Lab. Clin. Med. 124, 484–488 (1994).
Basnayake, K. et al. Resolution of cast nephropathy following free light chain removal by haemodialysis in a patient with multiple myeloma: a case report. J. Med. Case Reports 2, 380 (2008).
Wang, P. X. & Sanders P. W. Immunoglobulin light chains generate hydrogen peroxide. J. Am. Soc. Nephrol. 18, 1239–1245 (2007).
Sanders, P. W. Management of paraproteinemic renal disease. Curr. Opin. Nephrol. Hypertens. 14, 97–103 (2005).
Hutchison, C. A. et al. Treatment of acute renal failure secondary to multiple myeloma with chemotherapy and extended high cut-off hemodialysis. Clin. J. Am. Soc. Nephrol. 4, 745–754 (2009).
Chanan-Khan, A. A. et al. Activity and safety of bortezomib in multiple myeloma patients with advanced renal failure: a multicenter retrospective study. Blood 109, 2604–2606 (2007).
Ludwig, H. et al. Reversal of acute renal failure by bortezomib-based chemotherapy in patients with multiple myeloma. Haematologica 92, 1411–1414 (2007).
Kastritis, E. et al. Reversibility of renal failure in newly diagnosed multiple myeloma patients treated with high dose dexamethasone-containing regimes and the impact of novel agents. Haematologica 92, 546–549 (2007).
Drayson, M. et al. Serum free light-chain measurements for identifying and monitoring patients with nonsecretory multiple myeloma. Blood 97, 2900–2902 (2001).
Sanchorawala, V. et al. AL amyloidosis associated with B-cell lymphoproliferative disorders: frequency and treatment outcomes. Am. J. Hematol. 81, 692–695 (2006).
Rysavá, R. AL amyloidosis with renal involvement. Kidney Blood Press. Res. 30, 359–364 (2007).
Lachmann, H. J. et al. Outcome in systemic AL amyloidosis in relation to changes in concentration of circulating free immunoglobulin light chains following chemotherapy. Br. J. Haematol. 122, 78–84 (2003).
Matsuda, M. et al. Serum levels of free light chains before and after chemotherapy in primary systemic AL amyloidosis. Intern. Med. 44, 428–433 (2005).
Novak, L., Cook, W. J., Herrera, G. A. & Sanders, P. W. AL-amyloidosis is under-diagnosed in renal biopsies. Nephrol. Dial. Transplant. 19, 3050–3053 (2004).
Deret, S. et al. Molecular modeling of immunoglobulin light chains implicates hydrophobic residues in non-amyloid light chain deposition disease. Prot. Eng. 10, 1191–1197 (1997).
Zhu, L. et al. Pathogenesis of glomerulosclerosis in light chain deposition disease. Role for transforming growth factor-beta. Am. J. Pathol. 147, 375–385 (1995).
Pozzi, C. et al. Light chain deposition disease with renal involvement: clinical characteristics and prognostic factors. Am. J. Kidney Dis. 42, 1154–1163 (2003).
Montseny, J. J. et al. Long-term outcome according to renal histological lesions in 118 patients with monoclonal gammopathies. Nephrol. Dial. Transplant. 13, 1438–1445 (1998).
Lin, J. et al. Renal monoclonal immunoglobulin deposition disease: the disease spectrum. J. Am. Soc. Nephrol. 12, 1482–1492 (2001).
Acknowledgements
Charles P. Vega, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the MedscapeCME-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Hutchison, C., Basnayake, K. & Cockwell, P. Serum free light chain assessment in monoclonal gammopathy and kidney disease. Nat Rev Nephrol 5, 621–628 (2009). https://doi.org/10.1038/nrneph.2009.151
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2009.151
This article is cited by
-
Paraproteinämien und Nierenbeteiligung
Die Nephrologie (2022)
-
Light chain monoclonal gammopathy of undetermined significance is characterized by a high disappearance rate and low risk of progression on longitudinal analysis
Annals of Hematology (2018)
-
Prevalence and incidence of anemia in the German Heinz Nixdorf Recall Study
Annals of Hematology (2013)
-
The pathogenesis and diagnosis of acute kidney injury in multiple myeloma
Nature Reviews Nephrology (2012)
-
Prevalence and progression of monoclonal gammopathy of undetermined significance and light-chain MGUS in Germany
Annals of Hematology (2012)