Abstract
Chronic hypoxia induces sequential abnormalities in oxygen metabolism (for example, oxidative stress, nitrosative stress, advanced glycation, carbonyl stress, endoplasmic reticulum stress) in the kidneys of individuals with diabetes. Identification of these abnormalities improves our understanding of therapeutic benefits that can be achieved with antihypertensive agents, the control of hyperglycemia and/or hyperinsulinemia and the dietary correction of obesity. Key to the body's defense against hypoxia is hypoxia-inducible factor, the activity of which is modulated by prolyl hydroxylases (PHDs)—oxygen sensors whose inhibition may prove therapeutic. Renal benefits of small-molecule PHD inhibitors have been documented in several animal models, including those of diabetic nephropathy. Three different PHD isoforms have been identified (PHD1, PHD2 and PHD3) and their respective roles have been delineated in knockout mouse studies. Unfortunately, none of the current inhibitors is specific for a distinct PHD isoform. Nonspecific inhibition of PHDs might induce adverse effects, such as those associated with PHD2 inhibition. Specific disruption of PHD1 induces hypoxic tolerance, without angiogenesis and erythrocytosis, through the reprogramming of basal oxygen metabolism and decreased generation of oxidative stress in hypoxic mitochondria. A specific PHD1 inhibitor might, therefore, offer a novel therapy for abnormal oxygen metabolism not only in the diabetic kidney, but also in other diseases for which hypoxia is a final, common pathway.
Key Points
-
Chronic hypoxia induces sequential abnormalities in oxygen metabolism in the diabetic kidney, leading to oxidative stress, nitrosative stress, advanced glycation, carbonyl stress and endoplasmic reticulum stress
-
Understanding the key features of abnormal oxygen metabolism improves the interpretation of the therapeutic benefits achieved by antihypertensive therapy, the control of hyperglycemia and/or hyperinsulinemia and the dietary correction of obesity
-
Activity of hypoxia-inducible factor—central to the defense against hypoxia—is modulated by prolyl hydroxylases (PHDs), which act as oxygen sensors
-
Three PHD isoforms have been identified and their respective roles have been elucidated, but none of the current PHD inhibitors exhibits absolute specificity for any subtype
-
Disruption of PHD1 induces hypoxic tolerance by reducing oxidative stress in hypoxic mitochondria, indicating that a specific PHD1 inhibitor could be an innovative treatment for abnormal oxygen metabolism in the diabetic kidney
-
Treatment of chronic hypoxia might apply to other chronic diseases that share a final common pathway, including a wide variety of kidney disorders, ischemic heart disease, and stroke
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Dronavalli, S., Duka, I. & Bakris, G. L. The pathogenesis of diabetic nephropathy. Nat. Clin. Pract. Endocrinol. Metab. 4, 444–452 (2008).
Williamson, J. R. et al. Hyperglycemic pseudohypoxia and diabetic complications. Diabetes 42, 801–813 (1993).
Bravi, M. C. et al. Polyol pathway activation and glutathione redox status in non-insulin-dependent diabetic patients. Metabolism 46, 1194–1198 (1997).
Jennings, P. E., Chirico, S., Jones, A. F., Lunec, J. & Barnett, A. H. Vitamin C metabolites and microangiopathy in diabetes mellitus. Diabetes Res. 6, 151–154 (1987).
Suzuki, E. et al. Increased oxidized form of human serum albumin in patients with diabetes mellitus. Diabetes Res. Clin. Pract. 18, 153–158 (1992).
Wells-Knecht, M. C., Lyons, T. J., McCance, D. R., Thorpe, S. R. & Baynes, J. W. Age-dependent increase in ortho-tyrosine and methionine sulfoxide in human skin collagen is not accelerated in diabetes. Evidence against a generalized increase in oxidative stress in diabetes. J. Clin. Invest. 100, 839–846 (1997).
Horie, K. et al. Immunohistochemical co-localization of glycoxidation products and lipid peroxidation products in diabetic renal glomerular lesions. Implication for glycoxidative stress in the pathogenesis of diabetic nephropathy. J. Clin. Invest. 100, 2995–3004 (1997).
Suzuki, D. et al. Immunohistochemical evidence for an increased oxidative stress and carbonyl modification of proteins in diabetic glomerular lesions. J. Am. Soc. Nephrol. 10, 822–832 (1999).
Pennathur, S., Wagner, J. D., Leeuwenburgh, C., Litwak, K. N. & Heinecke, J. W. A hydroxyl radical-like species oxidizes cynomolgus monkey artery wall proteins in early diabetic vascular disease. J. Clin. Invest. 107, 853–860 (2001).
Shah, S. V., Baliga, R., Rajapurkar, M. & Fonseca, V. A. Oxidants in chronic kidney disease. J. Am. Soc. Nephrol. 18, 16–28 (2007).
Forbes, J. M., Coughlan, M. T. & Cooper, M. E. Oxidative stress as a major culprit in kidney disease in diabetes. Diabetes 57, 1446–1454 (2008).
Jaimes, E. A., Galceran, J. M. & Raij, L. Angiotensin II induces superoxide anion production by mesangial cells. Kidney Int. 54, 775–784 (1998).
Izuhara, Y. et al. Renoprotective properties of angiotensin receptor blockers beyond blood pressure lowering. J. Am. Soc. Nephrol. 16, 3631–3641 (2005).
Gorin, Y. et al. Nox4 NAD(P)H oxidase mediates hypertrophy and fibronectin expression in the diabetic kidney. J. Biol. Chem. 280, 39616–39626 (2005).
Asaba, K. et al. Effects of NADPH oxidase inhibitor in diabetic nephropathy. Kidney Int. 67, 1890–1898 (2005).
Thallas-Bonke, V. et al. Inhibition of NADPH oxidase prevents advanced glycation end product-mediated damage in diabetic nephropathy through a protein kinase C-alpha-dependent pathway. Diabetes 57, 460–469 (2008).
Szabo, C. Role of nitrosative stress in the pathogenesis of diabetic vascular dysfunction. Br. J. Pharmacol. 156, 713–727 (2009).
Nishikawa, T. et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycemic damage. Nature 404, 787–790 (2000).
Kiritoshi, S. et al. Reactive oxygen species from mitochondria induce cyclooxygenase-2 gene expression in human mesangial cells: potential role in diabetic nephropathy. Diabetes 52, 2570–2577 (2003).
Tan, A. L., Forbes, J. M. & Cooper, M. E. AGE, RAGE, and ROS in diabetic nephropathy. Semin. Nephrol. 27, 130–143 (2007).
Monnier, V. M. Transition metals redox: reviving an old plot for diabetic vascular disease. J. Clin. Invest. 107, 799–801 (2001).
Baynes, J. W. & Thorpe, S. R. Role of oxidative stress in diabetic complications: a new perspective on an old paradigm. Diabetes 48, 1–9 (1999).
Aragonés, J. et al. Deficiency or inhibition of oxygen sensor Phd1 induces hypoxia tolerance by reprogramming basal metabolism. Nat. Genet. 40, 170–180 (2008).
Kim, J. W., Tchernyshyov, I., Semenza, G. L. & Dang, C. V. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 3, 177–185 (2006).
Bonnet, S. et al. A mitochondria-K+ channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth. Cancer Cell 11, 37–51 (2007).
Kulkarni, A. C., Kuppusamy, P. & Parinandi, N. Oxygen, the lead actor in the pathophysiologic drama: enactment of the trinity of normoxia, hypoxia, and hyperoxia in disease and therapy. Antioxid. Redox. Signal. 9, 1717–1730 (2007).
Katavetin, P. et al. High glucose blunts vascular endothelial growth factor response to hypoxia via the oxidativestress-regulated hypoxia-inducible factor/hypoxia-responsible element pathway. J. Am. Soc. Nephrol. 17, 1405–1413 (2006).
Rosenberger, C. et al. Adaptation to hypoxia in the diabetic rat kidney. Kidney Int. 73, 34–42 (2008).
Yang, Z. Z., Zhang, A. Y., Yi, F. X., Li, P. L. & Zou, A. P. Redox regulation of HIF-1alpha levels and HO-1 expression in renal medullary interstitial cells. Am. J. Physiol. Renal Physiol. 284, F1207–F1215 (2003).
Fine, L. G., Orphanides, C. & Norman, J. T. Progressive renal disease: the chronic hypoxia hypothesis. Kidney Int. Suppl. 65, S74–S78 (1998).
Nangaku, M. Chronic hypoxia and tubulointerstitial injury: a final common pathway to end-stage renal failure. J. Am. Soc. Nephrol. 17, 17–25 (2006).
Singh, D. K., Winocour, P. & Farrington, K. Mechanisms of disease: the hypoxic tubular hypothesis of diabetic nephropathy. Nat. Clin. Pract. Nephrol. 4, 216–226 (2008).
Ries, M. et al. Renal diffusion and BOLD MRI in experimental diabetic nephropathy. Blood oxygen level-dependent. J. Magn. Reson. Imaging 17, 104–113 (2003).
Bernhardt, W. M. et al. Expression of hypoxia-inducible transcription factors in developing human and rat kidneys. Kidney Int. 69, 114–122 (2006).
Rosenberger, C. et al. Hypoxia-inducible factors and tubular cell survival in isolated perfused kidneys. Kidney Int. 70, 60–70 (2006).
Tanaka, T., Miyata, T., Inagi, R., Fujita, T. & Nangaku, M. Hypoxia in renal disease with proteinuria and/or glomerular hypertension. Am. J. Pathol. 165, 1979–1992 (2004).
Singh, P., Deng, A., Weir, M. R. & Blantz, R. C. The balance of angiotensin II and nitric oxide in kidney diseases. Curr. Opin. Nephrol. Hypertens. 17, 51–56 (2008).
Palm, F. et al. Reduced nitric oxide in diabetic kidneys due to increased hepatic arginine metabolism: implications for renomedullary oxygen availability. Am. J. Physiol. Renal Physiol. 294, F30–F37 (2008).
Nangaku, M. & Eckardt, K. U. Pathogenesis of renal anemia. Semin. Nephrol. 26, 261–268 (2006).
Bahlmann, F. H. & Fliser, D. Erythropoietin and renoprotection. Curr. Opin. Nephrol. Hypertens. 18, 15–20 (2009).
Nath, K. A., Croat, A. J. & Hostettor, T. H. Oxygen consumption and oxidant stress in surviving nephrons. Am. J. Physiol. 258, F1354–F1362 (1990).
Modlinger, P. S., Wilcox, C. S. & Aslam, S. Nitric oxide, oxidative stress, and progression of chronic renal failure. Semin. Nephrol. 24, 354–365 (2004).
Wilcox, C. S. Oxidative stress and nitric oxide deficiency in the kidney: a critical link to hypertension? Am. J. Physiol. Regul. Integr. Comp. Physiol. 289, R913–R935 (2005).
Prabhakar, S., Starnes, J., Shi, S., Lonis, B. & Tran, R. Diabetic nephropathy is associated with oxidative stress and decreased renal nitric oxide production. J. Am. Soc. Nephrol. 18, 2945–2952 (2007).
Palm, F., Teerlink, T. & Hansell, P. Nitric oxide and kidney oxygenation. Curr. Opin. Nephrol. Hypertens. 18, 68–73 (2009).
Garcia, S. F. et al. Diabetic endothelial dysfunction: the role of poly(ADP-ribose) polymerase activation. Nat. Med. 7, 108–113 (2001).
Du, X. et al. Inhibition of GAPDH activity by poly(ADP-ribose) polymerase activates three major pathways of hyperglycemic damage in endothelial cells. J. Clin. Invest. 112, 1049–1057 (2003).
Miyata, T., Kurokawa, K. & van Ypersele de Strihou, C. From molecular footprints of disease to new therapeutic interventions in diabetic nephropathy: a detective story (review). Curr. Drug Targets Immune Endocr. Metabol. Disord. 5, 323–329 (2005).
Miyata, T. et al. Implication of an increased oxidative stress in the formation of advanced glycation end products in patients with end-stage renal failure. Kidney Int. 51, 1170–1181 (1997).
Witko-Sarsat, V. et al. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 49, 1304–1313 (1996).
Miyata, T., Kurokawa, K. & van Ypersele de Strihou, C. Advanced glycation and lipoxidation end products: Role of reactive carbonyl compounds generated during carbohydrate and lipid metabolism (editorial review). J. Am. Soc. Nephrol. 11, 1744–1752 (2000).
Yan, S. D. et al. Enhanced cellular oxidant stress by the interaction of advanced glycation end products with their receptors/binding proteins. J. Biol. Chem. 269, 9889–9897 (1994).
Friederich, M., Hansell, P. & Palm, F. Diabetes, oxidative stress, nitric oxide and mitochondria function. Curr. Diabetes Rev. 5, 120–144 (2009).
Xu, C., Bailly-Maitre, B. & Reed, J. C. Endoplasmic reticulum stress: cell life and death decisions. J. Clin. Invest. 115, 2656–2664 (2005).
Lindenmeyer, M. T. et al. Proteinuria and hyperglycemia induce endoplasmic reticulum stress. J. Am. Soc. Nephrol. 19, 2225–2236 (2008).
Jafar, T. H. et al. Angiotensin-converting enzyme inhibitors and progression of nondiabetic renal disease. A meta-analysis of patient-level data. Ann. Intern. Med. 135, 73–87 (2001).
Lewis, E. J. et al. Collaborative Study Group. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N. Engl. J. Med. 345, 851–860 (2001).
Brenner, B. M. et al. RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N. Engl. J. Med. 345, 861–869 (2001).
Parving, H. H. et al. Irbesartan in Patients with type 2 diabetes and Microalbuminuria Study Group. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N. Engl. J. Med. 345, 870–878 (2001).
Viberti, G. & Wheeldon, N. M. Microalbuminuria reduction with valsartan (MARVAL) Study Investigators. Microalbuminuria reduction with valsartan in patients with type 2 diabetes mellitus. A blood pressure-independent effect. Circulation 106, 672–678 (2002).
Parving, H. H. et al. Aliskiren combined with losartan in type 2 diabetes and nephropathy. N. Engl. J. Med. 358, 2433–2446 (2008).
Seikaly, M. G., Arant, B. S. & Seney, F. D. Endogenous angiotensin concentrations in specific intrarenal fluid compartments of the rat. J. Clin. Invest. 86, 1352–1357 (1990).
Manotham, K. et al. Evidence of tubular hypoxia in the early phase in the remnant kidney model. J. Am. Soc. Nephrol. 15, 1277–1288 (2004).
Palm, F., Connors, S. G., Mendonca, M., Welch, W. J. & Wilcox, C. S. Angiotensin II type 2 receptors and nitric oxide sustain oxygenation in the clipped kidney of early Goldblatt hypertensive rats. Hypertension 51, 345–351 (2008).
Onozato, M. L., Tojo, A., Goto, A., Fujita, T. & Wilcox, C. S. Oxidative stress and nitric oxide synthase in rat diabetic nephropathy: effects of ACEI and ARB. Kidney Int. 61, 186–194 (2002).
Benigni, A. et al. Disruption of the Ang II type 1 receptor promotes longevity in mice. J. Clin. Invest. 119, 524–530 (2009).
Miyata, T. et al. Angiotensine II receptor antagonist and angiotensin converting enzyme (ACE) inhibitor scavenge oxidative radicals and lower the formation of advanced glycation end products. J. Am. Soc. Nephrol. 13, 2478–2487 (2002).
Nangaku, M. et al. Anti-hypertensive agents inhibit in vivo the formation of advanced glycation end products and improve renal damage in a type 2 diabetic nephropathy rat model. J. Am. Soc. Nephrol. 14, 1212–1222 (2003).
Chen, S. et al. Candesartan suppresses chronic renal inflammation by a novel antioxidant action independent of AT1R blockade. Kidney Int. 74, 1128–1138 (2008).
Sun, H. L. et al. ACE-inhibitor suppresses the apoptosis induced by endoplasmic reticulum stress in renal tubular in experimental diabetic rats. Exp. Clin. Endocrinol. Diabetes 117, 336–344 (2009).
Izuhara, Y. et al. A novel sartan derivative with very low angiotensin II type 1 receptor affinity protects the kidney in type 2 diabetic rats. Arterioscler. Thromb. Vasc. Biol. 28, 1767–1773 (2008).
Takizawa, S. et al. A sartan derivative with a very low angiotensin II receptor affinity ameliorates ischemic cerebral damage. J. Cereb. Blood Flow Metab. 29, 1665–1672 (2009).
Writing Team for the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA 290, 2159–2167 (2003).
Sarafidis, P. A. & Ruilope, L. M. Insulin resistance, hyperinsulinemia, and renal injury: mechanisms and implications. Am. J. Nephrol. 26, 232–234 (2006).
Makino, H. et al. Altered gene expression related to glomerulogenesis and podocyte structure in early diabetic nephropathy of db/db mice and its restoration by pioglitazone. Diabetes 55, 2747–2756 (2006).
Ohtomo, S. et al. Thiazolidinediones provide better renoprotection than insulin in an obese, hypertensive type II diabetic rat model. Kidney Int. 72, 1512–1519 (2007).
Rodriguez, W. E. et al. Pioglitazone mitigates renal glomerular vascular changes in high-fat, high-calorie-induced type 2 diabetes mellitus. Am. J. Physiol. Renal Physiol. 291, F694–F701 (2006).
Perico, N. & Remuzzi, G. Inhibition of TGF-beta expression: a novel role for thiazolidinediones to implement renoprotection in diabetes. Kidney Int. 72, 1419–1421 (2007).
Gredilla, R. & Barja, G. Minireview: the role of oxidative stress in relation to caloric restriction and longevity. Endocrinology 146, 3713–3717 (2005).
Cai, W. et al. Oral glycotoxins determine the effects of calorie restriction on oxidant stress, age-related diseases, and lifespan. Am. J. Pathol. 173, 327–336 (2008).
Trayhurn, P., Wang, B. & Wood, I. S. Hypoxia in adipose tissue: a basis for the dysregulation of tissue function in obesity? Br. J. Nutr. 100, 227–235 (2008).
Ye, J. Emerging role of adipose tissue hypoxia in obesity and insulin resistance. Int. J. Obes. (Lond.) 33, 54–66 (2009).
Crujeiras, A. B. et al. Energy restriction in obese subjects impact differently two mitochondrial function markers. J. Physiol. Biochem. 64, 211–219 (2008).
Nangaku, M. et al. In a type 2 diabetic nephropathy rat model, the improvement of obesity by a low calorie diet reduces oxidative/carbonyl stress and prevents diabetic nephropathy. Nephrol. Dial. Transplant. 20, 2661–2669 (2005).
Miyata, T. & van Ypersele de Strihou, C. Translation of basic science into clinical medicine: novel targets for diabetic nephropathy. Nephrol. Dial. Transplant. 24, 1373–1377 (2009).
Semenza, G. L. Targeting HIF-1 for cancer therapy. Nat. Rev. Cancer 3, 721–732 (2003).
Marx, J. How cells endure low oxygen. Science 303, 1454–1456 (2004).
Epstein, A. C. et al. Elegans EGL-9 and mammalian homologs define a family of dioxygenases that regulate HIF by prolyl hydroxylation. Cell 107, 43–54 (2001).
Schofield, C. J. & Ratcliffe, P. J. Oxygen sensing by HIF hydroxylases. Nat. Rev. Mol. Cell Biol. 5, 343–354 (2004).
Hon, W. C. et al. Structural basis for the recognition of hydroxyproline in HIF-1 alpha by pVHL. Nature 417, 975–978 (2002).
Ivan, M. et al. HIFa targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing. Science 292, 464–468 (2001).
Maxwell, P. H. et al. The tumor suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399, 271–275 (1999).
Ohh, M. et al. Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel–Lindau protein. Nat. Cell Biol. 2, 423–427 (2000).
Eguchi, H., Ikuta, T., Tachibana, T., Yoneda, Y. & Kawajiri, K. A nuclear localization signal of human aryl hydrocarbon receptor nuclear translocator/hypoxia-inducible factor 1beta is a novel bipartite type recognized by the two components of nuclear pore-targeting complex. J. Biol. Chem. 272, 17640–17647 (1997).
Kaelin, W. G. Jr & Ratcliffe, P. J. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol. Cell 30, 393–402 (2008).
Takeda, K. et al. Regulation of adult erythropoiesis by prolyl hydroxylase domain proteins. Blood 113, 3229–3235 (2008).
Percy, M. J. et al. A gain-of-function mutation in the HIF2A gene in familial erythrocytosis. N. Engl. J. Med. 358, 162–168 (2008).
Bernhardt, W. M. et al. Involvement of hypoxia-inducible transcription factors in polycystic kidney disease. Am. J. Pathol. 170, 830–842 (2007).
Kojima, I. et al. Protective role of hypoxia-inducible factor-2alpha against ischemic damage and oxidative stress in the kidney. J. Am. Soc. Nephrol. 18, 1218–1226 (2007).
Rosenberger, C. et al. Evidence for sustained renal hypoxia and transient hypoxia adaptation in experimental rhabdomyolysis-induced acute kidney injury. Nephrol. Dial. Transplant. 23, 1135–1143 (2008).
Hill, P. et al. Inhibition of hypoxia inducible factor hydroxylases protects against renal ischemia-reperfusion injury. J. Am. Soc. Nephrol. 19, 39–46 (2008).
Schofield, C. J. & Ratcliffe, P. J. Oxygen sensing by HIF hydroxylases. Nat. Rev. Mol. Cell Biol. 5, 343–354 (2004).
Koivunen, P., Hirsilä, M., Günzler, V., Kivirikko, K. I. & Myllyharju, J. Catalytic properties of the asparaginyl hydroxylase (FIH) in the oxygen sensing pathway are distinct from those of its prolyl 4-hydroxylases. J. Biol. Chem. 279, 9899–9904 (2004).
Fraisl, P., Aragonés, J. & Carmeliet, P. Inhibition of oxygen sensors as a therapeutic strategy for ischaemic and inflammatory disease. Nat. Rev. Drug Discov. 8, 139–152 (2009).
Salnikow, K. et al. Depletion of intracellular ascorbate by the carcinogenic metals nickel and cobalt results in the induction of hypoxic stress. J. Biol. Chem. 279, 40337–40344 (2004).
Matsumoto, M. et al. Induction of renoprotective gene expression by cobalt ameliorates ischemic injury of the kidney in rats. J. Am. Soc. Nephrol. 14, 1825–1832 (2003).
Tanaka, T. et al. Induction of protective genes by cobalt ameliorates tubulointerstitial injury in the progressive Thy1 nephritis. Kidney Int. 68, 2714–2725 (2005).
Tanaka, T. et al. Cobalt promotes angiogenesis via hypoxia-inducible factor and protects tubulointerstitium in the remnant kidney model. Lab. Invest. 85, 1292–1307 (2005).
Tanaka, T. et al. Hypoxia-inducible factor modulates tubular cell survival in cisplatin nephrotoxicity. Am. J. Physiol. Renal Physiol. 289, F1123–F1133 (2005).
Ohtomo, S. et al. Cobalt ameliorates renal injury in an obese, hypertensive type 2 diabetes rat model. Nephrol. Dial. Transplant. 23, 1166–1172 (2008).
Smith, E. L. Presence of cobalt in the anti-pernicious anemia factor. Nature 162, 144 (1948).
Berk, L., Burchebal, J. H. & Castle, W. B. Erythropoietic effect of cobalt in patients with or without anemia. N. Engl. J. Med. 240, 754–761 (1949).
Edwards, M. S. & Curtis, J. R. Use of cobaltous chloride in anemia of maintenance hemodialysis patients. Lancet 2, 582–583 (1971).
Ivan, M. et al. Biochemical purification and pharmacological inhibition of a mammalian prolyl hydroxylase acting on hypoxia-inducible factor. Proc. Natl Acad. Sci. USA 99, 13459–13464 (2002).
Mole, D. R. et al. 2-oxoglutarate analog inhibitors of HIF prolyl hydroxylase. Bioorg. Med. Chem. Lett. 13, 2677–2680 (2003).
McDonough, M. A. et al. Selective inhibition of factor inhibiting hypoxia-inducible factor. J. Am. Chem. Soc. 127, 7680–7681 (2005).
Nangaku, M. et al. A novel class of prolyl hydroxylase inhibitors induces angiogenesis and exerts organ protection against ischemia. Arterioscler. Thromb. Vasc. Biol. 27, 2548–2554 (2007).
Bernhardt, W. M. et al. Preconditional activation of hypoxia-inducible factors ameliorates ischemic acute renal failure. J. Am. Soc. Nephrol. 17, 1970–1978 (2006).
Watanabe, D. et al. Erythropoietin as a retinal angiogenic factor in proliferative diabetic retinopathy. N. Engl. J. Med. 353, 782–792 (2005).
Metzen, E. et al. Intracellular localisation of human HIF-1 alpha hydroxylases: implications for oxygen sensing. J. Cell. Sci. 116, 1319–1326 (2003).
Steinhoff, A. et al. Cellular oxygen sensing: Importins and exportins are mediators of intracellular localisation of prolyl-4-hydroxylases PHD1 and PHD2. Biochem. Biophys. Res. Commun. 387, 705–711 (2009).
Berchner-Pfannschmidt, U. et al. Nuclear oxygen sensing: induction of endogenous prolyl-hydroxylase 2 activity by hypoxia and nitric oxide. J. Biol. Chem. 283, 31745–31753 (2008).
Fong, G. H. & Takeda, K. Role and regulation of prolyl hydroxylase domain proteins. Cell Death Differ. 15, 635–641 (2008).
Tug, S., Delos, R. B., Fandrey, J. & Berchner-Pfannschmidt, U. Non-hypoxic activation of the negative regulatory feedback loop of prolyl-hydroxylase oxygen sensors. Biochem. Biophys. Res. Commun. 384, 519–523 (2009).
Berchner-Pfannschmidt, U., Yamac, H., Trinidad, B. & Fandrey, J. Nitric oxide modulates oxygen sensing by hypoxia-inducible factor 1-dependent induction of prolyl hydroxylase 2. J. Biol. Chem. 282, 1788–1796 (2007).
Schödel, J. et al. HIF-prolyl hydroxylases in the rat kidney: physiologic expression patterns and regulation in acute kidney injury. Am. J. Pathol. 174, 1663–1674 (2009).
Takeda, K., Cowan, A. & Fong, G. H. Essential role for prolyl hydroxylase domain protein 2 in oxygen homeostasis of the adult vascular system. Circulation 116, 774–781 (2007).
Loinard, C. et al. Inhibition of prolyl hydroxylase domain proteins promotes therapeutic revascularization. Circulation 120, 50–59 (2009).
Pouysségur, J. et al. Hypoxia signaling in cancer and approaches to enforce tumor regression. Nature 441, 437–443 (2006).
Mazzone, M. et al. Heterozygous deficiency of PHD2 restores tumor oxygenation and inhibits metastasis via endothelial normalization. Cell 136, 839–851 (2009).
Cummins, E. P. et al. Prolyl hydroxylase-1 negatively regulates IkappaB kinase-beta, giving insight into hypoxia-induced NFkappaB activity. Proc. Natl Acad. Sci. USA 103, 18154–18159 (2006).
Mikhaylova, O. et al. The von Hippel–Lindau tumor suppressor protein and Egl-9-Type proline hydroxylases regulate the large subunit of RNA polymerase II in response to oxidative stress. Mol. Cell. Biol. 28, 2701–2717 (2008).
Baranova, O. et al. Neuron-specific inactivation of the hypoxia inducible factor 1 alpha increases brain injury in a mouse model of transient focal cerebral ischemia. J. Neurosci. 27, 6320–6332 (2007).
Koditz, J. et al. Oxygen-dependent ATF-4 stability is mediated by the PHD3 oxygen sensor. Blood 110, 3610–3617 (2007).
Natarajan, R. et al. Prolyl hydroxylase inhibition attenuates post-ischemic cardiac injury via induction of endoplasmic reticulum stress genes. Vascul. Pharmacol. 51, 110–128 (2009).
Tong, K. I., Kobayashi, A., Katsuoka, F. & Yamamoto, M. Two-site substrate recognition model for the Keap1-Nrf2 system: a hinge and latch mechanism. Biol. Chem. 387, 1311–1320 (2006).
Zhou, F. et al. Hibernation, a model of neuroprotection. Am. J. Pathol. 158, 2145–2151 (2001).
Drew, K. L. et al. Hypoxia tolerance in mammalian heterotherms. J. Exp. Biol. 207, 3155–3162 (2004).
Siddiq, A. et al. Selective inhibition of hypoxia-inducible factor (HIF) prolyl-hydroxylase 1 mediates neuroprotection against normoxic oxidative death via HIF- and CREB-independent pathways. J. Neurosci. 29, 8828–8838 (2009).
Minamishima, Y. A. et al. Somatic inactivation of the PHD2 prolyl hydroxylase causes polycythemia and congestive heart failure. Blood 111, 3236–3244 (2008).
Ladroue, C. et al. PHD2 mutation and congenital erythrocytosis with paraganglioma. N. Engl. J Med. 359, 2685–2692 (2008).
Takeda, K. et al. Placental but not heart defects are associated with elevated hypoxia-inducible factor alpha levels in mice lacking prolyl hydroxylase domain protein 2. Mol. Cell. Biol. 26, 8336–8346 (2006).
Chen, J. X. & Stinnett, A. Ang-1 gene therapy inhibits hypoxia-inducible factor-1alpha (HIF-1alpha)-prolyl-4-hydroxylase-2, stabilizes HIF-1alpha expression, and normalizes immature vasculature in db/db mice. Diabetes 57, 3335–3343 (2008).
Fuchshofer, R. et al. Hypoxia/reoxygenation induces CTGF and PAI-1 in cultured human retinal pigment epithelium cells. Exp. Eye Res. 88, 889–899 (2009).
Kroening, S., Neubauer, E., Wessel, J., Wiesener, M. & Goppelt-Struebe, M. Hypoxia interferes with connective tissue growth factor (CTGF) gene expression in human proximal tubular cell lines. Nephrol. Dial. Transplant. 24, 3319–3325 (2009).
Ha, H., Oh, E. Y. & Lee, H. B. The role of plasminogen activator inhibitor 1 in renal and cardiovascular diseases. Nat. Rev. Nephrol. 5, 203–211 (2009).
Izuhara, Y. et al. Inhibition of plasminogen activator inhibitor-1: its mechanism and effectiveness on coagulation and fibrosis. Arterioscler. Thromb. Vasc. Biol. 28, 672–677 (2008).
Schwartz, S. et al. Phase 1 study of FG-3019, an anti-CTGF monoclonal antibody, in type 1/2 diabetes mellitus with microalbuminuria (abstract). Diabetes 56 (Suppl.1), A151 (2007).
Acknowledgements
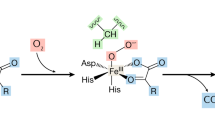
We thank Dr. Guo-Hua Fong for critically reviewing the manuscript, Drs. Masaomi Nangaku and Kotaro Takeda for helpful discussion, Dr. Noriaki Hirayama for kindly providing a representative picture of docking simulation (human PHD2 and PHD inhibitors), and Dr. Masashi Okamura for help with the preparation of figures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Miyata, T., van Ypersele de Strihou, C. Diabetic nephropathy: a disorder of oxygen metabolism?. Nat Rev Nephrol 6, 83–95 (2010). https://doi.org/10.1038/nrneph.2009.211
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2009.211
This article is cited by
-
Beraprost Sodium Delays the Decline of Glomerular Filtration Rate in Patients with Diabetic Nephropathy: A Retrospective Study
Diabetes Therapy (2023)
-
Down-regulation of Risa improves podocyte injury by enhancing autophagy in diabetic nephropathy
Military Medical Research (2022)
-
Maslinic acid activates renal AMPK/SIRT1 signaling pathway and protects against diabetic nephropathy in mice
BMC Endocrine Disorders (2022)
-
Rice bran protein hydrolysates attenuate diabetic nephropathy in diabetic animal model
European Journal of Nutrition (2018)
-
Radiologic imaging of the renal parenchyma structure and function
Nature Reviews Nephrology (2016)