Abstract
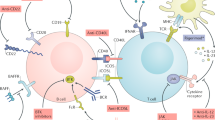
Suboptimal trial design and concurrent therapies are thought to account for the unexpected failure of two clinical trials of rituximab in patients with systemic lupus erythematosus (SLE). However, in this Opinion article we propose an alternative explanation: that rituximab can trigger a sequence of events that exacerbates disease in some patients with SLE. Post-rituximab SLE flares that are characterized by high levels of antibodies to double-stranded DNA are associated with elevated circulating BAFF (B-cell-activating factor, also known as TNF ligand superfamily member 13B or BLyS) levels, and a high proportion of plasmablasts within the B-cell pool. BAFF not only perpetuates autoreactive B cells (including plasmablasts), particularly when B-cell numbers are low, but also stimulates T follicular helper (TFH) cells. Moreover, plasmablasts and TFH cells promote each others' formation. Thus, repeated rituximab infusions can result in a feedback loop characterized by ever-rising BAFF levels, surges in autoantibody production and worsening of disease. We argue that B-cell depletion should be swiftly followed by BAFF inhibition in patients with SLE.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Thomas, G. et al. Mortality associated with systemic lupus erythematosus in France assessed by multiple cause-of-death analysis: the MORTALUP Study. Arthritis Rheumatol. 66, 2503–2511 (2014).
Furtado, J. & Isenberg, D. A. B cell elimination in systemic lupus erythematosus. Clin. Immunol. 146, 90–103 (2013).
Merrill, J. T. et al. Efficacy and safety of rituximab in moderately-to-severely active systemic lupus erythematosus: the randomized, double-blind, phase II/III systemic lupus erythematosus evaluation of rituximab trial. Arthritis Rheum. 62, 222–233 (2010).
Merrill, J. et al. Assessment of flares in lupus patients enrolled in a phase II/III study of rituximab (EXPLORER). Lupus 20, 709–716 (2011).
Rovin, B. H. et al. Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: the Lupus Nephritis Assessment with Rituximab study. Arthritis Rheum. 64, 1215–1226 (2012).
Lazarus, M. N., Turner-Stokes, T., Chavele, K. M., Isenberg, D. A. & Ehrenstein, M. R. B-cell numbers and phenotype at clinical relapse following rituximab therapy differ in SLE patients according to anti-dsDNA antibody levels. Rheumatology (Oxford) 51, 1208–1215 (2012).
Vital, E. M. et al. B cell biomarkers of rituximab responses in systemic lupus erythematosus. Arthritis Rheum. 63, 3038–3047 (2011).
Odendahl, M. et al. Disturbed peripheral B lymphocyte homeostasis in systemic lupus erythematosus. J. Immunol. 165, 5970–5979 (2000).
Jacobi, A. M. et al. Correlation between circulating CD27high plasma cells and disease activity in patients with systemic lupus erythematosus. Arthritis Rheum. 48, 1332–1342 (2003).
Bosma, A., Abdel-Gadir, A., Isenberg, D. A., Jury, E. C. & Mauri, C. Lipid-antigen presentation by CD1d+ B cells is essential for the maintenance of invariant natural killer T cells. Immunity 36, 477–490 (2012).
Chavele, K. M., Merry, E. & Ehrenstein, M. R. Cutting edge: circulating plasmablasts induce the differentiation of human T follicular helper cells via IL-6 production. J. Immunol. 194, 2482–2485 (2015).
de Wit, J. et al. Human B cells promote T-cell plasticity to optimize antibody response by inducing coexpression of TH1/TFH signatures. J. Allergy Clin. Immunol. 135, 1053–1060 (2015).
Karnowski, A. et al. B and T cells collaborate in antiviral responses via IL-6, IL-21, and transcriptional activator and coactivator, Oct2 and OBF-1. J. Exp. Med. 209, 2049–2064 (2012).
Feng, X. et al. Inhibition of aberrant circulating TFH cell proportions by corticosteroids in patients with systemic lupus erythematosus. PLoS ONE 7, e51982 (2012).
Simpson, N. et al. Expansion of circulating T cells resembling follicular helper T cells is a fixed phenotype that identifies a subset of severe systemic lupus erythematosus. Arthritis Rheum. 62, 234–244 (2010).
Choi, J. Y. et al. Circulating follicular helper-like T cells in systemic lupus erythematosus: association with disease activity. Arthritis Rheumatol. 67, 988–999 (2015).
Zhang, X. et al. Circulating CXCR5+CD4+ helper T cells in systemic lupus erythematosus patients share phenotypic properties with germinal center follicular helper T cells and promote antibody production. Lupus 24, 909–917 (2015).
Liarski, V. M. et al. Cell distance mapping identifies functional T follicular helper cells in inflamed human renal tissue. Sci. Transl. Med. 6, 230ra46 (2014).
Linterman, M. A. et al. Follicular helper T cells are required for systemic autoimmunity. J. Exp. Med. 206, 561–576 (2009).
Kim, Y. U., Lim, H., Jung, H. E., Wetsel, R. A. & Chung, Y. Regulation of autoimmune germinal center reactions in lupus-prone BXD2 mice by follicular helper T cells. PLoS ONE 10, e0120294 (2015).
Vital, E. M., Dass, S., Buch, M. H., Rawstron, A. C. & Emery, P. An extra dose of rituximab improves clinical response in rheumatoid arthritis patients with initial incomplete B cell depletion: a randomised controlled trial. Ann. Rheum. Dis. 74, 1195–1201 (2015).
Carter, L. M., Isenberg, D. A. & Ehrenstein, M. R. Elevated serum BAFF levels are associated with rising anti-double-stranded DNA antibody levels and disease flare following B cell depletion therapy in systemic lupus erythematosus. Arthritis Rheum. 65, 2672–2679 (2013).
Cancro, M. P., D'Cruz, D. P. & Khamashta, M. A. The role of B lymphocyte stimulator (BLyS) in systemic lupus erythematosus. J. Clin. Invest. 119, 1066–1073 (2009).
Cambridge, G. et al. B cell depletion therapy in systemic lupus erythematosus: relationships among serum B lymphocyte stimulator levels, autoantibody profile and clinical response. Ann. Rheum. Dis. 67, 1011–1016 (2008).
Cambridge, G. et al. Circulating levels of B lymphocyte stimulator in patients with rheumatoid arthritis following rituximab treatment: relationships with B cell depletion, circulating antibodies, and clinical relapse. Arthritis Rheum. 54, 723–732 (2006).
Thien, M. et al. Excess BAFF rescues self-reactive B cells from peripheral deletion and allows them to enter forbidden follicular and marginal zone niches. Immunity 20, 785–798 (2004).
Lesley, R. et al. Reduced competitiveness of autoantigen-engaged B cells due to increased dependence on BAFF. Immunity 20, 441–453 (2004).
Thorn, M., Lewis, R. H., Mumbey-Wafula, A., Kantrowitz, S. & Spatz, L. A. BAFF overexpression promotes anti-dsDNA B-cell maturation and antibody secretion. Cell. Immunol. 261, 9–22 (2010).
Liu, Z. & Davidson, A. BAFF and selection of autoreactive B cells. Trends Immunol. 32, 388–394 (2011).
Avery, D. T. et al. BAFF selectively enhances the survival of plasmablasts generated from human memory B cells. J. Clin. Invest. 112, 286–297 (2003).
Balázs, M., Martin, F., Zhou, T. & Kearney, J. Blood dendritic cells interact with splenic marginal zone B cells to initiate T-independent immune responses. Immunity 17, 341–352 (2002).
Joo, H. et al. Serum from patients with SLE instructs monocytes to promote IgG and IgA plasmablast differentiation. J. Exp. Med. 209, 1335–1348 (2012).
Mackay, F. et al. Mice transgenic for BAFF develop lymphocytic disorders along with autoimmune manifestations. J. Exp. Med. 190, 1697–1710 (1999).
Khare, S. D. et al. Severe B cell hyperplasia and autoimmune disease in TALL-1 transgenic mice. Proc. Natl Acad. Sci. USA 97, 3370–3375 (2000).
Gross, J. A. et al. TACI and BCMA are receptors for a TNF homologue implicated in B-cell autoimmune disease. Nature 404, 995–999 (2000).
Cheema, G. S., Roschke, V., Hilbert, D. M. & Stohl, W. Elevated serum B lymphocyte stimulator levels in patients with systemic immune-based rheumatic diseases. Arthritis Rheum. 44, 1313–1319 (2001).
Stohl, W. et al. B lymphocyte stimulator overexpression in patients with systemic lupus erythematosus: longitudinal observations. Arthritis Rheum. 48, 3475–3486 (2003).
Petri, M. et al. Association of plasma B lymphocyte stimulator levels and disease activity in systemic lupus erythematosus. Arthritis Rheum. 58, 2453–2459 (2008).
Zhao, L. D. et al. Expressions of BAFF/BAFF receptors and their correlation with disease activity in Chinese SLE patients. Lupus 19, 1534–1549 (2010).
Lopez, P. et al. Interferon-α-induced B-lymphocyte stimulator expression and mobilization in healthy and systemic lupus erthymatosus monocytes. Rheumatology (Oxford) 53, 2249–2258 (2014).
Panchanathan, R. & Choubey, D. Murine BAFF expression is up-regulated by estrogen and interferons: implications for sex bias in the development of autoimmunity. Mol. Immunol. 53, 15–23 (2013).
Litinskiy, M. B. et al. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nat. Immunol. 3, 822–829 (2002).
Abu-Rish, E. Y., Amrani, Y. & Browning, M. J. Toll-like receptor 9 activation induces expression of membrane-bound B-cell activating factor (BAFF) on human B cells and leads to increased proliferation in response to both soluble and membrane-bound BAFF. Rheumatology (Oxford) 52, 1190–1201 (2013).
Mosak, J. & Furie, R. Breaking the ice in systemic lupus erythematosus: belimumab, a promising new therapy. Lupus 22, 361–371 (2013).
Isenberg, D. A. et al. Efficacy and safety of subcutaneous tabalumab in patients with systemic lupus erythematosus: results from ILLUMINATE-1, a 52-week, phase III, multicentre, randomised, double-blind, placebo-controlled study. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2015-207653 (2015).
Merrill, J. T. et al. Efficacy and safety of subcutaneous tabalumab, a monoclonal antibody to B-cell activating factor, in patients with systemic lupus erythematosus: results from ILLUMINATE-2, a 52-week, phase III, multicentre, randomised, double-blind, placebo-controlled study. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2015-207654 (2015).
Jacob, C. O. et al. Development of systemic lupus erythematosus in NZM 2328 mice in the absence of any single BAFF receptor. Arthritis Rheum. 65, 1043–1054 (2013).
Zheng, N., Wang, D., Ming, H., Zhang, H. & Yu, X. BAFF promotes proliferation of human mesangial cells through interaction with BAFF-R. BMC Nephrol. 16, 72 (2015).
Figgett, W. A. et al. Deleting the BAFF receptor TACI protects against systemic lupus erythematosus without extensive reduction of B cell numbers. J. Autoimmun. 61, 9–16 (2015).
O'Connor, B. P. et al. BCMA is essential for the survival of long-lived bone marrow plasma cells. J. Exp. Med. 199, 91–98 (2004).
Vincent, F. B., Morand, E. F., Schneider, P. & Mackay, F. The BAFF/APRIL system in SLE pathogenesis. Nat. Rev. Rheumatol. 10, 365–373 (2014).
Coquery, C. M. & Erickson, L. D. Regulatory roles of the tumor necrosis factor receptor BCMA. Crit. Rev. Immunol. 32, 287–305 (2012).
Jiang, C., Loo, W. M., Greenley, E. J., Tung, K. S. & Erickson, L. D. B cell maturation antigen deficiency exacerbates lymphoproliferation and autoimmunity in murine lupus. J. Immunol. 186, 6136–6147 (2011).
Jacob, C. O. et al. Differential development of systemic lupus erythematosus in NZM 2328 mice deficient in discrete pairs of BAFF receptors. Arthritis Rheumatol. 67, 2523–2535 (2015).
Huard, B., Schneider, P., Mauri, D., Tschopp, J. & French, L. E. T cell costimulation by the TNF ligand BAFF. J. Immunol. 167, 6225–6231 (2001).
Ng, L. G. et al. B cell-activating factor belonging to the TNF family (BAFF)-R is the principal BAFF receptor facilitating BAFF costimulation of circulating T and B cells. J. Immunol. 173, 807–817 (2004).
Coquery, C. M. et al. BAFF regulates follicular helper T cells and affects their accumulation and interferon-γ production in autoimmunity. Arthritis Rheumatol. 67, 773–784 (2015).
Goenka, R. et al. Local BLyS production by T follicular cells mediates retention of high affinity B cells during affinity maturation. J. Exp. Med. 211, 45–56 (2014).
Goenka, R., Scholz, J. L., Sindhava, V. J. & Cancro, M. P. New roles for the BLyS/BAFF family in antigen-experienced B cell niches. Cytokine Growth Factor Rev. 25, 107–113 (2014).
Scholz, J. L. & Cancro, M. P. Resolve, revise, and relax: the 3 Rs of B cell repertoire adjustment. Immunol. Lett. 143, 2–8 (2012).
Turner-Stokes, T. et al. The efficacy of repeated treatment with B-cell depletion therapy in systemic lupus erythematosus: an evaluation. Rheumatology (Oxford) 50, 1401–1408 (2011).
Cambridge, G. et al. The effect of B-cell depletion therapy on serological evidence of B-cell and plasmablast activation in patients with rheumatoid arthritis over multiple cycles of rituximab treatment. J. Autoimmun. 50, 67–76 (2014).
Smilek, D. E., Ehlers, M. R. & Nepom, G. T. Restoring the balance: immunotherapeutic combinations for autoimmune disease. Dis. Model. Mech. 7, 503–513 (2014).
Gong, Q. et al. Importance of cellular microenvironment and circulatory dynamics in B cell immunotherapy. J. Immunol. 174, 817–826 (2005).
Lin, W. et al. Dual B cell immunotherapy is superior to individual anti-CD20 depletion or BAFF blockade in murine models of spontaneous or accelerated lupus. Arthritis Rheumatol. 67, 215–224 (2015).
Wild, J. et al. Neutralization of (NK-cell-derived) B-cell activating factor by belimumab restores sensitivity of chronic lymphoid leukemia cells to direct and rituximab-induced NK lysis. Leukemia 29, 1676–1683 (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Acknowledgements
The authors' research is funded by grants from Arthritis Research UK and University College London Hospitals Biomedical Research Centre.
Author information
Authors and Affiliations
Contributions
Both authors contributed to all aspects of the article: researching data for the article, writing the manuscript, discussions of its content and review or editing of manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.R.E. is Chief Investigator for BEAT-LUPUS, a trial combining rituximab and belimumab for patients with SLE. This trial is jointly funded by GlaxoSmithKline, Arthritis Research UK and University College London Hospitals Biomedical Research Centre.
Rights and permissions
About this article
Cite this article
Ehrenstein, M., Wing, C. The BAFFling effects of rituximab in lupus: danger ahead?. Nat Rev Rheumatol 12, 367–372 (2016). https://doi.org/10.1038/nrrheum.2016.18
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2016.18
This article is cited by
-
Treatment of lupus nephritis: consensus, evidence and perspectives
Nature Reviews Rheumatology (2023)
-
Clinical advances in immunotherapy for immune-mediated glomerular diseases
Clinical and Experimental Medicine (2023)
-
The role of BAFF and G-CSF for rituximab-induced late-onset neutropenia (LON) in lymphomas
Medical Oncology (2021)
-
Elevated T cell levels in peripheral blood predict poor clinical response following rituximab treatment in new-onset type 1 diabetes
Genes & Immunity (2019)
-
Phenotypically distinct anti-insulin B cells repopulate pancreatic islets after anti-CD20 treatment in NOD mice
Diabetologia (2019)