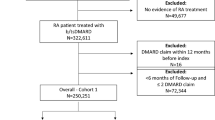
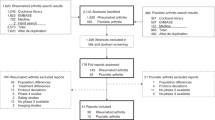
Abstract
The beginning of the 21st century saw a biopharmaceutical revolution in the treatment of inflammatory rheumatic diseases, particularly rheumatoid arthritis. The fast-evolving use of biologic therapies highlighted the need to develop registers at national and international levels with the aim of collecting long-term data on patient outcomes. Over the past 15 years, many biologics registers have contributed a wealth of data and provided robust and reliable evidence on the use, effectiveness and safety of these therapies. The unavoidable challenges posed by the continuous introduction of new therapies, particularly with regard to understanding their long-term safety, highlights the importance of learning from experience with established biologic therapies. In this Perspectives article, the role of biologics registers in bridging the evidence gap between efficacy in clinical trials and real-world effectiveness is discussed, with a focus on methodological aspects of registers, their unique features and challenges and their role going forward.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
van Vollenhoven, R. F. Treatment of rheumatoid arthritis: state of the art 2009. Nat. Rev. Rheumatol. 5, 531–541 (2009).
Choi, H. K. & Seeger, J. D. Observational research in rheumatic disorders. Rheum. Dis. Clin. North Am. 30, 685–699 (2004).
Watson, K., Symmons, D., Griffiths, I. & Silman, A. The British Society for Rheumatology Biologics Register. Ann. Rheum. Dis. 64 (Suppl. 4), iv42–iv43 (2005).
Zink, A. et al. Treatment continuation in patients receiving biological agents or conventional DMARD therapy. Ann. Rheum. Dis. 64, 1274–1279 (2005).
van Vollenhoven, R. F. & Askling, J. Rheumatoid arthritis registries in Sweden. Clin. Exp. Rheumatol. 23, S195–S200 (2005).
Hetland, M. L. et al. Direct comparison of treatment responses, remission rates, and drug adherence in patients with rheumatoid arthritis treated with adalimumab, etanercept, or infliximab: results from eight years of surveillance of clinical practice in the nationwide Danish DANBIO registry. Arthritis Rheum. 62, 22–32 (2010).
Gabay, C., Riek, M., Scherer, A. & Finckh, A. Effectiveness of biologic DMARDs in monotherapy versus in combination with synthetic DMARDs in rheumatoid arthritis: data from the Swiss Clinical Quality Management Registry. Rheumatology (Oxford) 54, 1664–1672 (2015).
Curtis, J. R. et al. A comparison of patient characteristics and outcomes in selected European and U.S. rheumatoid arthritis registries. Semin. Arthritis Rheum. 40, 2–14.e1 (2010).
Zink, A. et al. Effectiveness of tumor necrosis factor inhibitors in rheumatoid arthritis in an observational cohort study: comparison of patients according to their eligibility for major randomized clinical trials. Arthritis Rheum. 54, 3399–3407 (2006).
Kievit, W. et al. The efficacy of anti-TNF in rheumatoid arthritis, a comparison between randomised controlled trials and clinical practice. Ann. Rheum. Dis. 66, 1473–1478 (2007).
Kihara, M. et al. Use and effectiveness of tocilizumab among patients with rheumatoid arthritis: an observational study from the British Society for Rheumatology Biologics Register for rheumatoid arthritis. Clin. Rheumatol. 36, 241–250 (2017).
Hetland, M. L. et al. Do changes in prescription practice in patients with rheumatoid arthritis treated with biological agents affect treatment response and adherence to therapy? Results from the nationwide Danish DANBIO Registry. Ann. Rheum. Dis. 67, 1023–1026 (2008).
Hyrich, K. L., Watson, K. D., Lunt, M. & Symmons, D. P. M. Changes in disease characteristics and response rates among patients in the United Kingdom starting anti-tumour necrosis factor therapy for rheumatoid arthritis between 2001 and 2008. Rheumatology (Oxford) 50, 117–123 (2011).
Hyrich, K. L., Symmons, D. P. M., Watson, K. D. & Silman, A. J. Comparison of the response to infliximab or etanercept monotherapy with the response to cotherapy with methotrexate or another disease-modifying antirheumatic drug in patients with rheumatoid arthritis: results from the British Society for Rheumatology Biologics Register. Arthritis Rheum. 54, 1786–1794 (2006).
Virkki, L., Aaltonen, K. & Nordström, D. Biological therapy in rheumatoid arthritis based on ten years of registry surveillance in Finland [Finnish]. Duodecim 126, 1487–1495 (2010).
Soliman, M. M. et al. Impact of concomitant use of DMARDs on the persistence with anti-TNF therapies in patients with rheumatoid arthritis: results from the British Society for Rheumatology Biologics Register. Ann. Rheum. Dis. 70, 583–589 (2011).
Østergaard, M. et al. Low remission rates but long drug survival in rheumatoid arthritis patients treated with infliximab or etanercept: results from the nationwide Danish DANBIO database. Scand. J. Rheumatol. 36, 151–154 (2007).
Iannone, F. et al. Longterm retention of tumor necrosis factor-α inhibitor therapy in a large italian cohort of patients with rheumatoid arthritis from the GISEA registry: an appraisal of predictors. J. Rheumatol. 39, 1179–1184 (2012).
Gomez-Reino, J. J. & Carmona, L. Switching TNF antagonists in patients with chronic arthritis: an observational study of 488 patients over a four-year period. Arthritis Res. Ther. 8, R29 (2006).
van Vollenhoven, R., Harju, A., Brannemark, S. & Klareskog, L. Treatment with infliximab (Remicade) when etanercept (Enbrel) has failed or vice versa: data from the STURE registry showing that switching tumour necrosis factor alpha blockers can make sense. Ann. Rheum. Dis. 62, 1195–1198 (2003).
Hyrich, K. L., Lunt, M., Watson, K. D., Symmons, D. P. M. & Silman, A. J. Outcomes after switching from one anti-tumor necrosis factor alpha agent to a second anti-tumor necrosis factor alpha agent in patients with rheumatoid arthritis: results from a large UK national cohort study. Arthritis Rheum. 56, 13–20 (2007).
Soliman, M. M. et al. Rituximab or a second anti-tumor necrosis factor therapy for rheumatoid arthritis patients who have failed their first anti-tumor necrosis factor therapy? Comparative analysis from the British Society for Rheumatology Biologics Register. Arthritis Care Res. (Hoboken) 64, 1108–1115 (2012).
Finckh, A. et al. Which subgroup of patients with rheumatoid arthritis benefits from switching to rituximab versus alternative anti-tumour necrosis factor (TNF) agents after previous failure of an anti-TNF agent? Ann. Rheum. Dis. 69, 387–393 (2010).
Emery, P. et al. Rituximab versus an alternative TNF inhibitor in patients with rheumatoid arthritis who failed to respond to a single previous TNF inhibitor: SWITCH-RA, a global, observational, comparative effectiveness study. Ann. Rheum. Dis. 74, 979–984 (2015).
Gottenberg, J.-E. et al. Non-TNF-targeted biologic versus a second anti-TNF drug to treat rheumatoid arthritis in patients with insufficient response to a first anti-TNF drug: a randomized clinical trial. JAMA 316, 1172–1180 (2016).
Mariette, X., Gottenberg, J.-E., Ravaud, P. & Combe, B. Registries in rheumatoid arthritis and autoimmune diseases: data from the French registries. Rheumatology (Oxford) 50, 222–229 (2011).
Gabay, C. et al. Effectiveness of tocilizumab with and without synthetic disease-modifying antirheumatic drugs in rheumatoid arthritis: results from a European collaborative study. Ann. Rheum. Dis. 75, 1336–1342 (2016).
Lahaye, C. et al. Effectiveness and safety of abatacept in elderly patients with rheumatoid arthritis enrolled in the French Society of Rheumatology's ORA registry. Rheumatology (Oxford) 55, 874–882 (2016).
Gottenberg, J. E. et al. Brief report: association of rheumatoid factor and anti-citrullinated protein antibody positivity with better effectiveness of abatacept: results from the Pan-European Registry analysis. Arthritis Rheumatol. 68, 1346–1352 (2016).
Chatzidionysiou, K. et al. Highest clinical effectiveness of rituximab in autoantibody-positive patients with rheumatoid arthritis and in those for whom no more than one previous TNF antagonist has failed: pooled data from 10 European registries. Ann. Rheum. Dis. 70, 1575–1580 (2011).
Hyrich, K. L., Watson, K. D., Silman, A. J. & Symmons, D. P. M. Predictors of response to anti-TNF-alpha therapy among patients with rheumatoid arthritis: results from the British Society for Rheumatology Biologics Register. Rheumatology (Oxford) 45, 1558–1565 (2006).
Kristensen, L. E. et al. Predictors of response to anti-TNF therapy according to ACR and EULAR criteria in patients with established RA: results from the South Swedish Arthritis Treatment Group Register. Rheumatology (Oxford) 47, 495–499 (2008).
Sode, J. et al. Anti-TNF treatment response in rheumatoid arthritis patients is associated with genetic variation in the NLRP3-inflammasome. PLoS ONE 9, e100361 (2014).
Saevarsdottir, S. et al. Patients with early rheumatoid arthritis who smoke are less likely to respond to treatment with methotrexate and tumor necrosis factor inhibitors: observations from the Epidemiological Investigation of Rheumatoid Arthritis and the Swedish Rheumatology Register cohorts. Arthritis Rheum. 63, 26–36 (2011).
Söderlin, M. K., Petersson, I. F. & Geborek, P. The effect of smoking on response and drug survival in rheumatoid arthritis patients treated with their first anti-TNF drug. Scand. J. Rheumatol. 41, 1–9 (2012).
Canhão, H. et al. Comparative effectiveness and predictors of response to tumour necrosis factor inhibitor therapies in rheumatoid arthritis. Rheumatology (Oxford) 51, 2020–2026 (2012).
Neovius, M. et al. Drug survival on TNF inhibitors in patients with rheumatoid arthritis comparison of adalimumab, etanercept and infliximab. Ann. Rheum. Dis. 74, 354–360 (2015).
Plant, D. et al. Differential methylation as a biomarker of response to etanercept in patients with rheumatoid arthritis. Arthritis Rheumatol. 68, 1353–1360 (2016).
Listing, J. et al. Infections in patients with rheumatoid arthritis treated with biologic agents. Arthritis Rheum. 52, 3403–3412 (2005).
Askling, J. et al. Time-dependent increase in risk of hospitalisation with infection among Swedish RA patients treated with TNF antagonists. Ann. Rheum. Dis. 66, 1339–1344 (2007).
Salmon-Ceron, D. et al. Drug-specific risk of non-tuberculosis opportunistic infections in patients receiving anti-TNF therapy reported to the 3-year prospective French RATIO registry. Ann. Rheum. Dis. 70, 616–623 (2011).
Dixon, W. G. et al. Rates of serious infection, including site-specific and bacterial intracellular infection, in rheumatoid arthritis patients receiving anti-tumor necrosis factor therapy: results from the British Society for Rheumatology Biologics Register. Arthritis Rheum. 54, 2368–2376 (2006).
Galloway, J. B. et al. Anti-TNF therapy is associated with an increased risk of serious infections in patients with rheumatoid arthritis especially in the first 6 months of treatment: updated results from the British Society for Rheumatology Biologics Register with special emphasis on risks in the elderly. Rheumatology (Oxford) 50, 124–131 (2010).
Strangfeld, A. et al. Treatment benefit or survival of the fittest: what drives the time-dependent decrease in serious infection rates under TNF inhibition and what does this imply for the individual patient? Ann. Rheum. Dis. 70, 1914–1920 (2011).
Greenberg, J. D. et al. Tumour necrosis factor antagonist use and associated risk reduction of cardiovascular events among patients with rheumatoid arthritis. Ann. Rheum. Dis. 70, 576–582 (2011).
Mariette, X. et al. Malignancies associated with tumour necrosis factor inhibitors in registries and prospective observational studies: a systematic review and meta-analysis. Ann. Rheum. Dis. 70, 1895–1904 (2011).
Mercer, L. K. et al. Risk of invasive melanoma in patients with rheumatoid arthritis treated with biologics: results from a collaborative project of 11 European biologic registers. Ann. Rheum. Dis. 76, 386–391 (2017).
Askling, J. et al. Swedish registers to examine drug safety and clinical issues in RA. Ann. Rheum. Dis. 65, 707–712 (2006).
Mercer, L. K. et al. Risk of solid cancer in patients exposed to anti-tumour necrosis factor therapy: results from the British Society for Rheumatology Biologics Register for Rheumatoid Arthritis. Ann. Rheum. Dis. 74, 1087–1093 (2015).
Dreyer, L. et al. Incidences of overall and site specific cancers in TNFα inhibitor treated patients with rheumatoid arthritis and other arthritides — a follow-up study from the DANBIO Registry. Ann. Rheum. Dis. 72, 79–82 (2013).
Raaschou, P., Simard, J. F., Holmqvist, M. & Askling, J. Rheumatoid arthritis, anti-tumour necrosis factor therapy, and risk of malignant melanoma: nationwide population based prospective cohort study from Sweden. BMJ 346, f1939 (2013).
Strangfeld, A. et al. Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German Biologics Register RABBIT. Arthritis Res. Ther. 12, R5 (2010).
Pallavicini, F. B. et al. Tumour necrosis factor antagonist therapy and cancer development: analysis of the LORHEN registry. Autoimmun. Rev. 9, 175–180 (2010).
Dixon, W. G. et al. Influence of anti-tumor necrosis factor therapy on cancer incidence in patients with rheumatoid arthritis who have had a prior malignancy: results from the British Society for Rheumatology Biologics Register. Arthritis Care Res. (Hoboken) 62, 755–763 (2010).
Askling, J. et al. Cancer risk in patients with rheumatoid arthritis treated with anti-tumor necrosis factor alpha therapies: does the risk change with the time since start of treatment? Arthritis Rheum. 60, 3180–3189 (2009).
Askling, J. et al. Anti-tumour necrosis factor therapy in rheumatoid arthritis and risk of malignant lymphomas: relative risks and time trends in the Swedish Biologics Register. Ann. Rheum. Dis. 68, 648–653 (2009).
Askling, J. et al. Haematopoietic malignancies in rheumatoid arthritis: lymphoma risk and characteristics after exposure to tumour necrosis factor antagonists. Ann. Rheum. Dis. 64, 1414–1420 (2005).
Silva-Fernández, L. et al. The incidence of cancer in patients with rheumatoid arthritis and a prior malignancy who receive TNF inhibitors or rituximab: results from the British Society for Rheumatology Biologics Register — Rheumatoid Arthritis. Rheumatology (Oxford) 55, 2033–2039 (2016).
Verstappen, S. M. M., King, Y., Watson, K. D., Symmons, D. P. M. & Hyrich, K. L. Anti-TNF therapies and pregnancy: outcome of 130 pregnancies in the British Society for Rheumatology Biologics Register. Ann. Rheum. Dis. 70, 823–826 (2011).
Strangfeld, A. et al. Pregnancies in patients with long-standing rheumatoid arthritis and biologic DMARD treatment: course of disease during pregnancy and pregnancy outcomes [abstract]. Arthritis Rheumatol. 67 (Suppl. 10), a2521 (2015).
Lunt, M. et al. Different methods of balancing covariates leading to different effect estimates in the presence of effect modification. Am. J. Epidemiol. 169, 909–917 (2009).
Machado, M. A., Bernatsky, S., Bessette, L., Nedjar, H. & Rahme, E. Hospitalization for musculoskeletal disorders in rheumatoid arthritis patients: a population-based study. BMC Musculoskelet. Disord. 17, 298 (2016).
Dixon, W. G. et al. EULAR points to consider when establishing, analysing and reporting safety data of biologics registers in rheumatology. Ann. Rheum. Dis. 69, 1596–1602 (2010).
Kearsley-Fleet, L. et al. The EULAR Study Group for Registers and Observational Drug Studies: comparability of the patient case mix in the European biologic disease modifying anti-rheumatic drug registers. Rheumatology (Oxford) 54, 1074–1079 (2014).
Askling, J. & Dixon, W. The safety of anti-tumour necrosis factor therapy in rheumatoid arthritis. Curr. Opin. Rheumatol. 20, 138–144 (2008).
Chatzidionysiou, K. et al. Effectiveness of disease-modifying antirheumatic drug co-therapy with methotrexate and leflunomide in rituximab- treated rheumatoid arthritis patients: results of a 1-year follow-up study from the CERERRA collaboration. Ann. Rheum. Dis. 71, 374–377 (2012).
Askling, J. & Dixon, W. Influence of biological agents on cardiovascular disease in rheumatoid arthritis. Ann. Rheum. Dis. 70, 561–562 (2011).
Hyrich, K. L., Deighton, C., Watson, K. D., Symmons, D. P. M. & Lunt, M. Benefit of anti-TNF therapy in rheumatoid arthritis patients with moderate disease activity. Rheumatology (Oxford) 48, 1323–1327 (2009).
Tak, P. P. A personalized medicine approach to biologic treatment of rheumatoid arthritis: a preliminary treatment algorithm. Rheumatology (Oxford) 51, 600–609 (2012).
Hellgren, K. et al. Ankylosing spondylitis, psoriatic arthritis, and risk of malignant lymphoma: a cohort study based on nationwide prospectively recorded data from Sweden. Arthritis Rheumatol. 66, 1282–1290 (2014).
Verstappen, S. M. M. et al. Working status in patients with rheumatoid arthritis, ankylosing spondylitis and psoriatic arthritis: results from the British Society for Rheumatology Biologics Register. Rheumatology (Oxford) 49, 1570–1577 (2010).
Fonseca, J. E. et al. The Portuguese Society of Rheumatology position paper on the use of biosimilars. Acta Reum. Port. 39, 60–71 (2014).
Atzeni, F. et al. Position paper of Italian rheumatologists on the use of biosimilar drugs. Clin. Exp. Rheumatol. 33, 1–4 (2015).
Sjöwall, C. et al. Svensk Reumatologisk Förenings policydokument avseende biosimilarer [Swedish]. Svensk Reumatologisk Förening http://svenskreumatologi.se/srfs-riktlinjer/policydokument-behandling/ (2017).
James, S., Rao, S. V. & Granger, C. B. Registry-based randomized clinical trials — a new clinical trial paradigm. Nat. Rev. Cardiol. 12, 312–316 (2015).
Ibfelt, E. H., Jensen, D. V. & Hetland, M. L. The Danish nationwide clinical register for patients with rheumatoid arthritis: DANBIO. Clin. Epidemiol. 8, 737–742 (2016).
Kvien, T. K. et al. A Norwegian DMARD register: prescriptions of DMARDs and biological agents to patients with inflammatory rheumatic diseases. Clin. Exp. Rheumatol. 23, S188–S194 (2005).
Lie, E. et al. First-time prescriptions of biological disease-modifying antirheumatic durgs in rheumatoid arthritis, psoriatic arthritis and axial spondyloarthritis 2002-2011: data from the NOR-DMARD register. Ann. Rheum. Dis. 73, 1905–1906 (2014).
Uitz, E., Fransen, J., Langenegger, T. & Stucki, G. Clinical quality management in rheumatoid arthritis: putting theory into practice. Swiss Clinical Quality Management in Rheumatoid Arthritis. Rheumatology (Oxford) 39, 542–549 (2000).
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided substantial contributions to discussions of its content, wrote the article and undertook review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
K.L.H. declares that she has received honoraria from Abbvie and Pfizer. M.H.B. declares that she has received grants from Pfizer and Roche and has been on the advisory board and/or provided lectures for Abbvie, Bristol-Myers Squibb, Pfizer, Roche and UCB. E.N. declares no competing interests.
Related links
Supplementary information
Supplementary information S1 (table)
Key findings on overall and solid organ cancer risk in biologic-exposed patients with rheumatoid arthritis (RA) (PDF 210 kb)
Supplementary information S2 (table)
Key findings on haematological cancer* risk in biologic-exposed patients with rheumatoid arthritis (RA) (PDF 154 kb)
Supplementary information S3 (table)
Key findings on skin cancer risk in biologic-exposed patients with rheumatoid arthritis (RA) (PDF 85 kb)
Rights and permissions
About this article
Cite this article
Nikiphorou, E., Buch, M. & Hyrich, K. Biologics registers in RA: methodological aspects, current role and future applications. Nat Rev Rheumatol 13, 503–510 (2017). https://doi.org/10.1038/nrrheum.2017.81
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2017.81
This article is cited by
-
Do the drug doses of conventional synthetic DMARDs used for the treatment of biologic/targeted-synthetic DMARDs naive rheumatoid arthritis patients affect QuantiFERON-TB Gold Plus test results?
Rheumatology International (2023)
-
Hematologic Malignancy Risk in Inflammatory Arthritis Patients Treated with TNF Inhibitors: The Real-Life Data from the HUR-BIO Registry
Rheumatology and Therapy (2023)
-
Anxiety levels before biologic initiation and changes with treatment in patients with psoriatic arthritis: HUR-BIO biologic registry results
Clinical Rheumatology (2022)
-
APPA (apocynin and paeonol) modulates pathological aspects of human neutrophil function, without supressing antimicrobial ability, and inhibits TNFα expression and signalling
Inflammopharmacology (2020)
-
Persistence rates of abatacept and TNF inhibitors used as first or second biologic DMARDs in the treatment of rheumatoid arthritis: 9 years of experience from the Rhumadata® clinical database and registry
Arthritis Research & Therapy (2019)