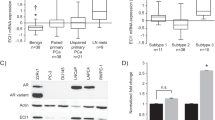
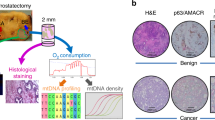
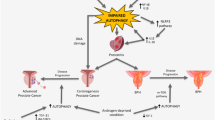
Abstract
Prostate cancer is a prevalent disease that is characterized by a presumably long latency period and a moderate propensity to metastasize. Although a range of mechanisms have been implicated in prostate carcinogenesis, the factors determining the initiation of metastasis remain obscure. The synchronized function of prostate cells depends on their metabolic and electrical coupling; disturbance of these functions has long been suggested to be integral to prostate carcinogenesis. However, although connexins form intercellular channels involved in gap-junction-mediated intercellular coupling (GJIC), whether these proteins also have GJIC-independent roles in cancer progression and metastasis remains a matter of debate. Some data indicate a correlation between connexin expression and the invasive potential of prostate cancer cells, which points to stage-specific functions of connexins during prostate cancer development. For example, restoration of connexin expression seems to be crucial for the formation of invasive cell subsets within heterogeneous prostate cancer cell populations that have undergone aberrant differentiation. Consequently, the clinical application of therapeutic and prophylactic approaches focused on the modulation of connexin expression in prostate cancer cells should be reconsidered.
Key Points
-
Connexins are integral membrane proteins that form gap junctions, which are clusters of aqueous channels that directly link cytoplasmic compartments of neighboring cells and enable intercellular communication
-
Connexins can also modulate cellular traits crucial for cancer development, such as proliferation and migration, in a manner independent of gap junctions
-
The differential expression and functional incompatibility of connexons composed of connexin 32 and connexin 43 establish functional compartments within the prostate epithelium that determine its synchronized action
-
Inhibition of connexin expression promotes prostate carcinogenesis, whereas restoration of connexin function in subpopulations of prostate cancer cells seems to drive prostate cancer progression and metastasis
-
Treatment regimens that target connexin expression and function in prostate cancer cells should take into account the stage-specific roles of connexins and gap junctions in prostate cancer development
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Joshua, A. M. et al. Prostatic preneoplasia and beyond. Biochim. Biophys. Acta 1785, 156–181 (2008).
Schröder, F. H. et al. Screening and prostate-cancer mortality in a randomized European study. N. Engl. J. Med. 360, 1320–1328 (2009).
Siegel, R., Ward, E., Brawley, O. & Jemal, A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J. Clin. 61, 212–236 (2011).
Sell, S. On the stem cell origin of cancer. Am. J. Pathol. 176, 2584–2594 (2010).
Sottoriva, A. et al. Cancer stem cell tumor model reveals invasive morphology and increased phenotypical heterogeneity. Cancer Res. 70, 46–56 (2010).
Lang, S. H., Frame, F. M. & Collins, A. T. Prostate cancer stem cells. J. Pathol. 217, 299–306 (2009).
Maitland, N. J. & Collins, A. A tumour stem cell hypothesis for the origins of prostate cancer. BJU Int. 96, 1219–1223 (2005).
Bonkhoff, H. & Remberger, K. Differentiation pathways and histogenetic aspects of normal and abnormal prostatic growth: a stem cell model. Prostate 28, 98–106 (1996).
Bonkhoff, H. Role of the basal cells in premalignant changes of the human prostate: a stem cell concept for the development of prostate cancer. Eur. Urol. 30, 201–205 (1996).
Goldstein, A. S., Huang, J., Guo, C., Garraway, I. P. & Witte, O. N. Identification of a cell of origin for human prostate cancer. Science 329, 568–571 (2010).
Nauseef, J. T. & Henry, M. D. Epithelial-to-mesenchymal transition in prostate cancer: paradigm or puzzle? Nat. Rev. Urol. 8, 428–439 (2011).
Moore, K. A. & Lemischka, I. R. Stem cells and their niches. Science 311, 1880–1885 (2006).
Voog, J. & Jones, D. L. Stem cells and the niche: a dynamic duo. Cell Stem Cell 6, 103–115 (2010).
Mol, A. J., Geldof, A. A., Meijer, G. A., van der Poel, H. G. & van Moorselaar, R. J. New experimental markers for early detection of high-risk prostate cancer: role of cell–cell adhesion and cell migration. J. Cancer Res. Clin. Oncol. 133, 687–695 (2007).
Wei, C. J., Xu, X. & Lo, C. W. Connexins and cell signaling in development and disease. Annu. Rev. Cell Dev. Biol. 20, 811–838 (2004).
Trosko, J. E. Gap junctional intercellular communication as a biological “Rosetta stone” in understanding, in a systems biological manner, stem cell behavior, mechanisms of epigenetic toxicology, chemoprevention and chemotherapy. J. Membr. Biol. 218, 93–100 (2007).
Loewenstein, W. R. Junctional intercellular communication and the control of growth. Biochim. Biophys. Acta 560, 1–65 (1979).
Chipman, J. K., Mally, A. & Edwards, G. O. Disruption of gap junctions in toxicity and carcinogenicity. Toxicol. Sci. 71, 146–153 (2003).
Luebeck, E. G., Buchmann, A., Schneider, D., Moolgavkar, S. H. & Schwarz, M. Modulation of liver tumorigenesis in connexin32-deficient mouse. Mutat. Res. 570, 33–47 (2005).
Czyż, J. The stage-specific function of gap junctions during tumourigenesis. Cell. Mol. Biol. Lett. 13, 92–102 (2008).
Cronier, L., Crespin, S., Strale, P. O., Defamie, N. & Mesnil, M. Gap junctions and cancer: new functions for an old story. Antioxid. Redox. Signal. 11, 323–338 (2009).
Dbouk, H. A., Mroue, R. M., El-Sabban, M. E. & Talhouk, R. S. Connexins: a myriad of functions extending beyond assembly of gap junction channels. Cell Commun. Signal. 7, 4 (2009).
Mroue, R. M., El-Sabban, M. E. & Talhouk, R. S. Connexins and the gap in context. Integr. Biol. (Camb.) 3, 255–266 (2011).
Hossain, M. Z. et al. Impaired expression and posttranslational processing of connexin43 and downregulation of gap junctional communication in neoplastic human prostate cells. Prostate 38, 55–59 (1999).
Habermann, H., Ray, V., Habermann, W. & Prins, G. S. Alterations in gap junction protein expression in human benign prostatic hyperplasia and prostate cancer. J. Urol. 167, 655–660 (2002).
Miekus, K., Czernik, M., Sroka, J., Czyż, J. & Madeja, Z. Contact stimulation of prostate cancer cell migration: the role of gap junctional coupling and migration stimulated by heterotypic cell-to-cell contacts in determination of the metastatic phenotype of Dunning rat prostate cancer cells. Biol. Cell 97, 893–903 (2005).
Tate, A. W. et al. Changes in gap junctional connexin isoforms during prostate cancer progression. Prostate 66, 19–31 (2006).
Tarakanov, A. O. & Goncharova, L. B. Cell–cell nanotubes: Tunneling through several types of synapses. Commun. Integr. Biol. 2, 359–361 (2009).
Maeda, S. & Tsukihara, T. Structure of the gap junction channel and its implications for its biological functions. Cell. Mol. Life Sci. 68, 1115–1129 (2011).
Evans, W. H. & Martin, P. E. Gap junctions: structure and function (review). Mol. Membr. Biol. 19, 121–136 (2002).
Burra, S. & Jiang, J. X. Regulation of cellular function by connexin hemichannels. Int. J. Biochem. Mol. Biol. 2, 119–128 (2011).
Burra, S. et al. Dendritic processes of osteocytes are mechanotransducers that induce the opening of hemichannels. Proc. Natl Acad. Sci. USA 107, 13648–13653 (2010).
Cotrina, M. L., Lin, J. H. & Nedergaard, M. Adhesive properties of connexin hemichannels. Glia 56, 1791–1798 (2008).
Goldberg, G. S., Moreno, A. P. & Lampe, P. D. Gap junctions between cells expressing connexin 43 or 32 show inverse permselectivity to adenosine and ATP. J. Biol. Chem. 277, 36725–36730 (2002).
Salameh, A. Life cycle of connexins: regulation of connexin synthesis and degradation. Adv. Cardiol. 42, 57–70 (2006).
Peracchia, C. Chemical gating of gap junction channels; roles of calcium, pH and calmodulin. Biochim. Biophys. Acta 1662, 61–80 (2004).
Laird, D. W. Life cycle of connexins in health and disease. Biochem. J. 394, 527–543 (2006).
Solan, J. L. & Lampe, P. D. Connexin43 phosphorylation: structural changes and biological effects. Biochem. J. 419, 261–272 (2009).
Xu, X. et al. Modulation of mouse neural crest cell motility by N-cadherin and connexin 43 gap junctions. J. Cell Biol. 154, 217–230 (2001).
Talhouk, R. S. et al. Heterocellular interaction enhances recruitment of α and β-catenins and ZO-2 into functional gap-junction complexes and induces gap junction-dependent differentiation of mammary epithelial cells. Exp. Cell Res. 314, 3275–3291 (2008).
Francis, R. et al. Connexin43 modulates cell polarity and directional cell migration by regulating microtubule dynamics. PLoS ONE 6, e26379 (2011).
Elias, L. A., Wang, D. D. & Kriegstein, A. R. Gap junction adhesion is necessary for radial migration in the neocortex. Nature 448, 901–907 (2007).
Cina, C. et al. Involvement of the cytoplasmic C-terminal domain of connexin43 in neuronal migration. J. Neurosci. 29, 2009–2021 (2009).
Zhang, Y. W., Nakayama, K., Nakayama, K. & Morita, I. A novel route for connexin 43 to inhibit cell proliferation: negative regulation of S-phase kinase-associated protein (Skp 2). Cancer Res. 63, 1623–1630 (2003).
Kardami, E. et al. The role of connexins in controlling cell growth and gene expression. Prog. Biophys. Mol. Biol. 94, 245–264 (2007).
Dang, X., Doble, B. W. & Kardami, E. The carboxy-tail of connexin-43 localizes to the nucleus and inhibits cell growth. Mol. Cell Biochem. 242, 35–38 (2003).
Jerónimo, C. et al. Epigenetics in prostate cancer: biologic and clinical relevance. Eur. Urol. 60, 753–766 (2011).
Naus, C. C. & Laird, D. W. Implications and challenges of connexin connections to cancer. Nat. Rev. Cancer 10, 435–441 (2010).
Leithe, E., Sirnes, S., Omori, Y. & Rivedal, E. Downregulation of gap junctions in cancer cells. Crit. Rev. Oncog. 12, 225–256 (2006).
Zhu, D., Caveney, S., Kidder, G. M. & Naus, C. C. Transfection of C6 glioma cells with connexin 43 cDNA: analysis of expression, intercellular coupling, and cell proliferation. Proc. Natl Acad. Sci. USA 88, 1883–1887 (1991).
Chen, S. C., Pelletier, D. B., Ao, P. & Boynton, A. L. Connexin43 reverses the phenotype of transformed cells and alters their expression of cyclin/cyclin-dependent kinases. Cell Growth Differ. 6, 681–690 (1995).
Goldberg, G. S. et al. Connexin43 suppresses MFG-E8 while inducing contact growth inhibition of glioma cells. Cancer Res. 60, 6018–6026 (2000).
Fujimoto, E. et al. Negative growth control of renal cell carcinoma cell by connexin 32: possible involvement of Her-2. Mol. Carcinog. 40, 135–142 (2004).
Mehta, P. P. et al. Suppression of human prostate cancer cell growth by forced expression of connexin genes. Dev. Genet. 24, 91–110 (1999).
Hirschi, K. K., Xu, C. E., Tsukamoto, T. & Sager, R. Gap junction genes Cx26 and Cx43 individually suppress the cancer phenotype of human mammary carcinoma cells and restore differentiation potential. Cell Growth Differ. 7, 861–870 (1996).
McLachlan, E., Shao, Q. & Laird, D. W. Connexins and gap junctions in mammary gland development and breast cancer progression. J. Membr. Biol. 218, 107–121 (2007).
Trosko, J. E., Chang, C. C., Upham, B. L. & Tai, M. H. Ignored hallmarks of carcinogenesis: stem cells and cell-cell communication. Ann. NY Acad. Sci. 1028, 192–201 (2004).
Graeber, S. H. & Hülser, D. F. Connexin transfection induces invasive properties in HeLa cells. Exp. Cell Res. 243, 142–149 (1998).
Ito, A. et al. A role for heterologous gap junctions between melanoma and endothelial cells in metastasis. J. Clin. Invest. 105, 1189–1197 (2000).
Lin, J. H. et al. Connexin 43 enhances the adhesivity and mediates the invasion of malignant glioma cells. J. Neurosci. 22, 4302–4311 (2002).
Bates, D. C., Sin, W. C., Aftab, Q. & Naus, C. C. Connexin43 enhances glioma invasion by a mechanism involving the carboxy terminus. Glia 55, 1554–1564 (2007).
Ito, A. et al. Increased expression of connexin 26 in the invasive component of lung squamous cell carcinoma: significant correlation with poor prognosis. Cancer Lett. 234, 239–248 (2006).
Kanczuga-Koda, L. et al. Increased expression of connexins 26 and 43 in lymph node metastases of breast cancer. J. Clin. Pathol. 59, 429–433 (2006).
Baklaushev, V. P. et al. Visualization of connexin 43-positive cells of glioma and the periglioma zone by means of intravenously injected monoclonal antibodies. Drug Deliv. 18, 331–337 (2011).
Han, Y. et al. Connexin43 expression increases in the epithelium and stroma along the colonic neoplastic progression pathway: implications for its oncogenic role. Gastroenterol. Res. Pract. 2011, 561719 (2011).
Tang, B., Peng, Z. H., Yu, P. W., Yu, G. & Qian, F. Expression and significance of Cx43 and E-cadherin in gastric cancer and metastatic lymph nodes. Med. Oncol. 28, 502–508 (2011).
Naoi, Y. et al. Connexin26 expression is associated with lymphatic vessel invasion and poor prognosis in human breast cancer. Breast Cancer Res. Treat. 106, 11–17 (2007).
Lewalle, J. M., Cataldo, D., Bajou, K., Lambert, C. A. & Foidart, J. M. Endothelial cell intracellular Ca2+ concentration is increased upon breast tumor cell contact and mediates tumor cell transendothelial migration. Clin. Exp. Metastasis 16, 21–29 (1998).
Elzarrad, M. K. et al. Connexin-43 upregulation in micrometastases and tumor vasculature and its role in tumor cell attachment to pulmonary endothelium. BMC Med. 6, 20 (2008).
Pollmann, M.-A., Shao, Q., Laird, D. W. & Sandig, M. Connexin 43 mediated gap junctional communication enhances breast tumor cell diapedesis in culture. Breast Cancer Res. 7, R522–R534 (2005).
Langley, R. R. & Fidler, I. J. Tumor cell-organ microenvironment interactions in the pathogenesis of cancer metastasis. Endocr. Rev. 28, 297–321 (2007).
Yano, T. et al. Connexin 32 as an anti-invasive and anti-metastatic gene in renal cell carcinoma. Biol. Pharm. Bull. 29, 1991–1994 (2006).
Sato, H. et al. The inhibitory effect of connexin 32 gene on metastasis in renal cell carcinoma. Mol. Carcinog. 47, 403–409 (2008).
Li, Z., Zhou, Z., Welch, D. R. & Donahue, H. J. Expressing connexin 43 in breast cancer cells reduces their metastasis to lungs. Clin. Exp. Metastasis 25, 893–901 (2008).
Xu, H. T. et al. Connexin 43 recruits E-cadherin expression and inhibits the malignant behaviour of lung cancer cells. Folia Histochem. Cytobiol. 46, 315–321 (2008).
Liang, Q. L., Wang, B. R., Chen, G. Q., Li, G. H. & Xu, Y. Y. Clinical significance of vascular endothelial growth factor and connexin43 for predicting pancreatic cancer clinicopathologic parameters. Med. Oncol. 27, 1164–1170 (2010).
Van der Aa, F., Roskams, T., Blyweert, W. & De Ridder, D. Interstitial cells in the human prostate: a new therapeutic target? Prostate 56, 250–255 (2003).
Dey, A., Kusljic, S., Lang, R. J. & Exintaris, B. Role of connexin 43 in the maintenance of spontaneous activity in the guinea pig prostate gland. Br. J. Pharmacol. 161, 1692–1707 (2010).
Bonkhoff, H. & Remberger, K. Morphogenetic aspects of normal and abnormal prostatic growth. Pathol. Res. Pract. 191, 833–835 (1995).
El-Alfy, M., Pelletier, G., Hermo, L. S. & Labrie, F. Unique features of the basal cells of human prostate epithelium. Microsc. Res. Tech. 51, 436–446 (2000).
Kyprianou, N. & Isaacs, J. T. Identification of a cellular receptor for transforming growth factor-β in rat ventral prostate and its negative regulation by androgens. Endocrinology 123, 2124–2131 (1988).
Sar, M., Lubahn, D. B., French, F. S. & Wilson, E. M. Immunohistochemical localization of the androgen receptor in rat and human tissues. Endocrinology 127, 3180–3186 (1990).
Pointis, G., Fiorini, C., Defamie, N. & Segretain, D. Gap junctional communication in the male reproductive system. Biochim. Biophys. Acta 1719, 102–116 (2005).
Meda, P. et al. Differential expression of gap junction connexins in endocrine and exocrine glands. Endocrinology 133, 2371–2378 (1993).
Habermann, H., Chang, W. Y., Birch, L., Mehta, P. & Prins, G. S. Developmental exposure to estrogens alters epithelial cell adhesion and gap junction proteins in the adult rat prostate. Endocrinology 142, 359–369 (2001).
Gemel, J., Valiunas, V., Brink, P. R. & Beyer, E. C. Connexin43 and connexin26 form gap junctions, but not heteromeric channels in co-expressing cells. J. Cell Sci. 117, 2469–2480 (2004).
Huynh, H. T. et al. Regulation of the gap junction connexin 43 gene by androgens in the prostate. J. Mol. Endocrinol. 26, 1–10 (2001).
Prins, G. S. et al. Influence of neonatal estrogens on rat prostate development. Reprod. Fertil. Dev. 13, 241–252 (2001).
Mitra, S. et al. Androgen-regulated formation and degradation of gap junctions in androgen-responsive human prostate cancer cells. Mol. Biol. Cell 17, 5400–5416 (2006).
Sottoriva, A., Sloot, P. M., Medema, J. P. & Vermeulen, L. Exploring cancer stem cell niche directed tumor growth. Cell Cycle 9, 1472–1479 (2010).
Cocciadiferro, L. et al. Profiling cancer stem cells in androgen-responsive and refractory human prostate tumor cell lines. Ann. NY Acad. Sci. 1155, 257–262 (2009).
Tsai, H. et al. Reduced connexin 43 expression in high grade, human prostatic adenocarcinoma cells. Biochem. Biophys. Res. Commun. 227, 64–69 (1996).
Benko, G. et al. Prognostic value of connexin43 expression in patients with clinically localized prostate cancer. Prostate Cancer Prostatic Dis. 14, 90–95 (2011).
Shackleton, M., Quintana, E., Fearon, E. R. & Morrison, S. J. Heterogeneity in cancer: cancer stem cells versus clonal evolution. Cell 138, 822–829 (2009).
Hernandez, M. et al. A histone deacetylation-dependent mechanism for transcriptional repression of the gap junction gene cx43 in prostate cancer cells. Prostate 66, 1151–1161 (2006).
Chen, W. et al. ATRA enhances bystander effect of suicide gene therapy in the treatment of prostate cancer. Urol. Oncol. 26, 397–405 (2008).
Carruba, G. et al. Intercellular communication and human prostate carcinogenesis. Ann. NY Acad. Sci. 963, 156–168 (2002).
Carruba, G. et al. Regulation of cell-to-cell communication in non-tumorigenic and malignant human prostate epithelial cells. Prostate 50, 73–82 (2002).
Tanaka, M. & Grossman, H. B. Connexin 26 induces growth suppression, apoptosis and increased efficacy of doxorubicin in prostate cancer cells. Oncol. Rep. 11, 537–541 (2004).
Mehta, P. P. et al. Gap-junctional communication in normal and neoplastic prostate epithelial cells and its regulation by cAMP. Mol. Carcinog. 15, 18–32 (1996).
Lamiche, C. et al. The gap junction protein Cx43 is involved in the bone-targeted metastatic behaviour of human prostate cancer cells. Clin. Exp. Metastasis 29, 111–122 (2012).
Szpak, K. et al. DU-145 prostate carcinoma cells that selectively transmigrate narrow obstacles express elevated levels of Cx43. Cell. Mol. Biol. Lett. 16, 625–637 (2011).
Saladino, F. et al. Connexin expression in nonneoplastic human prostate epithelial cells. Ann. NY Acad. Sci. 963, 213–217 (2002).
Carter, H. B., Partin, A. W. & Coffey, D. S. Prediction of metastatic potential in an animal model of prostate cancer: flow cytometric quantification of cell surface charge. J. Urol. 142, 1338–1341 (1989).
Miękus, K. & Madeja, Z. Genistein inhibits the contact-stimulated migration of prostate cancer cells. Cell. Mol. Biol. Lett. 12, 348–361 (2007).
Daniel-Wójcik, A. et al. Cell motility affects the intensity of gap junctional coupling in prostate carcinoma and melanoma cell populations. Int. J. Oncol. 33, 309–315 (2008).
Olk, S., Zoidl, G. & Dermietzel, R. Connexins, cell motility, and the cytoskeleton. Cell. Motil. Cytoskeleton 66, 1000–1016 (2009).
Kandouz, M. & Batist, G. Gap junctions and connexins as therapeutic targets in cancer. Expert Opin. Ther. Targets 14, 681–692 (2010).
Clere, N., Faure, S., Martinez, M. C. & Andriantsitohaina, R. Anticancer properties of flavonoids: roles in various stages of carcinogenesis. Cardiovasc. Hematol. Agents Med. Chem. 9, 62–77 (2011).
Nishiumi, S. et al. Dietary flavonoids as cancer-preventive and therapeutic biofactors. Front. Biosci. (Schol. Ed.) 3, 1332–1362 (2011).
Chaumontet, C. et al. Flavonoids (apigenin, tangeretin) counteract tumor promoter-induced inhibition of intercellular communication of rat liver epithelial cells. Cancer Lett. 114, 207–210 (1997).
Czyż, J., Irmer, U., Zappe, C., Mauz, M. & Hülser, D. F. Hierarchy of carcinoma cell responses to apigenin: gap junctional coupling versus proliferation. Oncol. Rep. 11, 739–744 (2004).
Czyż, J., Madeja, Z., Irmer, U., Korohoda, W. & Hülser, D. F. Flavonoid apigenin inhibits motility and invasiveness of carcinoma cells in vitro. Int. J. Cancer 114, 12–18 (2005).
King, T. J. & Bertram, J. S. Connexins as targets for cancer chemoprevention and chemotherapy. Biochim. Biophys. Acta 1719, 146–160 (2005).
Conklin, C. M. et al. Genistein and quercetin increase connexin43 and suppress growth of breast cancer cells. Carcinogenesis 28, 93–100 (2007).
Czernik, M., Sroka, J., Madeja, Z. & Czyż, J. Apigenin inhibits growth and motility but increases gap junctional coupling intensity in rat prostate carcinoma (MAT-LyLu) cell populations. Cell. Mol. Biol. Lett. 13, 327–338 (2008).
Wertz, K., Siler, U. & Goralczyk, R. Lycopene: modes of action to promote prostate health. Arch. Biochem. Biophys. 430, 127–134 (2004).
Forbes, K., Gillette, K. & Sehgal, I. Lycopene increases urokinase receptor and fails to inhibit growth or connexin expression in a metastatically passaged prostate cancer cell line: a brief communication. Exp. Biol. Med. (Maywood) 228, 967–971 (2003).
Gitenay, D. et al. Serum from rats fed red or yellow tomatoes induces Connexin43 expression independently from lycopene in a prostate cancer cell line. Biochem. Biophys. Res. Commun. 364, 578–582 (2007).
Sell, S. Stem cell origin of cancer and differentiation therapy. Crit. Rev. Oncol. Hematol. 51, 1–28 (2004).
Hattori, Y. & Maitani, Y. Folate-linked nanoparticle-mediated suicide gene therapy in human prostate cancer and nasopharyngeal cancer with herpes simplex virus thymidine kinase. Cancer Gene Ther. 12, 796–809 (2005).
Wang, M., Berthoud, V. M. & Beyer, E. C. Connexin43 increases the sensitivity of prostate cancer cells to TNFα-induced apoptosis. J. Cell Sci. 120, 320–329 (2007).
Govindarajan, R. et al. Impaired trafficking of connexins in androgen-independent human prostate cancer cell lines and its mitigation by α-catenin. J. Biol. Chem. 277, 50087–50097 (2002).
Fukushima, M., Hattori, Y., Yoshizawa, T. & Maitani, Y. Combination of non-viral connexin 43 gene therapy and docetaxel inhibits the growth of human prostate cancer in mice. Int. J. Oncol. 30, 225–231 (2007).
Wybieralska, E. et al. Fenofibrate attenuates contact-stimulated cell motility and gap junctional coupling in DU-145 human prostate cancer cell populations. Oncol. Rep. 26, 447–453 (2011).
Chao, Y., Wu, Q., Acquafondata, M., Dhir, R. & Wells, A. Partial mesenchymal to epithelial reverting transition in breast and prostate cancer metastases. Cancer Microenviron. http://dx.doi.org/10.1007/s12307-011-0085-4.
Kedrin, D., Wyckoff, J., Sahai, E., Condeelis, J. & Segall, J. E. Imaging tumor cell movement in vivo. Curr. Protoc. Cell Biol. 35, 19.7.1–19.7.17 (2007).
Acknowledgements
The current authors' research on the topics discussed in this review is supported by the Polish National Science Centre (grant 2011/01/B/NZ3/00004). The authors' research is also financed by Polish Ministry of Scientific Research and Higher Education, (N N302 061936 and N N301 050236) and the European Regional Development Fund within the Operational Programme Innovative Economy (grant UDA-POIG.01.03.01-14-036/09-00). The Faculty of Biochemistry, Biophysics and Biotechnology of Jagiellonian University is a beneficiary of structural funds from the European Union (grant POIG.02.01.00-12-064/08 and POIG 01.02-00-109/99).
Author information
Authors and Affiliations
Contributions
J. Czyż, K. Szpak and Z. Madeja researched data for this article. J. Czyż wrote the manuscript, K. Szpak designed the figures and all authors reviewed and edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Czyż, J., Szpak, K. & Madeja, Z. The role of connexins in prostate cancer promotion and progression. Nat Rev Urol 9, 274–282 (2012). https://doi.org/10.1038/nrurol.2012.14
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2012.14
This article is cited by
-
SNHG3 could promote prostate cancer progression through reducing methionine dependence of PCa cells
Cellular & Molecular Biology Letters (2022)
-
Gap junction-mediated cell-to-cell communication in oral development and oral diseases: a concise review of research progress
International Journal of Oral Science (2020)
-
Connexins: substrates and regulators of autophagy
BMC Cell Biology (2016)
-
All-trans retinoic acid arrests cell cycle in leukemic bone marrow stromal cells by increasing intercellular communication through connexin 43-mediated gap junction
Journal of Hematology & Oncology (2015)
-
Connexins: junctional and non-junctional modulators of proliferation
Cell and Tissue Research (2015)