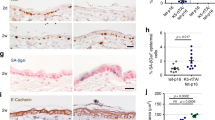
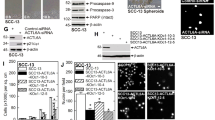
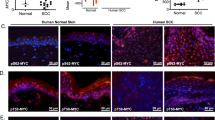
Abstract
Overexpression of the epidermal growth factor receptor (EGFR) in human papillomavirus type 16-immortalized human keratinocytes (HKc) is caused by the viral oncoprotein E6, which targets p53 for degradation. We have previously observed that expression of p53 RNAi in normal HKc is associated with an increase in EGFR mRNA and protein. We now report that p53 RNAi induces EGFR promoter activity up to approximately 10-fold in normal HKc, and this effect does not require intact p53 binding sites on the EGFR promoter. Exogenous wild-type p53 inhibits the EGFR promoter at low levels, and activates it at higher concentrations. Yin Yang 1 (YY1), which negatively regulates p53, induces EGFR promoter activity, and this effect is augmented by p53 RNAi. Intact p53 binding sites on the EGFR promoter are not required for activation by YY1. In addition, Sp1 and YY1 synergistically induce the EGFR promoter in normal HKc, indicating that Sp1 may recruit YY1 as a co-activator. Wild-type p53 suppressed Sp1- and YY1-mediated induction of the EGFR promoter. We conclude that acute loss of p53 in normal HKc induces EGFR expression by a mechanism that involves YY1 and Sp1 and does not require p53 binding to the EGFR promoter.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ai W, Narahari J, Roman A . (2000). Yin yang 1 negatively regulates the differentiation-specific E1 promoter of human papillomavirus type 6. J Virol 74: 5198–5205.
Akerman GS, Tolleson WH, Brown KL, Zyzak LL, Mourateva E, Engin TS et al. (2001). Human papillomavirus type 16 E6 and E7 cooperate to increase epidermal growth factor receptor (EGFR) mRNA levels, overcoming mechanisms by which excessive EGFR signaling shortens the life span of normal human keratinocytes. Cancer Res 61: 3837–3843.
Arteaga CL . (2002). Epidermal growth factor receptor dependence in human tumors: more than just expression? Oncologist 7 (Suppl 4): 31–39.
Baldwin A, Pirisi L, Creek KE . (2004). NFI-Ski interactions mediate transforming growth factor beta modulation of human papillomavirus type 16 early gene expression. J Virol 78: 3953–3964.
Deb SP, Munoz RM, Brown DR, Subler MA, Deb S . (1994). Wild-type human p53 activates the human epidermal growth factor receptor promoter. Oncogene 9: 1341–1349.
Dykxhoorn DM, Lieberman J . (2005). The silent revolution: RNA interference as basic biology, research tool, and therapeutic. Annu Rev Med 56: 401–423.
Gordon S, Akopyan G, Garban H, Bonavida B . (2006). Transcription factor YY1: structure, function, and therapeutic implications in cancer biology. Oncogene 25: 1125–1142.
Gronroos E, Terentiev AA, Punga T, Ericsson J . (2004). YY1 inhibits the activation of the p53 tumor suppressor in response to genotoxic stress. Proc Natl Acad Sci USA 101: 12165–12170.
Haley J, Whittle N, Bennet P, Kinchington D, Ullrich A, Waterfield M . (1987). The human EGF receptor gene: structure of the 110 kb locus and identification of sequences regulating its transcription. Oncogene Res 1: 375–396.
Hu G, Liu W, Mendelsohn J, Ellis LM, Radinsky R, Andreeff M et al. (1997). Expression of epidermal growth factor receptor and human papillomavirus E6/E7 proteins in cervical carcinoma cells. J Natl Cancer Inst 89: 1271–1276.
Innocente SA, Lee JM . (2005). p53 is a NF-Y- and p21-independent, Sp1-dependent repressor of cyclin B1 transcription. FEBS Lett 579: 1001–1007.
Ishii S, Xu YH, Stratton RH, Roe BA, Merlino GT, Pastan I . (1985). Characterization and sequence of the promoter region of the human epidermal growth factor receptor gene. Proc Natl Acad Sci USA 82: 4920–4924.
Johnson AC, Murphy BA, Matelis CM, Rubinstein Y, Piebenga EC, Akers LM et al. (2000). Activator protein-1 mediates induced but not basal epidermal growth factor receptor gene expression. Mol Med 6: 17–27.
Kageyama R, Merlino GT, Pastan I . (1988). Epidermal growth factor (EGF) receptor gene transcription. Requirement for Sp1 and an EGF receptor-specific factor. J Biol Chem 263: 6329–6336.
Kawada H, Nishiyama C, Takagi A, Tokura T, Nakano N, Maeda K et al. (2005). Transcriptional regulation of ATP2C1 gene by Sp1 and YY1 and reduced function of its promoter in Hailey-Hailey disease keratinocytes. J Invest Dermatol 124: 1206–1214.
Kilic G, Cardillo M, Ozdemirli M, Arun B . (1999). Human papillomavirus 18 oncoproteins E6 and E7 enhance irradiation- and chemotherapeutic agent-induced apoptosis in p53 and Rb mutated cervical cancer cell lines. Eur J Gynaecol Oncol 20: 167–171.
Lee JS, Galvin KM, Shi Y . (1993). Evidence for physical interaction between the zinc-finger transcription factors YY1 and Sp1. Proc Natl Acad Sci USA 90: 6145–6149.
Ludes-Meyers JH, Subler MA, Shivakumar CV, Munoz RM, Jiang P, Bigger JE et al. (1996). Transcriptional activation of the human epidermal growth factor receptor promoter by human p53. Mol Cell Biol 16: 6009–6019.
Mathur SP, Mathur RS, Rust PF, Young RC . (2001). Human papilloma virus (HPV)-E6/E7 and epidermal growth factor receptor (EGF-R) protein levels in cervical cancer and cervical intraepithelial neoplasia (CIN). Am J Reprod Immunol 46: 280–287.
Pirisi L, Creek KE, Doniger J, DiPaolo JA . (1988). Continuous cell lines with altered growth and differentiation properties originate after transfection of human keratinocytes with human papillomavirus type 16 DNA. Carcinogenesis 9: 1573–1579.
Pirisi L, Yasumoto S, Feller M, Doniger J, DiPaolo JA . (1987). Transformation of human fibroblasts and keratinocytes with human papillomavirus type 16 DNA. J Virol 61: 1061–1066.
Sheikh MS, Carrier F, Johnson AC, Ogdon SE, Fornace Jr AJ . (1997). Identification of an additional p53-responsive site in the human epidermal growth factor receptor gene promoter. Oncogene 15: 1095–1101.
Shi Y, Lee JS, Galvin KM . (1997). Everything you have ever wanted to know about Yin Yang 1. Biochim Biophys Acta 1332: F49–F66.
Sui G, Affar el B, Shi Y, Brignone C, Wall NR, Yin P et al. (2004). Yin Yang 1 is a negative regulator of p53. Cell 117: 859–872.
Wang X, Bolotin D, Chu DH, Polak L, Williams T, Fuchs E . (2006). AP-2alpha: a regulator of EGF receptor signaling and proliferation in skin epidermis. J Cell Biol 172: 409–421.
Xu J, Thompson KL, Shephard LB, Hudson LG, Gill GN . (1993). T3 receptor suppression of Sp1-dependent transcription from the epidermal growth factor receptor promoter via overlapping DNA-binding sites. J Biol Chem 268: 16065–16073.
Yakovleva T, Kolesnikova L, Vukojevic V, Gileva I, Tan-No K, Austen M et al. (2004). YY1 binding to a subset of p53 DNA-target sites regulates p53-dependent transcription. Biochem Biophys Res Commun 318: 615–624.
Yao YL, Yang WM, Seto E . (2001). Regulation of transcription factor YY1 by acetylation and deacetylation. Mol Cell Biol 21: 5979–5991.
Yarden Y, Sliwkowski MX . (2001). Untangling the ErbB signalling network. Nat Rev Mol Cell Biol 2: 127–137.
Yu J, Zhang L, Hwang PM, Rago C, Kinzler KW, Vogelstein B . (1999). Identification and classification of p53-regulated genes. Proc Natl Acad Sci USA 96: 14517–14522.
Zhao R, Gish K, Murphy M, Yin Y, Notterman D, Hoffman WH et al. (2000). Analysis of p53-regulated gene expression patterns using oligonucleotide arrays. Genes Dev 14: 981–993.
Zyzak LL, MacDonald LM, Batova A, Forand R, Creek KE, Pirisi L . (1994). Increased levels and constitutive tyrosine phosphorylation of the epidermal growth factor receptor contribute to autonomous growth of human papillomavirus type 16 immortalized human keratinocytes. Cell Growth Differ 5: 537–547.
Acknowledgements
We thank Dr Omar Bagasra for discussion and advice in the construction and use of siRNA. This work was supported by grants from the National Institutes of Health: R01CA89502 and P20CA096427 to KEC, and EXPORT Center P20MD001770.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bheda, A., Creek, K. & Pirisi, L. Loss of p53 induces epidermal growth factor receptor promoter activity in normal human keratinocytes. Oncogene 27, 4315–4323 (2008). https://doi.org/10.1038/onc.2008.65
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.65
Keywords
This article is cited by
-
Human papillomavirus-mediated carcinogenesis and tumor progression
Genome Instability & Disease (2021)
-
The Role of p53 Expression in Patients with RAS/BRAF Wild-Type Metastatic Colorectal Cancer Receiving Irinotecan and Cetuximab as Later Line Treatment
Targeted Oncology (2021)
-
VHL-HIF-2α axis-induced SMYD3 upregulation drives renal cell carcinoma progression via direct trans-activation of EGFR
Oncogene (2020)
-
Inhibition of DYRK1A-EGFR axis by p53-MDM2 cascade mediates the induction of cellular senescence
Cell Death & Disease (2019)
-
Synergistic antitumour activity of HDAC inhibitor SAHA and EGFR inhibitor gefitinib in head and neck cancer: a key role for ΔNp63α
British Journal of Cancer (2019)