Abstract
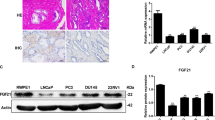
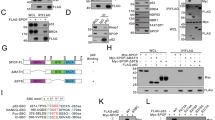
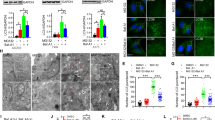
The ubiquitin-proteasome and lysosome-autophagy pathways are the two major intracellular protein degradation systems that work cooperatively to maintain homeostasis. Proteasome inhibitors (PIs) have clinical activity in hematological tumors, and inhibitors of autophagy are also being evaluated as potential antitumor therapies. In this study, we found that chemical PIs and small interfering RNA-mediated knockdown of the proteasome's enzymatic subunits promoted autophagosome formation, stimulated autophagic flux, and upregulated expression of the autophagy-specific genes (ATGs) (ATG5 and ATG7) in some human prostate cancer cells and immortalized mouse embryonic fibroblasts (MEFs). Upregulation of ATG5 and ATG7 only occurred in cells displaying PI-induced phosphorylation of the eukaryotic translation initiation factor 2 alpha (eIF2α), an important component of the unfolded protein responses. Furthermore, PIs did not induce autophagy or upregulate ATG5 in MEFs expressing a phosphorylation-deficient mutant form of eIF2α. Combined inhibition of autophagy and the proteasome induced an accumulation of intracellular protein aggregates reminiscent of neuronal inclusion bodies and caused more cancer cell death than blocking either degradation pathway alone. Overall, our data show that proteasome inhibition activates autophagy through a phospho-eIF2α-dependent mechanism to eliminate protein aggregates and alleviate proteotoxic stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Adams J . (2004). The development of proteasome inhibitors as anticancer drugs. Cancer Cell 5: 417–421.
Amaravadi RK, Yu D, Lum JJ, Bui T, Christophorou MA, Evan GI et al. (2007). Autophagy inhibition enhances therapy-induced apoptosis in a Myc-induced model of lymphoma. J Clin Invest 117: 326–336.
Anderson KC . (2004). Bortezomib therapy for myeloma. Curr Hematol Rep 3: 65.
Apel A, Herr I, Schwarz H, Rodemann HP, Mayer A . (2008). Blocked autophagy sensitizes resistant carcinoma cells to radiation therapy. Cancer Res 68: 1485–1494.
Carew JS, Nawrocki ST, Kahue CN, Zhang H, Yang C, Chung L et al. (2007). Targeting autophagy augments the anticancer activity of the histone deacetylase inhibitor SAHA to overcome Bcr-Abl-mediated drug resistance. Blood 110: 313–322.
Chauhan D, Catley L, Li G, Podar K, Hideshima T, Velankar M et al. (2005). A novel orally active proteasome inhibitor induces apoptosis in multiple myeloma cells with mechanisms distinct from Bortezomib. Cancer Cell 8: 407–419.
Chauhan D, Singh A, Brahmandam M, Podar K, Hideshima T, Richardson P et al. (2008). Combination of proteasome inhibitors bortezomib and NPI-0052 trigger in vivo synergistic cytotoxicity in multiple myeloma. Blood 111: 1654–1664.
Ding WX, Ni HM, Gao W, Yoshimori T, Stolz DB, Ron D et al. (2007). Linking of autophagy to ubiquitin-proteasome system is important for the regulation of endoplasmic reticulum stress and cell viability. Am J Pathol 171: 513–524.
Fels DR, Ye J, Segan AT, Kridel SJ, Spiotto M, Olson M et al. (2008). Preferential cytotoxicity of bortezomib toward hypoxic tumor cells via overactivation of endoplasmic reticulum stress pathways. Cancer Res 68: 9323–9330.
Geng J, Baba M, Nair U, Klionsky DJ . (2008). Quantitative analysis of autophagy-related protein stoichiometry by fluorescence microscopy. J Cell Biol 182: 129–140.
Goldberg AL . (2007). Functions of the proteasome: from protein degradation and immune surveillance to cancer therapy. Biochem Soc Trans 35: 12–17.
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R et al. (2006). Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441: 885–889.
Ito H, Daido S, Kanzawa T, Kondo S, Kondo Y . (2005). Radiation-induced autophagy is associated with LC3 and its inhibition sensitizes malignant glioma cells. Int J Oncol 26: 1401–1410.
Iwata A, Riley BE, Johnston JA, Kopito RR . (2005). HDAC6 and microtubules are required for autophagic degradation of aggregated huntingtin. J Biol Chem 280: 40282–40292.
Jiang HY, Wek RC . (2005). Phosphorylation of the alpha-subunit of the eukaryotic initiation factor-2 (eIF2alpha) reduces protein synthesis and enhances apoptosis in response to proteasome inhibition. J Biol Chem 280: 14189–14202.
Kirkin V, McEwan DG, Novak I, Dikic I . (2009). A role for ubiquitin in selective autophagy. Mol Cell 34: 259–269.
Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I et al. (2006). Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441: 880–884.
Komatsu M, Waguri S, Ueno T, Iwata J, Murata S, Tanida I et al. (2005). Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. J Cell Biol 169: 425–434.
Kondo Y, Kanzawa T, Sawaya R, Kondo S . (2005). The role of autophagy in cancer development and response to therapy. Nat Rev Cancer 5: 726–734.
Kouroku Y, Fujita E, Tanida I, Ueno T, Isoai A, Kumagai H et al. (2007). ER stress (PERK/eIF2alpha phosphorylation) mediates the polyglutamine-induced LC3 conversion, an essential step for autophagy formation. Cell Death Differ 14: 230–239.
Krysko DV, Vanden Berghe T, Parthoens E, D'Herde K, Vandenabeele P . (2008). Methods for distinguishing apoptotic from necrotic cells and measuring their clearance. Methods Enzymol 442: 307–341.
Levine B, Kroemer G . (2008). Autophagy in the pathogenesis of disease. Cell 132: 27–42.
McConkey DJ, Zhu K . (2008). Mechanisms of proteasome inhibitor action and resistance in cancer. Drug Resist Updat 11: 164–179.
Miller CP, Ban K, Dujka ME, McConkey DJ, Munsell M, Palladino M et al. (2007). NPI-0052, a novel proteasome inhibitor, induces caspase-8 and ROS-dependent apoptosis alone and in combination with HDAC inhibitors in leukemia cells. Blood 110: 267–277.
Mizushima N, Levine B, Cuervo AM, Klionsky DJ . (2008). Autophagy fights disease through cellular self-digestion. Nature 451: 1069–1075.
Mizushima N, Yoshimori T . (2007). How to interpret LC3 immunoblotting. Autophagy 3: 542–545.
Nawrocki ST, Carew JS, Dunner Jr K, Boise LH, Chiao PJ, Huang P et al. (2005). Bortezomib inhibits PKR-like endoplasmic reticulum (ER) kinase and induces apoptosis via ER stress in human pancreatic cancer cells. Cancer Res 65: 11510–11519.
Nawrocki ST, Carew JS, Pino MS, Highshaw RA, Andtbacka RH, Dunner Jr K et al. (2006). Aggresome disruption: a novel strategy to enhance bortezomib-induced apoptosis in pancreatic cancer cells. Cancer Res 66: 3773–3781.
Nelson DA, White E . (2004). Exploiting different ways to die. Genes Dev 18: 1223–1226.
Ogata M, Hino S, Saito A, Morikawa K, Kondo S, Kanemoto S et al. (2006). Autophagy is activated for cell survival after endoplasmic reticulum stress. Mol Cell Biol 26: 9220–9231.
Pandey UB, Nie Z, Batlevi Y, McCray BA, Ritson GP, Nedelsky NB et al. (2007). HDAC6 rescues neurodegeneration and provides an essential link between autophagy and the UPS. Nature 447: 859–863.
Reyes S, Rembao D, Sotelo J . (2001). The antimalarials quinacrine and chloroquine potentiate the transplacental carcinogenic effect of ethylnitrosourea on ependymal cells. Brain Tumor Pathol 18: 83–87.
Ron D, Walter P . (2007). Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol 8: 519–529.
Rubinsztein DC . (2006). The roles of intracellular protein-degradation pathways in neurodegeneration. Nature 443: 780–786.
Rubinsztein DC . (2007). Autophagy induction rescues toxicity mediated by proteasome inhibition. Neuron 54: 854–856.
Sakaki K, Wu J, Kaufman RJ . (2008). Protein kinase Ctheta is required for autophagy in response to stress in the endoplasmic reticulum. J Biol Chem 283: 15370–15380.
Savarino A, Lucia MB, Giordano F, Cauda R . (2006). Risks and benefits of chloroquine use in anticancer strategies. Lancet Oncol 7: 792–793.
Scarlatti F, Bauvy C, Ventruti A, Sala G, Cluzeaud F, Vandewalle A et al. (2004). Ceramide-mediated macroautophagy involves inhibition of protein kinase B and up-regulation of beclin 1. J Biol Chem 279: 18384–18391.
Shao Y, Gao Z, Marks PA, Jiang X . (2004). Apoptotic and autophagic cell death induced by histone deacetylase inhibitors. Proc Natl Acad Sci USA 101: 18030–18035.
Talloczy Z, Jiang W, Virgin HWt, Leib DA, Scheuner D, Kaufman RJ et al. (2002). Regulation of starvation- and virus-induced autophagy by the eIF2alpha kinase signaling pathway. Proc Natl Acad Sci USA 99: 190–195.
Wek RC, Jiang HY, Anthony TG . (2006). Coping with stress: eIF2 kinases and translational control. Biochem Soc Trans 34: 7–11.
Zhou D, Palam LR, Jiang L, Narasimhan J, Staschke KA, Wek RC . (2008). Phosphorylation of eIF2 directs ATF5 translational control in response to diverse stress conditions. J Biol Chem 283: 7064–7073.
Zhu K, Chan W, Heymach J, Wilkinson M, McConkey DJ . (2009). Control of HIF-1alpha expression by eIF2 alpha phosphorylation-mediated translational repression. Cancer Res 69: 1836–1843.
Acknowledgements
This work was supported by a grant from the Department of Defense Prostate Cancer Research Program (PC050288). The TEM studies were supported by the MD Anderson Cancer Center Support Grant (CA16672) to the High Resolution Electron Microscopy Facility. We also acknowledge Dr Woonyoung Choi and Maosheng Huang's help with the array data analyses.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Zhu, K., Dunner, K. & McConkey, D. Proteasome inhibitors activate autophagy as a cytoprotective response in human prostate cancer cells. Oncogene 29, 451–462 (2010). https://doi.org/10.1038/onc.2009.343
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.343
Keywords
This article is cited by
-
Nanomedicine for autophagy modulation in cancer therapy: a clinical perspective
Cell & Bioscience (2023)
-
Systematic approach to identify therapeutic targets and functional pathways for the cervical cancer
Journal of Genetic Engineering and Biotechnology (2023)
-
Proteasomal Stimulation by MK886 and Its Derivatives Can Rescue Tau-Induced Neurite Pathology
Molecular Neurobiology (2023)
-
Therapeutic potential of autophagy in immunity and inflammation: current and future perspectives
Pharmacological Reports (2023)
-
Targeting autophagy increases the efficacy of proteasome inhibitor treatment in multiple myeloma by induction of apoptosis and activation of JNK
BMC Cancer (2022)