Abstract
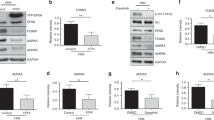
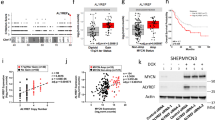
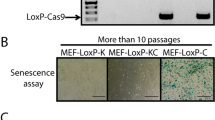
As an oncoprotein, Eps8 participates in v-Src-induced cellular transformation. To delineate the underlying mechanism, we conducted a yeast two-hybrid screening and identified IRSp53S, a protein critical in cell mobilization, as one of the Eps8-binding partners from a human brain cDNA library. The association was mediated by the multiple proline-rich regions of Eps8 and the C-terminal SH3-WWB containing domains of IRSp53S. In this study, we observed that Eps8 modulated the expression of IRSp53 in v-Src-transformed cells (IV5), raising the question of whether Eps8/IRSp53 interaction was crucial in carcinogenesis. To address this issue, we generated IV5-expressing irsp53 siRNA cells. Attenuation of IRSp53 reduced cell proliferation of IV5 in culture dish and tumor formation in mice, which could be partly rescued by ectopically expressed human IRSp53S. In addition, IRSp53 knockdown impaired activity of phosphatidylinositol 3-kinase (as reflected by Pi-Ser473 AKT) and Stat3 (as reflected by Pi-Tyr705 Stat3), and reduced cyclin D1 expression that culminated to impede G1-phase cell-cycle progression. Ectopically expressed human IRSp53S, but not its Eps8-binding defective mutants (that is, Δ363 and PPPDA), rescued these defects and partly restored cell proliferation. Remarkably, through activation of Src, EGF increased the formation of Eps8/IRSp53 complex and Stat3 activation in HeLa cells. With these results, we show for the first time that IRSp53, through its interaction with Eps8, not only affects cell migration but also dictates cellular growth in cancer cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout












Similar content being viewed by others
References
Abbott MA, Wells DG, Fallon JR . (1999). The insulin receptor tyrosine kinase substrate p58/53 and the insulin receptor are components of CNS synapses. J Neurosci 19: 7300–7308.
Aitio O, Hellman M, Kesti T, Kleino L, Samuilova O, Paakkonen K et al. (2008). Structural basis of PxxDY motif recognition in SH3 binding. J Mol Biol 382: 167–178.
Blake RA, Broome MA, Liu X, Wu J, Gishizky M, Sun L et al. (2000). SU6656, a selective Src family kinase inhibitor, used to probe growth factor signaling. Mol Cell Biol 20: 9018–9027.
Bowman T, Broome MA, Sinibaldi D, Wharton W, Pledger WJ, Sedivy JM et al. (2001). Stat3-mediated Myc expression is required for Src transformation and PDGF-induced mitogenesis. Proc Natl Acad Sci USA 98: 7319–7324.
Chen YJ, Shen MR, Chen YJ, Maa MC, Leu TH . (2008). Eps8 decreases chemosensitivity and affects survival of cervical cancer patients. Mol Cancer Ther 7: 1376–1385.
Disanza A, Carlier MF, Stradal TE, Didry D, Frittoli E, Confalonieri S et al. (2004). Eps8 controls actin-based motility by capping the barbed ends of actin filaments. Nat Cell Biol 6: 1180–1188.
Funato Y, Terabayashi T, Suenaga N, Seiki M, Takenawa T, Miki H . (2004). IRSp53/Eps8 complex is important for positive regulation of Rac and cancer cell motility/invasiveness. Cancer Res 64: 5237–5244.
Gallo R, Provenzano C, Carbone R, Di Fiore PP, Castellani L, Falcone G et al. (1997). Regulation of the tyrosine kinase substrate Eps8 expression by growth factor, v-Src and terminal differentiation. Oncogene 15: 1929–1936.
Harlow E, Lane D (eds). (1988). Antibodies: A Laboratory Manual. Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, pp 522–523.
Lee SH, Kerff F, Chereau D, Ferron F, Klug A, Dominguez R . (2007). Structural basis for actin-binding function of missing-in-metastasis. Structure 15: 145–155.
Leu TH, Yeh HH, Huang CC, Chuang YC, Su SL, Maa MC . (2004). Participation of p97Eps8 in Src-mediated transformation. J Biol Chem 279: 9875–9881.
Maa MC, Leu TH . (1998). Vanadate-dependent FAK activation is accomplished by the sustained FAK Tyr-576/577 phosphorylation. Biochem Biophy Res Commun 251: 344–349.
Maa MC, Lai JR, Lin RW, Leu TH . (1999). Enhancement of tyrosyl phosphorylation and protein expression of eps8 by v-Src. Biochim Biophys Acta 1450: 341–351.
Maa MC, Hsieh CY, Leu TH . (2001). Overexpression of p97Eps8 leads to cellular transformation: implication of pleckstrin homology domain in p97Eps8-mediated ERK activation. Oncogene 20: 106–112.
Maa MC, Lee JC, Chen YJ, Chen YJ, Lee YC, Wang ST et al. (2007). Eps8 facilitates cellular growth and motility of colon cancer cells by increasing the expression and activity of focal adhesion kinase. J Biol Chem 282: 19399–19409.
Martin GS . (2001). Timeline: the hunting of the Src. Nat Rev Mol Cell Biol 2: 467–475.
Mattila PK, Pykalainen A, Saarikangas J, Paavilainen VO, Vihinen H, Jokitalo E et al. (2007). Missing-in-metastasis and IRSp53 deform PIP2-rich membranes by an inverse BAR domain-like mechanism. J Cell Biol 7: 953–964.
Millard TH, Bompard G, Heung MY, Dafforn TR, Scott DJ, Machesky LM et al. (2005). Structural basis of filopodia formation induced by the IRSp53/MIM homology domain of human IRSp53. EMBO J 24: 240–250.
Miyahara A, Okamura-Oho Y, Miyahita T, Hoshika A, Yamada M . (2003). Genomic structure and alternative splicing of the insulin receptor tyrosine kinase substrate of 53-kDa protein. J Hum Genet 48: 410–414.
Mongiovi AM, Romaon PR, Panni S, Mendoza M, Wong WT, Musacchio A et al. (1999). A novel peptide-SH3 interaction. EMBO J 18: 5300–5309.
Oda K, Shiratsuchi T, Nishimori H, Inazawa J, Yoshikawa H, Taketani Y et al. (1999). Identification of BAIAP2 (BAI-associated protein 2), a novel human homologue of hamster IRSp53, whose SH3 interacts with the cytoplasmic domain of BAI1. Cytogenet Cell Genet 84: 75–82.
Okamura-Oho Y, Miyashita T, Ohmi K, Yamada M . (1999). Dentatorubral-pallidoluysian atrophy protein interacts through a proline-rich region near polyglutamine with the SH3 domain of an insulin receptor tyrosine kinase substrate. Hum Mol Genet 8: 947–957.
Okamura-Oho Y, Miyashita T, Yamada M . (2001). Distinctive tissue distribution and phosphorylation of IRSp53 isoforms. Biochem Biophys Res Commun 289: 957–960.
Scita G, Nordstrom J, Carbone R, Tenca P, Giardina G, Gutkind S et al. (1999). EPS8 and E3B1 transduce signals from Ras to Rac. Nature 401: 290–293.
Scita G, Confalonieri S, Lappalainen P, Suetsugu S . (2008). IRSp53: crossing the road of membrane and actin dynamics in the formation of membrane protrusions. Trends Cell Biol 18: 52–60.
Silva CM . (2004). Role of STATs as downstream signal transducers in Src family kinase-mediated tumorigensis. Oncogene 23: 8017–8023.
Suetsugu S, Murayama K, Sakamoto A, Hanawa-Suetsugu K, Seto A, Oikawa T et al. (2006). The RAC binding domain/IRSp53-MIM homology domain of IRSp53 induces RAC-dependent membrane deformation. J Biol Chem 46: 35347–35358.
Xu M, Shorts-Cary L, Knox AJ, Kleinsmidt-DeMasters B, Lillehei K, Wierman ME . (2009). Epidermal growth factor receptor pathway substrate 8 is overexpressed in human pituitary tumors: role in proliferation and survival. Endocrinology 150: 2064–2071.
Yap LF, Jenei V, Robinson CM, Moutasim K, Benn TM, Threadgold SP et al. (2009). Upregulation of Eps8 in oral squamous cell carcinoma promotes cell migration and invasion through integrin-dependent Rac1 activation. Oncogene 28: 2524–2534.
Yeh TC, Ogawa W, Danielsen AG, Roth RA . (1996). Characterization and cloning of a 58/53-kDa substrate of the insulin receptor tyrosine kinase. J Biol Chem 271: 2921–2928.
Acknowledgements
This work was supported by National Science Council grants to M-CM (NSC98-2311-B-039-002-MY3) and T-HL (NSC97-2320-B-006-024-MY3). Additional support came from NHRI (NHRI-EX-98-9828BI to T-H Leu), Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH99-TD-B-111-004 to M-C Maa) and China Medical University (CMU98-C-05 to M-C Maa). The DNA constructs of pLKO.1-mirsp53-b and pLKO.1-mirsp53-c were provided by the National RNAi Core Facility located at the Institute of Molecular Biology/Genomic Research Center, Academia Sinica, supported by the National Research Program for Genomic Medicine Grants of NSC (NSC 94-3112-B-001-003 and NSC 94-3112-B-001-018-Y).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Liu, PS., Jong, TH., Maa, MC. et al. The interplay between Eps8 and IRSp53 contributes to Src-mediated transformation. Oncogene 29, 3977–3989 (2010). https://doi.org/10.1038/onc.2010.144
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.144
Keywords
This article is cited by
-
Mechanism of IRSp53 inhibition by 14-3-3
Nature Communications (2019)
-
A synthetic cell-penetrating peptide derived from nuclear localization signal of EPS8 exerts anticancer activity against acute myeloid leukemia
Journal of Experimental & Clinical Cancer Research (2018)
-
Salt Bridge Formation between the I-BAR Domain and Lipids Increases Lipid Density and Membrane Curvature
Scientific Reports (2017)
-
Mechanism of IRSp53 inhibition and combinatorial activation by Cdc42 and downstream effectors
Nature Structural & Molecular Biology (2014)