Abstract
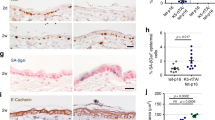
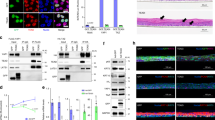
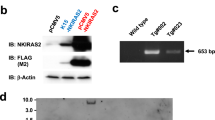
The tumor suppressor Fbxw7 (also known as Sel-10, hCdc4, hAgo, or Fbw7) is an F-box protein that functions as the substrate-recognition subunit of an SCF ubiquitin ligase complex and targets a group of oncoproteins for degradation. We now show that Fbxw7 regulates the proliferation and differentiation of keratinocytes by mediating the degradation of c-Myc and Notch proteins. Fbxw7-deficient keratinocytes showed an increased proliferative capacity that was dependent on the accumulation of c-Myc but not on that of Notch. Fbxw7 deficiency also resulted in the premature differentiation of keratinocytes in a manner dependent on both c-Myc and Notch. Although Fbxw7-deficient keratinocytes proliferated excessively in vitro, loss of Fbxw7 did not predispose keratinocytes to the formation of squamous cell carcinoma in vivo induced by the expression of oncogenic Ras, possibly because the stem cell population of keratinocytes becomes exhausted as a result of enhanced Notch activity. Indeed, suppression of Notch signaling by additional ablation of RBP-J in Fbxw7-deficient keratinocytes conferred a more aggressive tumorigenic capacity. Collectively, these results indicate that Fbxw7 controls the proliferation and differentiation of keratinocytes, and that it exerts both inhibitory and stimulatory actions in skin carcinogenesis by counteracting the proliferation-promoting effect of c-Myc and the tumor-suppressive effect of Notch, respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Nakayama KI, Nakayama K . Ubiquitin ligases: cell-cycle control and cancer. Nat Rev Cancer 2006; 6: 369–381.
Jin J, Cardozo T, Lovering RC, Elledge SJ, Pagano M, Harper JW . Systematic analysis and nomenclature of mammalian F-box proteins. Genes Dev 2004; 18: 2573–2580.
Ang XL, Wade Harper J . SCF-mediated protein degradation and cell cycle control. Oncogene 2005; 24: 2860–2870.
Welcker M, Orian A, Jin J, Grim JE, Harper JW, Eisenman RN et al. The Fbw7 tumor suppressor regulates glycogen synthase kinase 3 phosphorylation-dependent c-Myc protein degradation. Proc Natl Acad Sci USA 2004; 101: 9085–9090.
Yada M, Hatakeyama S, Kamura T, Nishiyama M, Tsunematsu R, Imaki H et al. Phosphorylation-dependent degradation of c-Myc is mediated by the F-box protein Fbw7. EMBO J 2004; 23: 2116–2125.
Koepp DM, Schaefer LK, Ye X, Keyomarsi K, Chu C, Harper JW et al. Phosphorylation-dependent ubiquitination of cyclin E by the SCFFbw7 ubiquitin ligase. Science 2001; 294: 173–177.
Moberg KH, Bell DW, Wahrer DC, Haber DA, Hariharan IK . Archipelago regulates Cyclin E levels in Drosophila and is mutated in human cancer cell lines. Nature 2001; 413: 311–316.
Strohmaier H, Spruck CH, Kaiser P, Won KA, Sangfelt O, Reed SI . Human F-box protein hCdc4 targets cyclin E for proteolysis and is mutated in a breast cancer cell line. Nature 2001; 413: 316–322.
Gupta-Rossi N, Le Bail O, Gonen H, Brou C, Logeat F, Six E et al. Functional interaction between SEL-10, an F-box protein, and the nuclear form of activated Notch1 receptor. J Biol Chem 2001; 276: 34371–34378.
O'Neil J, Grim J, Strack P, Rao S, Tibbitts D, Winter C et al. FBW7 mutations in leukemic cells mediate NOTCH pathway activation and resistance to gamma-secretase inhibitors. J Exp Med 2007; 204: 1813–1824.
Oberg C, Li J, Pauley A, Wolf E, Gurney M, Lendahl U . The Notch intracellular domain is ubiquitinated and negatively regulated by the mammalian Sel-10 homolog. J Biol Chem 2001; 276: 35847–35853.
Thompson BJ, Buonamici S, Sulis ML, Palomero T, Vilimas T, Basso G et al. The SCFFBW7 ubiquitin ligase complex as a tumor suppressor in T cell leukemia. J Exp Med 2007; 204: 1825–1835.
Wu G, Lyapina S, Das I, Li J, Gurney M, Pauley A et al. SEL-10 is an inhibitor of notch signaling that targets notch for ubiquitin-mediated protein degradation. Mol Cell Biol 2001; 21: 7403–7415.
Akhoondi S, Sun D, von der Lehr N, Apostolidou S, Klotz K, Maljukova A et al. FBXW7/hCDC4 is a general tumor suppressor in human cancer. Cancer Res 2007; 67: 9006–9012.
Tan Y, Sangfelt O, Spruck C . The Fbxw7/hCdc4 tumor suppressor in human cancer. Cancer Lett 2008.
Matsuoka S, Oike Y, Onoyama I, Iwama A, Arai F, Takubo K et al. Fbxw7 acts as a critical fail-safe against premature loss of hematopoietic stem cells and development of T-ALL. Genes Dev 2008; 22: 986–991.
Onoyama I, Tsunematsu R, Matsumoto A, Kimura T, de Alboran IM, Nakayama K et al. Conditional inactivation of Fbxw7 impairs cell-cycle exit during T cell differentiation and results in lymphomatogenesis. J Exp Med 2007; 204: 2875–2888.
Thompson BJ, Jankovic V, Gao J, Buonamici S, Vest A, Lee JM et al. Control of hematopoietic stem cell quiescence by the E3 ubiquitin ligase Fbw7. J Exp Med 2008; 205: 1395–1408.
Mao JH, Perez-Losada J, Wu D, Delrosario R, Tsunematsu R, Nakayama KI et al. Fbxw7/Cdc4 is a p53-dependent, haploinsufficient tumour suppressor gene. Nature 2004; 432: 775–779.
Perez-Losada J, Mao JH, Balmain A . Control of genomic instability and epithelial tumor development by the p53-Fbxw7/Cdc4 pathway. Cancer Res 2005; 65: 6488–6492.
Ishikawa Y, Onoyama I, Nakayama KI, Nakayama K . Notch-dependent cell cycle arrest and apoptosis in mouse embryonic fibroblasts lacking Fbxw7. Oncogene 2008.
Masuda K, Ishikawa Y, Onoyama I, Unno M, de Alboran IM, Nakayama KI et al. Complex regulation of cell-cycle inhibitors by Fbxw7 in mouse embryonic fibroblasts. Oncogene 2010; 29: 1798–1809.
Artavanis-Tsakonas S, Rand MD, Lake RJ . Notch signaling: cell fate control and signal integration in development. Science 1999; 284: 770–776.
Koch U, Radtke F . Notch and cancer: a double-edged sword. Cell Mol Life Sci 2007; 64: 2746–2762.
Nicolas M, Wolfer A, Raj K, Kummer JA, Mill P, van Noort M et al. Notch1 functions as a tumor suppressor in mouse skin. Nat Genet 2003; 33: 416–421.
Rangarajan A, Talora C, Okuyama R, Nicolas M, Mammucari C, Oh H et al. Notch signaling is a direct determinant of keratinocyte growth arrest and entry into differentiation. EMBO J 2001; 20: 3427–3436.
Lefort K, Mandinova A, Ostano P, Kolev V, Calpini V, Kolfschoten I et al. Notch1 is a p53 target gene involved in human keratinocyte tumor suppression through negative regulation of ROCK1/2 and MRCKalpha kinases. Genes Dev 2007; 21: 562–577.
Tetzlaff MT, Yu W, Li M, Zhang P, Finegold M, Mahon K et al. Defective cardiovascular development and elevated cyclin E and Notch proteins in mice lacking the Fbw7 F-box protein. Proc Natl Acad Sci USA 2004; 101: 3338–3345.
Tsunematsu R, Nakayama K, Oike Y, Nishiyama M, Ishida N, Hatakeyama S et al. Mouse Fbw7/Sel-10/Cdc4 is required for notch degradation during vascular development. J Biol Chem 2004; 279: 9417–9423.
Yu L, Hitchler MJ, Sun W, Sarsour EH, Goswami PC, Klingelhutz AJ et al. AP-2alpha inhibits c-MYC induced oxidative stress and apoptosis in HaCaT human keratinocytes. J Oncol 2009. 780874.
Mandinova A, Lefort K, Tommasi di Vignano A, Stonely W, Ostano P, Chiorino G et al. The FoxO3a gene is a key negative target of canonical Notch signalling in the keratinocyte UVB response. EMBO J 2008; 27: 1243–1254.
Dlugosz AA, Yuspa SH . Coordinate changes in gene expression which mark the spinous to granular cell transition in epidermis are regulated by protein kinase C. J Cell Biol 1993; 120: 217–225.
Land H, Parada LF, Weinberg RA . Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature 1983; 304: 596–602.
Rounbehler RJ, Schneider-Broussard R, Conti CJ, Johnson DG . Myc lacks E2F1's ability to suppress skin carcinogenesis. Oncogene 2001; 20: 5341–5349.
Owens DM, Watt FM . Contribution of stem cells and differentiated cells to epidermal tumours. Nat Rev Cancer 2003; 3: 444–451.
Fuchs E . Skin stem cells: rising to the surface. J Cell Biol 2008; 180: 273–284.
Barrandon Y, Green H . Cell size as a determinant of the clone-forming ability of human keratinocytes. Proc Natl Acad Sci USA 1985; 82: 5390–5394.
Morris RJ, Tacker KC, Fischer SM, Slaga TJ . Quantitation of primary in vitro clonogenic keratinocytes from normal adult murine epidermis, following initiation, and during promotion of epidermal tumors. Cancer Res 1988; 48: 6285–6290.
Matsumoto A, Onoyama I, Sunabori T, Kageyama R, Okano H, Nakayama KI . Fbxw7-dependent degradation of Notch is required for control of "stemness" and neuronal-glial differentiation in neural stem cells. J Biol Chem 2011; 286: 13754–13764.
Babaei-Jadidi R, Li N, Saadeddin A, Spencer-Dene B, Jandke A, Muhammad B et al. FBXW7 influences murine intestinal homeostasis and cancer, targeting Notch, Jun, and DEK for degradation. J Exp Med 2011; 208: 295–312.
Nguyen BC, Lefort K, Mandinova A, Antonini D, Devgan V, Della Gatta G et al. Cross-regulation between Notch and p63 in keratinocyte commitment to differentiation. Genes Dev 2006; 20: 1028–1042.
Okuyama R, Ogawa E, Nagoshi H, Yabuki M, Kurihara A, Terui T et al. p53 homologue, p51/p63, maintains the immaturity of keratinocyte stem cells by inhibiting Notch1 activity. Oncogene 2007; 26: 4478–4488.
Senoo M, Pinto F, Crum CP, McKeon F . p63 Is essential for the proliferative potential of stem cells in stratified epithelia. Cell 2007; 129: 523–536.
Benitah SA, Frye M, Glogauer M, Watt FM . Stem cell depletion through epidermal deletion of Rac1. Science 2005; 309: 933–935.
Dotto GP, Cotsarelis G . Developmental biology. Rac1 up for epidermal stem cells. Science 2005; 309: 890–891.
Tanigaki K, Han H, Yamamoto N, Tashiro K, Ikegawa M, Kuroda K et al. Notch-RBP-J signaling is involved in cell fate determination of marginal zone B cells. Nat Immunol 2002; 3: 443–450.
de Alboran IM, O'Hagan RC, Gartner F, Malynn B, Davidson L, Rickert R et al. Analysis of C-MYC function in normal cells via conditional gene-targeted mutation. Immunity 2001; 14: 45–55.
Morita S, Kojima T, Kitamura T . Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther 2000; 7: 1063–1066.
Kitagawa M, Hatakeyama S, Shirane M, Matsumoto M, Ishida N, Hattori K et al. An F-box protein, FWD1, mediates ubiquitin-dependent proteolysis of beta-catenin. EMBO J 1999; 18: 2401–2410.
Acknowledgements
We thank T Honjo for RbpjF/F mice; IM de Alborán for c-MycF/F mice; R Tsunematsu for the NICD1 plasmid; T Kitamura for pMX-puro and Plat-E cells; Y Gotoh for pMX-Cre-SV40-GFP; S Ikawa for the cDNA-encoding H-RasG12V; E Ogawa and E Nishimura for technical advice; Y Ono, T Senga and N Yanagihara for technical assistance; and other laboratory members for discussion. This study was supported in part by a Grant-in-Aid from the Ministry of Education, Culture, Sports, Science, and Technology of Japan and by the Global Center of Excellence Program of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ishikawa, Y., Hosogane, M., Okuyama, R. et al. Opposing functions of Fbxw7 in keratinocyte growth, differentiation and skin tumorigenesis mediated through negative regulation of c-Myc and Notch. Oncogene 32, 1921–1932 (2013). https://doi.org/10.1038/onc.2012.213
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.213
Keywords
This article is cited by
-
Mutations observed in somatic evolution reveal underlying gene mechanisms
Communications Biology (2023)
-
Comprehensive Study of Human FBXW7 Deleterious nsSNP’s Functional Inference and Susceptibility to Gynaecological Cancer
Applied Biochemistry and Biotechnology (2022)
-
The role of FBXW7, a cell-cycle regulator, as a predictive marker of recurrence of gastrointestinal stromal tumors
Gastric Cancer (2019)
-
FBXW7 regulates glucocorticoid response in T-cell acute lymphoblastic leukaemia by targeting the glucocorticoid receptor for degradation
Leukemia (2013)