Abstract
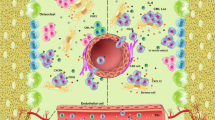
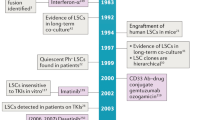
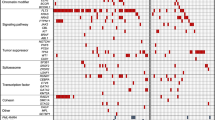
Human acute myeloid leukemias (AMLs) are sustained by leukemic stem cells (LSCs) that generate through aberrant differentiation the blast cells that make up the bulk of the malignant clone. LSCs were first identified as rare cells with an immunophenotype shared with normal hematopoietic stem cells (HSCs). However, refinements of xenotransplantation assays, alternative methods of quantitation and syngeneic murine models have all led to an appreciation that LSCs display marked variability in frequency, immunophenotype and differentiation potential, both between and even within leukemias. Insights from next-generation sequencing efforts have dramatically extended understanding of the mutational landscape and clonal organization of AML and have added an additional layer of complexity to the biology of LSCs: a requirement to consider the effect of the various recurrently occurring genetic lesions in AML on the initiation and maintenance of leukemic subclones. Despite these advances, cure rates in AML remain substantially unchanged in recent years. A renewed focus on the biological properties of chemotherapy-resistant LSCs, a cellular entity of prime clinical importance, will be required to develop additional therapeutic strategies to enhance patient outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Spangrude GJ, Smith L, Uchida N, Ikuta K, Heimfeld S, Friedman J et al. Mouse hematopoietic stem cells. Blood 1991; 78: 1395–1402.
Orlic D, Bodine DM . What defines a pluripotent hematopoietic stem cell (PHSC): will the real PHSC please stand up!. Blood 1994; 84: 3991–3994.
Osawa M, Hanada K, Hamada H, Nakauchi H . Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell. Science 1996; 273: 242–245.
Kiel MJ, Yilmaz OH, Iwashita T, Terhorst C, Morrison SJ . SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 2005; 121: 1109–1121.
Dicke KA, Spitzer G, Ahearn MJ . Colony formation in vitro by leukaemic cells in acute myelogenous leukaemia with phytohaemagglutinin as stimulating factor. Nature 1976; 259: 129–130.
Griffin JD, Lowenberg B . Clonogenic cells in acute myeloblastic leukemia. Blood 1986; 68: 1185–1195.
Buick RN, Minden MD, McCulloch EA . Self-renewal in culture of proliferative blast progenitor cells in acute myeloblastic leukemia. Blood 1979; 54: 95–104.
Sabbath KD, Ball ED, Larcom P, Davis RB, Griffin JD . Heterogeneity of clonogenic cells in acute myeloblastic leukemia. J Clin Invest 1985; 75: 746–753.
Bonnet D, Dick JE . Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997; 3: 730–737.
Spitzer G, Dicke KA, Gehan EA, Smith T, McCredie KB, Barlogie B et al. A simplified in vitro classification for prognosis in adult acute leukemia: the application of in vitro results in remission-predictive models. Blood 1976; 48: 795–807.
Swart K, Hagemeijer A, Lowenberg B . Acute myeloid leukemia colony growth in vitro: differences of colony-forming cells in PHA-supplemented and standard leukocyte feeder cultures. Blood 1982; 59: 816–821.
Harris WJ, Huang X, Lynch JT, Spencer GJ, Hitchin JR, Li Y et al. The histone demethylase KDM1A sustains the oncogenic potential of MLL-AF9 leukemia stem cells. Cancer Cell 2012; 21: 473–487.
Shultz LD, Lyons BL, Burzenski LM, Gott B, Chen X, Chaleff S et al. Human lymphoid and myeloid cell development in NOD/LtSz-scid IL2R gamma null mice engrafted with mobilized human hemopoietic stem cells. J Immunol 2005; 174: 6477–6489.
Sanchez PV, Perry RL, Sarry JE, Perl AE, Murphy K, Swider CR et al. A robust xenotransplantation model for acute myeloid leukemia. Leukemia 2009; 23: 2109–2117.
Taussig DC, Miraki-Moud F, Anjos-Afonso F, Pearce DJ, Allen K, Ridler C et al. Anti-CD38 antibody-mediated clearance of human repopulating cells masks the heterogeneity of leukemia-initiating cells. Blood 2008; 112: 568–575.
Ishikawa F, Yoshida S, Saito Y, Hijikata A, Kitamura H, Tanaka S et al. Chemotherapy-resistant human AML stem cells home to and engraft within the bone-marrow endosteal region. Nat Biotechnol 2007; 25: 1315–1321.
Wunderlich M, Chou FS, Link KA, Mizukawa B, Perry RL, Carroll M et al. AML xenograft efficiency is significantly improved in NOD/SCID-IL2RG mice constitutively expressing human SCF, GM-CSF and IL-3. Leukemia 2010; 24: 1785–1788.
Vargaftig J, Taussig DC, Griessinger E, Anjos-Afonso F, Lister TA, Cavenagh J et al. Frequency of leukemic initiating cells does not depend on the xenotransplantation model used. Leukemia 2012; 26: 858–860.
Ailles LE, Gerhard B, Kawagoe H, Hogge DE . Growth characteristics of acute myelogenous leukemia progenitors that initiate malignant hematopoiesis in nonobese diabetic/severe combined immunodeficient mice. Blood 1999; 94: 1761–1772.
Pearce DJ, Taussig D, Zibara K, Smith LL, Ridler CM, Preudhomme C et al. AML engraftment in the NOD/SCID assay reflects the outcome of AML: implications for our understanding of the heterogeneity of AML. Blood 2006; 107: 1166–1173.
Patel S, Zhang Y, Cassinat B, Zassadowski F, Ferre N, Cuccuini W et al. Successful xenografts of AML3 samples in immunodeficient NOD/shi-SCID IL2Rgamma(−)/(−) mice. Leukemia 2012; 26: 2432–2435.
Sarry JE, Murphy K, Perry R, Sanchez PV, Secreto A, Keefer C et al. Human acute myelogenous leukemia stem cells are rare and heterogeneous when assayed in NOD/SCID/IL2Rgammac-deficient mice. J Clin Invest 2011; 121: 384–395.
Notta F, Mullighan CG, Wang JC, Poeppl A, Doulatov S, Phillips LA et al. Evolution of human BCR-ABL1 lymphoblastic leukaemia-initiating cells. Nature 2011; 469: 362–367.
Somervaille TC, Cleary ML . Identification and characterization of leukemia stem cells in murine MLL-AF9 acute myeloid leukemia. Cancer Cell 2006; 10: 257–268.
Krivtsov AV, Twomey D, Feng Z, Stubbs MC, Wang Y, Faber J et al. Transformation from committed progenitor to leukaemia stem cell initiated by MLL-AF9. Nature 2006; 442: 818–822.
Kelly PN, Dakic A, Adams JM, Nutt SL, Strasser A . Tumor growth need not be driven by rare cancer stem cells. Science 2007; 317: 337.
Somervaille TC, Matheny CJ, Spencer GJ, Iwasaki M, Rinn JL, Witten DM et al. Hierarchical maintenance of MLL myeloid leukemia stem cells employs a transcriptional program shared with embryonic rather than adult stem cells. Cell Stem Cell 2009; 4: 129–140.
Wilhelm BT, Briau M, Austin P, Faubert A, Boucher G, Chagnon P et al. RNA-seq analysis of 2 closely related leukemia clones that differ in their self-renewal capacity. Blood 2011; 117: e27–e38.
Yilmaz OH, Valdez R, Theisen BK, Guo W, Ferguson DO, Wu H et al. Pten dependence distinguishes haematopoietic stem cells from leukaemia-initiating cells. Nature 2006; 441: 475–482.
Lange B, Ferrero D, Pessano S, Palumbo A, Faust J, Meo P et al. Surface phenotype of clonogenic cells in acute myeloid leukemia defined by monoclonal antibodies. Blood 1984; 64: 693–700.
Terpstra W, Prins A, Ploemacher RE, Wognum BW, Wagemaker G, Lowenberg B et al. Long-term leukemia-initiating capacity of a CD34-subpopulation of acute myeloid leukemia. Blood 1996; 87: 2187–2194.
Blair A, Hogge DE, Ailles LE, Lansdorp PM, Sutherland HJ . Lack of expression of Thy-1 (CD90) on acute myeloid leukemia cells with long-term proliferative ability in vitro and in vivo. Blood 1997; 89: 3104–3112.
Saito Y, Kitamura H, Hijikata A, Tomizawa-Murasawa M, Tanaka S, Takagi S et al. Identification of therapeutic targets for quiescent, chemotherapy-resistant human leukemia stem cells. Sci Transl Med 2010; 2: 17ra19.
Martelli MP, Pettirossi V, Thiede C, Bonifacio E, Mezzasoma F, Cecchini D et al. CD34+ cells from AML with mutated NPM1 harbor cytoplasmic mutated nucleophosmin and generate leukemia in immunocompromised mice. Blood 2010; 116: 3907–3922.
Taussig DC, Vargaftig J, Miraki-Moud F, Griessinger E, Sharrock K, Luke T et al. Leukemia-initiating cells from some acute myeloid leukemia patients with mutated nucleophosmin reside in the CD34(−) fraction. Blood 2010; 115: 1976–1984.
Goardon N, Marchi E, Atzberger A, Quek L, Schuh A, Soneji S et al. Coexistence of LMPP-like and GMP-like leukemia stem cells in acute myeloid leukemia. Cancer Cell 2011; 19: 138–152.
Eppert K, Takenaka K, Lechman ER, Waldron L, Nilsson B, van Galen P et al. Stem cell gene expression programs influence clinical outcome in human leukemia. Nat Med 2011; 17: 1086–1093.
Notta F, Doulatov S, Laurenti E, Poeppl A, Jurisica I, Dick JE . Isolation of single human hematopoietic stem cells capable of long-term multilineage engraftment. Science 2011; 333: 218–221.
Bereshchenko O, Mancini E, Moore S, Bilbao D, Mansson R, Luc S et al. Hematopoietic stem cell expansion precedes the generation of committed myeloid leukemia-initiating cells in C/EBPalpha mutant AML. Cancer Cell 2009; 16: 390–400.
Gibbs KD Jr., Jager A, Crespo O, Goltsev Y, Trejo A, Richard CE et al. Decoupling of tumor-initiating activity from stable immunophenotype in HoxA9-Meis1-driven AML. Cell Stem Cell 2012; 10: 210–217.
Jamieson CH, Ailles LE, Dylla SJ, Muijtjens M, Jones C, Zehnder JL et al. Granulocyte–macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N Engl J Med 2004; 351: 657–667.
Cozzio A, Passegue E, Ayton PM, Karsunky H, Cleary ML, Weissman IL . Similar MLL-associated leukemias arising from self-renewing stem cells and short-lived myeloid progenitors. Genes Dev 2003; 17: 3029–3035.
Passegue E, Jamieson CH, Ailles LE, Weissman IL . Normal and leukemic hematopoiesis: are leukemias a stem cell disorder or a reacquisition of stem cell characteristics? Proc Natl Acad Sci USA 2003; 100 (Suppl 1): 11842–11849.
Huntly BJ, Shigematsu H, Deguchi K, Lee BH, Mizuno S, Duclos N et al. MOZ-TIF2, but not BCR-ABL, confers properties of leukemic stem cells to committed murine hematopoietic progenitors. Cancer Cell 2004; 6: 587–596.
Fialkow PJ, Jacobson RJ, Papayannopoulou T . Chronic myelocytic leukemia: clonal origin in a stem cell common to the granulocyte, erythrocyte, platelet and monocyte/macrophage. Am J Med 1977; 63: 125–130.
Martin PJ, Najfeld V, Hansen JA, Penfold GK, Jacobson RJ, Fialkow PJ . Involvement of the B-lymphoid system in chronic myelogenous leukaemia. Nature 1980; 287: 49–50.
Jonas D, Lubbert M, Kawasaki ES, Henke M, Bross KJ, Mertelsmann R et al. Clonal analysis of bcr-abl rearrangement in T lymphocytes from patients with chronic myelogenous leukemia. Blood 1992; 79: 1017–1023.
Janssen JW, Buschle M, Layton M, Drexler HG, Lyons J, van den Berghe H et al. Clonal analysis of myelodysplastic syndromes: evidence of multipotent stem cell origin. Blood 1989; 73: 248–254.
Blackstock AM, Garson OM . Direct evidence for involvement of erythroid cells in acute myeloblastic leukaemia. Lancet 1974; 2: 1178–1179.
Keinanen M, Griffin JD, Bloomfield CD, Machnicki J, de la Chapelle A . Clonal chromosomal abnormalities showing multiple-cell-lineage involvement in acute myeloid leukemia. N Engl J Med 1988; 318: 1153–1158.
WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues 4th edn. WHO Press, Geneva, Switzerland, 2008.
Gale RE, Wheadon H, Goldstone AH, Burnett AK, Linch DC . Frequency of clonal remission in acute myeloid leukaemia. Lancet 1993; 341: 138–142.
Fialkow PJ, Janssen JW, Bartram CR . Clonal remissions in acute nonlymphocytic leukemia: evidence for a multistep pathogenesis of the malignancy. Blood 1991; 77: 1415–1417.
Lo Coco F, Pelicci PG, D'Adamo F, Diverio D, Alimena G, Montefusco E et al. Polyclonal hematopoietic reconstitution in leukemia patients at remission after suppression of specific gene rearrangements. Blood 1993; 82: 606–612.
Jowitt SN, Liu Yin JA, Saunders MJ, Lucas GS . Clonal remissions in acute myeloid leukaemia are commonly associated with features of trilineage myelodysplasia during remission. Br J Haematol 1993; 85: 698–705.
Jinnai I, Nagai K, Yoshida S, Toyoda A, Murohashi I, Bessho M et al. Incidence and characteristics of clonal hematopoiesis in remission of acute myeloid leukemia in relation to morphological dysplasia. Leukemia 1995; 9: 1756–1761.
Fearon ER, Burke PJ, Schiffer CA, Zehnbauer BA, Vogelstein B . Differentiation of leukemia cells to polymorphonuclear leukocytes in patients with acute nonlymphocytic leukemia. N Engl J Med 1986; 315: 15–24.
Jan M, Snyder TM, Corces-Zimmerman MR, Vyas P, Weissman IL, Quake SR et al. Clonal evolution of preleukemic hematopoietic stem cells precedes human acute myeloid leukemia. Sci Transl Med 2012; 4: 149ra118.
Delhommeau F, Dupont S, Della Valle V, James C, Trannoy S, Masse A et al. Mutation in TET2 in myeloid cancers. N Engl J Med 2009; 360: 2289–2301.
Busque L, Patel JP, Figueroa ME, Vasanthakumar A, Provost S, Hamilou Z et al. Recurrent somatic TET2 mutations in normal elderly individuals with clonal hematopoiesis. Nat Genet 2012; 44: 1179–1181.
Miyamoto T, Weissman IL, Akashi K . AML1/ETO-expressing nonleukemic stem cells in acute myelogenous leukemia with 8;21 chromosomal translocation. Proc Natl Acad Sci USA 2000; 97: 7521–7526.
Ley TJ, Mardis ER, Ding L, Fulton B, McLellan MD, Chen K et al. DNA sequencing of a cytogenetically normal acute myeloid leukaemia genome. Nature 2008; 456: 66–72.
Mardis ER, Ding L, Dooling DJ, Larson DE, McLellan MD, Chen K et al. Recurring mutations found by sequencing an acute myeloid leukemia genome. N Engl J Med 2009; 361: 1058–1066.
Ley TJ, Ding L, Walter MJ, McLellan MD, Lamprecht T, Larson DE et al. DNMT3A mutations in acute myeloid leukemia. N Engl J Med 2010; 363: 2424–2433.
Yan XJ, Xu J, Gu ZH, Pan CM, Lu G, Shen Y et al. Exome sequencing identifies somatic mutations of DNA methyltransferase gene DNMT3A in acute monocytic leukemia. Nat Genet 2011; 43: 309–315.
Ding L, Ley TJ, Larson DE, Miller CA, Koboldt DC, Welch JS et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature 2012; 481: 506–510.
Walter MJ, Shen D, Ding L, Shao J, Koboldt DC, Chen K et al. Clonal architecture of secondary acute myeloid leukemia. N Engl J Med 2012; 366: 1090–1098.
Mullighan CG, Phillips LA, Su X, Ma J, Miller CB, Shurtleff SA et al. Genomic analysis of the clonal origins of relapsed acute lymphoblastic leukemia. Science 2008; 322: 1377–1380.
Meyer JA, Wang J, Hogan LE, Yang JJ, Dandekar S, Patel JP et al. Relapse-specific mutations in NT5C2 in childhood acute lymphoblastic leukemia. Nat Genet 2013; 45: 290–294.
Tzoneva G, Perez-Garcia A, Carpenter Z, Khiabanian H, Tosello V, Allegretta M et al. Activating mutations in the NT5C2 nucleotidase gene drive chemotherapy resistance in relapsed ALL. Nat Med 2013; 19: 368–371.
Beekman R, Valkhof MG, Sanders MA, van Strien PM, Haanstra JR, Broeders L et al. Sequential gain of mutations in severe congenital neutropenia progressing to acute myeloid leukemia. Blood 2012; 119: 5071–5077.
Welch JS, Ley TJ, Link DC, Miller CA, Larson DE, Koboldt DC et al. The origin and evolution of mutations in acute myeloid leukemia. Cell 2012; 150: 264–278.
Gilliland DG, Griffin JD . The roles of FLT3 in hematopoiesis and leukemia. Blood 2002; 100: 1532–1542.
Shih AH, Abdel-Wahab O, Patel JP, Levine RL . The role of mutations in epigenetic regulators in myeloid malignancies. Nat Rev Cancer 2012; 12: 599–612.
Nakano Y, Kiyoi H, Miyawaki S, Asou N, Ohno R, Saito H et al. Molecular evolution of acute myeloid leukaemia in relapse: unstable N-ras and FLT3 genes compared with p53 gene. Br J Haematol 1999; 104: 659–664.
Kottaridis PD, Gale RE, Langabeer SE, Frew ME, Bowen DT, Linch DC . Studies of FLT3 mutations in paired presentation and relapse samples from patients with acute myeloid leukemia: implications for the role of FLT3 mutations in leukemogenesis, minimal residual disease detection, and possible therapy with FLT3 inhibitors. Blood 2002; 100: 2393–2398.
Anderson K, Lutz C, van Delft FW, Bateman CM, Guo Y, Colman SM et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature 2011; 469: 356–361.
Crowther D, Bateman CJ, Vartan CP, Whitehouse JM, Malpas JS, Fairley GH et al. Combination chemotherapy using L-asparaginase, daunorubicin, and cytosine arabinoside in adults with acute myelogenous leukaemia. BMJ 1970; 4: 513–517.
Lowenberg B, Downing JR, Burnett A . Acute myeloid leukemia. N Engl J Med 1999; 341: 1051–1062.
Burnett A, Wetzler M, Lowenberg B . Therapeutic advances in acute myeloid leukemia. J Clin Oncol 2011; 29: 487–494.
Terpstra W, Ploemacher RE, Prins A, van Lom K, Pouwels K, Wognum AW et al. Fluorouracil selectively spares acute myeloid leukemia cells with long-term growth abilities in immunodeficient mice and in culture. Blood 1996; 88: 1944–1950.
Minden MD, Till JE, McCulloch EA . Proliferative state of blast cell progenitors in acute myeloblastic leukemia (AML). Blood 1978; 52: 592–600.
Guzman ML, Neering SJ, Upchurch D, Grimes B, Howard DS, Rizzieri DA et al. Nuclear factor-kappaB is constitutively activated in primitive human acute myelogenous leukemia cells. Blood 2001; 98: 2301–2307.
Guan Y, Gerhard B, Hogge DE . Detection, isolation, and stimulation of quiescent primitive leukemic progenitor cells from patients with acute myeloid leukemia (AML). Blood 2003; 101: 3142–3149.
Zuber J, Radtke I, Pardee TS, Zhao Z, Rappaport AR, Luo W et al. Mouse models of human AML accurately predict chemotherapy response. Genes Dev 2009; 23: 877–889.
Weiden PL, Sullivan KM, Flournoy N, Storb R, Thomas ED . Antileukemic effect of chronic graft-versus-host disease: contribution to improved survival after allogeneic marrow transplantation. N Engl J Med 1981; 304: 1529–1533.
Vago L, Perna SK, Zanussi M, Mazzi B, Barlassina C, Stanghellini MT et al. Loss of mismatched HLA in leukemia after stem-cell transplantation. N Engl J Med 2009; 361: 478–488.
Taussig DC, Pearce DJ, Simpson C, Rohatiner AZ, Lister TA, Kelly G et al. Hematopoietic stem cells express multiple myeloid markers: implications for the origin and targeted therapy of acute myeloid leukemia. Blood 2005; 106: 4086–4092.
Burnett AK, Hills RK, Milligan D, Kjeldsen L, Kell J, Russell NH et al. Identification of patients with acute myeloblastic leukemia who benefit from the addition of gemtuzumab ozogamicin: results of the MRC AML15 trial. J Clin Oncol 2011; 29: 369–377.
Castaigne S, Pautas C, Terre C, Raffoux E, Bordessoule D, Bastie JN et al. Effect of gemtuzumab ozogamicin on survival of adult patients with de-novo acute myeloid leukaemia (ALFA-0701): a randomised, open-label, phase 3 study. Lancet 2012; 379: 1508–1516.
Majeti R, Chao MP, Alizadeh AA, Pang WW, Jaiswal S, Gibbs KD Jr. et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell 2009; 138: 286–299.
Hosen N, Park CY, Tatsumi N, Oji Y, Sugiyama H, Gramatzki M et al. CD96 is a leukemic stem cell-specific marker in human acute myeloid leukemia. Proc Natl Acad Sci USA 2007; 104: 11008–11013.
van Rhenen A, van Dongen GA, Kelder A, Rombouts EJ, Feller N, Moshaver B et al. The novel AML stem cell associated antigen CLL-1 aids in discrimination between normal and leukemic stem cells. Blood 2007; 110: 2659–2666.
Kikushige Y, Shima T, Takayanagi S, Urata S, Miyamoto T, Iwasaki H et al. TIM-3 is a promising target to selectively kill acute myeloid leukemia stem cells. Cell Stem Cell 2010; 7: 708–717.
The Cancer Genome Atlas Research Network, Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N Engl J Med 2013; 368: 2059–2074.
Acknowledgements
We apologize to the many clinicians and scientists who have contributed to this field whose work we were unable to cite because of space constraints. DW and BG are recipients of Leukaemia and Lymphoma Research Clinical Training Fellowships. TS is supported by Cancer Research UK grant number C5759/A12328.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Wiseman, D., Greystoke, B. & Somervaille, T. The variety of leukemic stem cells in myeloid malignancy. Oncogene 33, 3091–3098 (2014). https://doi.org/10.1038/onc.2013.269
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.269
Keywords
This article is cited by
-
Regulation of HOX gene expression in AML
Blood Cancer Journal (2024)
-
Bone marrow-targetable Green Tea Catechin-Based Micellar Nanocomplex for synergistic therapy of Acute myeloid leukemia
Journal of Nanobiotechnology (2022)
-
ASXL1/2 mutations and myeloid malignancies
Journal of Hematology & Oncology (2022)
-
Leukemic stem cell signatures in Acute myeloid leukemia- targeting the Guardians with novel approaches
Stem Cell Reviews and Reports (2022)
-
FOXM1 regulates leukemia stem cell quiescence and survival in MLL-rearranged AML
Nature Communications (2020)