Abstract
MRI of the sacroiliac joints is increasingly acknowledged as being indispensable in the early diagnosis of axial spondyloarthritis (axSpA) and as having a prominent role in the prognosis and classification of axSpA. Technological advances include improvements in the resolution of structural lesions and in methodologies for the quantification of lesions. Limited access and expertise in interpretation of MRI have led to a resurgence of interest in CT, especially the development of low radiation protocols for assessing the sacroiliac joints. Trials of TNF inhibitors in patients with non-radiographic axSpA have led to greater understanding of the role of MRI in selecting which patients might respond well to this therapy. The role of MRI features as target end points in treat-to-target strategies remains unclear because the effect of such targeting on structural damage parameters has only recently been explored. The relative importance of active and structural lesions for prognostic risk assessment and selection of appropriate treatment is also an area of current research. Given the increased capacity to visualize a broad array of lesions in both the sacroiliac joints and the spine using MRI and CT, these modalities will probably be increasingly employed for assessment of the disease-modifying activity of new therapies.
Key points
-
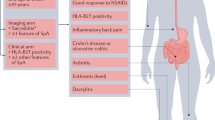
Low-dose CT of the sacroiliac joints has superior diagnostic accuracy to plain radiography for axial spondyloarthritis (axSpA) and can be widely implemented.
-
MRI scans of the sacroiliac joint should be interpreted contextually using complementary diagnostic information provided by T1-weighted and fat-suppressed sequences.
-
Routine MRI evaluation of patients presenting with back pain and suspected axSpA should be confined to the pelvis because imaging of the spine is of little additional benefit.
-
The utility of the assessment of sacroiliac joint structural lesions or spinal lesions by MRI for improving current classification for axSpA is still uncertain.
-
Lesions in the sacroiliac joints and spine on MRI have potential prognostic value for structural progression, especially the presence of fat metaplasia.
-
MRI inflammatory scores can discriminate between active therapy and placebo and have been a consistent predictor of the efficacy of novel treatments in clinical trials over the past two decades.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Maksymowych, W. P. Seronegative spondyloarthritis: diagnosis and management. DeckerIP https://www.deckerip.com/products/medicine/table-of-contents/ (2018).
Van Mens, L. J. J., van de Sande, M. G. H. & Baeten, D. L. P. New treatment paradigms in spondyloarthritis. Curr. Opin. Rheumatol. 30, 79–86 (2018).
Dougados, M. et al. The DESIR cohort: a 10-year follow-up of early inflammatory back pain in France: study design and baseline characteristics of the 708 recruited patients. Joint Bone Spine 78, 598–603 (2011).
Dougados, M. et al. Sacroiliac radiographic progression in recent onset axial spondyloarthritis: the 5-year data of the DESIR cohort. Ann. Rheum. Dis. 76, 1823–1828 (2017).
van den Berg, R. et al. Percentage of patients with spondyloarthritis in patients referred because of chronic back pain and performance of classification criteria: experience from the Spondyloarthritis Caught Early (SPACE) cohort. Rheumatology 52, 1492–1499 (2013).
Ez-Zaitouni, Z. et al. Presence of multiple spondyloarthritis (SpA) features is important but not sufficient for a diagnosis of axial spondyloarthritis: data from the SPondyloArthritis Caught Early (SPACE) cohort. Ann. Rheum. Dis. 76, 1086–1092 (2017).
van den Berg, R. et al. Agreement between clinical practice and trained central reading in reading of sacroiliac joints on plain pelvic radiographs. Results from the DESIR cohort. Arthritis Rheumatol. 66, 2403–2411 (2014).
Christiansen, A. A. et al. Limited reliability of radiographic assessment of sacroiliac joints in patients with suspected early spondyloarthritis. J. Rheumatol. 44, 70–77 (2017).
Jaremko, J. L. et al. Diagnostic utility of magnetic resonance imaging and radiography in juvenile spondyloarthritis: evaluation of the sacroiliac joints in controls and affected subjects. J. Rheumatol. 41, 963–970 (2014).
Weiss, P. et al. Radiographs in screening for sacroiliitis in children: what is the value? Arthritis Res. Ther. 20, 141 (2018).
Diekhoff, T. et al. Comparison of MRI with radiography for detecting structural lesions of the sacroiliac joint using CT as standard of reference: results from the SIMACT study. Ann. Rheum. Dis. 76, 1502–1508 (2017).
Mandl, P. et al. EULAR recommendations for the use of imaging in the diagnosis and management of spondyloarthritis in clinical practice. Ann. Rheum. Dis. 74, 1327–1339 (2015).
Sudoł-Szopińska, I. et al. Recommendations of the ESSR Arthritis Subcommittee for the use of magnetic resonance imaging in musculoskeletal rheumatic diseases. Semin. Musculoskelet. Radiol. 19, 396–411 (2015).
Maksymowych, W. P. et al. Suppression of inflammation and effects on new bone formation in ankylosing spondylitis: evidence for a window of opportunity in disease modification. Ann. Rheum. Dis. 72, 23–28 (2013).
Park, J. W. et al. Impact of tumor necrosis factor inhibitor versus nonsteroidal antiinflammatory drug treatment on radiographic progression in early ankylosing spondylitis: its relationship to inflammation control during treatment. Arthritis Rheumatol. 71, 82–90 (2019).
Devauchelle-Pensec, V. et al. Computed tomography scanning facilitates the diagnosis of sacroiliitis in patients with suspected spondylarthritis. Arthritis Rheum. 64, 1412–1419 (2012).
Niemann, T., Kollmann, T. & Bongartz, G. Diagnostic performance of low-dose CT for the detection of urolithiasis: a meta-analysis. Am. J. Radiol. 191, 396–401 (2008).
Christe, A. et al. CT screening and follow-up of lung nodules: effects of tube, current-time setting, and nodule size and density on detectability and of tube current-time setting on apparent size. AJR Am. J. Roentgenol. 197, 623–630 (2011).
Chahal, B. S. et al. Radiation exposure to the sacroiliac joint from low-dose CT compared with radiography. Am. J. Radiol. 211, 1058–1062 (2018).
Martin, C. J. Effective dose: how should it be applied to medical exposures? Br. J. Radiol. 80, 639–647 (2007).
United States Environmental Protection Agency. Radiation sources and doses. EPA https://www.epa.gov/radiation/radiation-sources-and-doses (2019).
Jurik, A. G. et al. Effective radiation dose from semicoronal CT of the sacroiliac joints in comparison with axial CT and conventional radiography. Eur. Radiol. 12, 2820–2825 (2002).
Li, S. G., Liu, X., Zhou, H. & Zhang, Q. Interrater reliability and radiation dosage of oblique coronal computed tomography. Br. J. Radiol. 91, 20150700 (2018).
de Koning, A. et al. Low-dose CT detects more progression of bone formation in comparison to conventional radiography in patients with ankylosing spondylitis: results from the SIAS cohort. Ann. Rheum. Dis. 77, 293–299 (2018).
Ez-Zaitouni, Z. et al. Imaging of the sacroiliac joints is important for diagnosing early axial spondyloarthritis but not all-decisive. Rheumatology 57, 1173–1179 (2018).
Rudwaleit, M. et al. The development of Assessment of SpondyloArthritis international Society classification criteria for axial spondyloarthritis (part II): validation and final selection. Ann. Rheum. Dis. 68, 777–783 (2009).
Greese, J. et al. Detection of sacroiliitis by short-tau inversion recovery and T2-weighted turbo spin echo sequences: results from the SIMACT Study. J. Rheumatol. 46, 376–383 (2019).
Weber, U. et al. The diagnostic utility of magnetic resonance imaging in spondylarthritis: an international multicenter evaluation of one hundred eighty-seven subjects. Arthritis Rheum. 62, 3048–3058 (2010).
Rudwaleit, M. et al. How to diagnose axial spondyloarthritis early. Ann. Rheum. Dis. 63, 535–543 (2004).
De Hooge, M. et al. Magnetic resonance imaging of the sacroiliac joints in the early detection of spondyloarthritis: no added value of gadolinium compared with short tau inversion recovery sequence. Rheumatology 52, 1220–1224 (2013).
Weiss, P. F., Xiao, R., Biko, D. M., Johnson, A. M. & Chauvin, N. A. Detection of inflammatory sacroiliitis in children with magnetic resonance imaging is gadolinium contrast enhancement necessary? Arthritis Rheumatol. 67, 2250–2256 (2015).
Maksymowych, W. P. et al. Spondyloarthritis Research Consortium of Canada magnetic resonance imaging index for assessment of sacroiliac joint inflammation in ankylosing spondylitis. Arthritis. Care Res. 53, 703–709 (2005).
Maksymowych, W. P., Wichuk, S., Chiowchanwisawakit, P., Lambert, R. G. & Pedersen, S. J. Development and preliminary validation of the Spondyloarthritis Research Consortium of Canada magnetic resonance imaging sacroiliac joint structural score. J. Rheumatol. 42, 79–86 (2015).
Maksymowych, W. P. et al. MRI lesions in the sacroiliac joints of patients with spondyloarthritis: update of definitions and validation by the ASAS MRI working group. Ann. Rheum. Dis. https://doi.org/10.1136/annrheumdis-2019-215589 (2019).
Ciurea, A. et al. Tumor necrosis factor α inhibition in radiographic and nonradiographic axial spondyloarthritis: results from a large observational cohort. Arthritis Rheum. 65, 3096–3106 (2013).
Rudwaleit, M. et al. Defining active sacroiliitis on magnetic resonance imaging (MRI) for classification of axial spondyloarthritis: a consensual approach by the ASAS/OMERACT MRI group. Ann. Rheum. Dis. 68, 1520–1527 (2009).
van den Berg, R. et al. Classification of axial SpA based on positive imaging (radiographs and/or MRI of the sacroiliac joints) by local rheumatologists or radiologists versus central trained readers in the DESIR cohort. Ann. Rheum. Dis. 74, 2016–2021 (2015).
Bakker, P. A. C. et al. Is it useful to repeat magnetic resonance imaging of the sacroiliac joints after three months or one year in the diagnosis of patients with chronic back pain and suspected axial spondyloarthritis? Arthritis Rheumatol. 71, 382–391 (2019).
Weber, U. et al. Frequency and anatomic distribution of magnetic resonance imaging features in the sacroiliac joints of young athletes: exploring “background noise” toward a data-driven definition of sacroiliitis in early spondyloarthritis. Arthritis Rheumatol. 70, 736–745 (2018).
De Winter, J. et al. Magnetic resonance imaging of the sacroiliac joints indicating sacroiliitis according to the Assessment of SpondyloArthritis international Society definition in healthy individuals, runners, and women with postpartum back pain. Arthritis Rheumatol. 70, 1042–1048 (2018).
Weber, U. et al. Candidate lesion-based criteria for defining a positive sacroiliac joint MRI in two cohorts of patients with axial spondyloarthritis. Ann. Rheum. Dis. 74, 1976–1982 (2015).
Gong, Y. et al. Ten years’ experience with needle biopsy in the early diagnosis of sacroiliitis. Arthritis Rheum. 64, 1399–1406 (2012).
Varkas, G. et al. Effect of mechanical stress on magnetic resonance imaging of the sacroiliac joints: assessment of military recruits by magnetic resonance imaging study. Rheumatology 57, 508–513 (2018).
Arnbak, B. et al. Associations between spondyloarthritis features and magnetic resonance imaging findings: a cross-sectional analysis of 1,020 patients with persistent low back pain. Arthritis Rheumatol. 68, 892–900 (2016).
Eshed, I. et al. Peripartum changes of the sacroiliac joints on MRI: increasing mechanical load correlating with signs of edema and inflammation kindling spondyloarthropathy in the genetically prone. Clin. Rheumatol. 34, 1419–1426 (2015).
Ling, M. A. et al. Osteitis condensans ilii may demonstrate bone marrow edema on sacroiliac joint magnetic resonance imaging. Int. J. Rheum. Dis. 21, 299–307 (2018).
Weber, U. et al. Fat infiltration on magnetic resonance imaging of the sacroiliac joints has limited diagnostic utility in nonradiographic axial spondyloarthritis. J. Rheumatol. 41, 75–83 (2014).
de Hooge, M. et al. Patients with chronic back pain of short duration from the SPACE cohort: which MRI structural lesions in the sacroiliac joints and inflammatory and structural lesions in the spine are most specific for axial spondyloarthritis? Ann. Rheum. Dis. 75, 1308–1314 (2016).
Weber, U. et al. Assessment of structural lesions in sacroiliac joints enhances diagnostic utility of magnetic resonance imaging in early spondylarthritis. Arthritis Care Res. 62, 1763–1771 (2010).
Maksymowych, W. P. et al. MRI evidence of structural changes in the sacroiliac joints of patients with nonradiographic axial spondyloarthritis even in the absence of MRI inflammation. Arthritis Res. Ther. 19, 126 (2017).
Maksymowych, W. P. et al. Fat metaplasia and backfill are key intermediaries in the development of sacroiliac joint ankylosis in patients with ankylosing spondylitis. Arthritis Rheumatol. 66, 2958–2967 (2014).
Pedersen, S. J., Wichuk, S., Chiowchanwisawakit, P., Lambert, R. G. & Maksymowych, W. P. Tumor necrosis factor inhibitor therapy but not standard therapy is associated with resolution of erosion in the sacroiliac joints of patients with axial spondyloarthritis. Arthritis Res. Ther. 16, R100 (2014).
Maksymowych, W. P. et al. Modification of structural lesions on magnetic resonance imaging of the sacroiliac joints by etanercept in the EMBARK trial: a 12-week randomised placebo-controlled trial in patients with non-radiographic axial spondyloarthritis. Ann. Rheum. Dis. 77, 78–84 (2018).
Laloo, F. et al. MR signal in the sacroiliac joint space in spondyloarthritis: a new sign. Eur. Radiol. 27, 2024–2030 (2017).
Azmat, O., Lambert, R. G., Jibri, Z. & Maksymowych, W. P. Subchondral bone sclerosis on computed tomography – does it have any value in the diagnosis of inflammatory sacroiliitis or is it a non-specific finding? [abstract 684]. Arthritis Rheumatol. 68 (Suppl. 10), 899–901 (2016).
Hermann, K. G. et al. Descriptions of spinal MRI lesions and definition of a positive MRI of the spine in axial spondyloarthritis: a consensual approach by the ASAS/OMERACT MRI study group. Ann. Rheum. Dis. 71, 1278–1288 (2012).
Østergaard, M., Maksymowych, W. P., Pedersen, S. J., Chiowchanwisawakit, P. & Lambert, R. G. W. Structural lesions detected by magnetic resonance imaging in the spine of patients with spondyloarthritis – definitions, assessment system, and reference image set. J. Rheumatol. Suppl. 84, 18–34 (2009).
Weber, U. et al. Does spinal MRI add incremental diagnostic value to MRI of the sacroiliac joints alone in patients with non-radiographic axial spondyloarthritis? Ann. Rheum. Dis. 74, 985–992 (2015).
Weber, U. et al. Diagnostic utility of candidate definitions for a positive MRI of the spine in patients with axial spondyloarthritis. Arthritis Rheumatol. 67, 924–933 (2015).
Ez-Zaitouni, Z. et al. The yield of a positive MRI of the spine as imaging criterion in the ASAS classification criteria for axial spondyloarthritis: results from the SPACE and DESIR cohorts. Ann. Rheum. Dis. 76, 1731–1736 (2017).
de Bruin, F. et al. Prevalence of degenerative changes and overlap with spondyloarthritis-associated lesions in the spine of patients from the DESIR cohort. RMD Open. 4, e000657 (2018).
Weber, U. et al. Sensitivity and specificity of spinal inflammatory lesions assessed by whole-body magnetic resonance imaging in patients with ankylosing spondylitis or recent-onset inflammatory back pain. Arthritis Rheum. 61, 900–908 (2009).
Rennie, W. J. et al. Magnetic resonance imaging assessment of spinal inflammation in ankylosing spondylitis: standard clinical protocols may omit inflammatory lesions in thoracic vertebrae. Arthritis Rheum. 61, 1187–1193 (2009).
Blachier, M. et al. Does the site of magnetic resonance imaging abnormalities match the site of recent-onset inflammatory backpain? The DESIR cohort. Ann. Rheum. Dis. 72, 979–985 (2013).
de Hooge, M. et al. Is the site of back pain related to the location of magnetic resonance imaging lesions in patients with chronic back pain? Results from the Spondyloarthritis Caught Early Cohort. Arthritis Care Res. 69, 717–723 (2017).
Sepriano, A. et al. Performance of the ASAS classification criteria for axial and peripheral spondyloarthritis: a systematic literature review and meta-analysis. Ann. Rheum. Dis. 76, 886–890 (2017).
Sepriano, A. et al. Predictive validity of the ASAS classification criteria for axial and peripheral spondyloarthritis after follow-up in the ASAS cohort: a final analysis. Ann. Rheum. Dis. 75, 1034–1042 (2016).
Ez-Zaitouni, Z. et al. The influence of discrepant imaging judgements on the classification of axial spondyloarthritis is limited: a replication in the SpondyloArthritis Caught Early (SPACE) cohort. Ann. Rheum. Dis. 77, e1 (2018).
Bakker, P. A. Can we use structural lesions seen on MRI of the sacroiliac joints reliably for the classification of patients according to the ASAS axial spondyloarthritis criteria? Data from the DESIR cohort. Ann. Rheum. Dis. 76, 392–398 (2017).
Bakker, P. A. et al. Impact of replacing radiographic sacroiliitis by magnetic resonance imaging structural lesions on the classification of patients with axial spondyloarthritis. Rheumatology 57, 1186–1193 (2018).
Molnar, C. et al. TNF blockers inhibit spinal radiographic progression in ankylosing spondylitis by reducing disease activity: results from the Swiss Clinical Quality Management cohort. Ann. Rheum. Dis. 77, 63–69 (2018).
Ramiro, S. et al. Evolution of radiographic damage in ankylosing spondylitis: a 12-year prospective follow-up of the OASIS study. Ann. Rheum. Dis. 74, 52–59 (2015).
Ramiro, S. et al. Spinal radiographic progression in early axial spondyloarthritis: five-year results from the DESIR cohort. Arthritis Care Res. https://doi.org/10.1002/acr.23796 (2018).
Sieper, J. et al. Efficacy and safety of adalimumab in patients with non-radiographic axial spondyloarthritis: results of a randomised placebo-controlled trial (ABILITY-1). Ann. Rheum. Dis. 72, 815–822 (2013).
Dougados, M. et al. Symptomatic efficacy of etanercept and its effects on objective signs of inflammation in early nonradiographic axial spondyloarthritis: a multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheumatol. 66, 2091–2102 (2014).
Sieper, J. et al. A randomized, double-blind, placebo-controlled, sixteen-week study of subcutaneous golimumab in patients with active nonradiographic axial spondyloarthritis. Arthritis Rheumatol. 67, 2702–2712 (2015).
Molto, A., Paternotte, S., Claudepierre, P., Breban, M. & Dougados, M. Effectiveness of tumor necrosis factor α blockers in early axial spondyloarthritis: data from the DESIR cohort. Arthritis Rheumatol. 66, 1734–1744 (2014).
Baraliakos, X. et al. Non-radiographic axial spondyloarthritis patients without initial evidence of inflammation may develop objective inflammation over time. Rheumatology 56, 1162–1166 (2017).
Sengupta, R. et al. Short-term repeat magnetic resonance imaging scans in suspected early axial spondyloarthritis are clinically relevant only in HLA-B27-positive male subjects. J. Rheumatol. 45, 202–205 (2018).
Bennett, A. N. et al. Severity of baseline magnetic resonance imaging – evident sacroiliitis and HLA–B27 status in early inflammatory back pain predict radiographically evident ankylosing spondylitis at eight years. Arthritis Rheum. 58, 3413–3418 (2008).
Poddubnyy, D. et al. Rates and predictors of radiographic sacroiliitis progression over 2 years in patients with axial spondyloarthritis. Ann. Rheum. Dis. 70, 1369–1374 (2011).
Sepriano, A. et al. Is active sacroiliitis on MRI associated with radiographic damage in axial spondyloarthritis? Real-life data from the ASAS and DESIR cohorts. Rheumatology 58, 798–802 (2019).
Machado, P. M. et al. MRI vertebral corner inflammation followed by fat deposition is the strongest contributor to the development of new bone at the same vertebral corner: a multilevel longitudinal analysis in patients with ankylosing spondylitis. Ann. Rheum. Dis. 75, 1486–1493 (2016).
Baraliakos, X. et al. Which spinal lesions are associated with new bone formation in patients with ankylosing spondylitis treated with anti-TNF agents? A long-term observational study using MRI and conventional radiography. Ann. Rheum. Dis. 73, 1819–1825 (2014).
Maksymowych, W. P. et al. Fat metaplasia on MRI of the sacroiliac joints increases the propensity for disease progression in the spine of patients with spondyloarthritis. RMD Open 3, e000399 (2017).
Dougados, M. et al. Rate and predisposing factors for sacroiliac joint radiographic progression after a two-year follow-up period in recent-onset spondyloarthritis. Arthritis Rheumatol. 68, 1904–1913 (2016).
Sepriano, A. et al. Five-year follow-up of radiographic sacroiliitis: progression as well as improvement? Ann. Rheum. Dis. 75, 1262–1263 (2016).
Sepriano, A. et al. Percentage of progressors in imaging: can we ignore regressors? RMD Open 5, e000848 (2019).
Dougados, M. et al. Evaluation of the change in structural radiographic sacroiliac joint damage after 2 years of etanercept therapy (EMBARK trial) in comparison to a contemporary control cohort (DESIR cohort) in recent onset axial spondyloarthritis. Ann. Rheum. Dis. 77, 221–227 (2018).
Creemers, M. C. et al. Assessment of outcome in ankylosing spondylitis: an extended radiographic scoring system. Ann. Rheum. Dis. 64, 127–129 (2005).
van der Heijde, D. et al. Modified stoke ankylosing spondylitis spinal score as an outcome measure to assess the impact of treatment on structural progression in ankylosing spondylitis. Rheumatology 58, 388–400 (2019).
Ramiro, S. et al. Which scoring method depicts spinal radiographic damage in early axial spondyloarthritis best? Five-year results from the DESIR cohort. Rheumatology 57, 1991–2000 (2018).
Maksymowych, W. P. et al. Development and validation of the spondyloarthritis radiography module for calibration of readers using the modified Stoke Ankylosing Spondylitis Spine Score. Arthritis Care Res. 66, 55–62 (2014).
Baraliakos, X. et al. Inflammation in ankylosing spondylitis: a systematic description of the extent and frequency of acute spinal changes using magnetic resonance imaging. Ann. Rheum. Dis. 64, 730–734 (2005).
Maas, F. et al. Incorporating assessment of the cervical facet joints in the modified Stoke Ankylosing Spondylitis Spine Score is of additional value in the evaluation of spinal radiographic outcome in ankylosing spondylitis. Arthritis Res. Ther. 19, 77 (2017).
De Bruin, F. et al. Development of the CT Syndesmophyte Score (CTSS) in patients with ankylosing spondylitis: data from the SIAS cohort. Ann. Rheum. Dis. 77, 371–377 (2018).
Maksymowych, W. P. et al. Development and validation of web-based training modules for systematic evaluation of active inflammatory lesions in the spine and sacroiliac joints in spondyloarthritis. J. Rheumatol. 36, 48–57 (2009).
Deodhar, A. et al. A fifty-two-week randomized placebo-controlled trial of certolizumab pegol in non-radiographic axial spondyloarthritis. Arthritis Rheumatol. 71, 1101–1111 (2019).
Weiss, P. et al. Feasibility and reliability of the spondyloarthritis research consortium of canada sacroiliac joint inflammation score in children. Arthritis Res. Ther. 20, 56 (2018).
Van der Heijde, D. et al. Efficacy and safety of filgotinib, a selective Janus kinase 1 inhibitor, in patients with active ankylosing spondylitis (TORTUGA): results from a randomised, placebo-controlled, phase 2 trial. Lancet 392, 2378–2387 (2018).
Van der Heijde, D. et al. Ixekizumab, an interleukin-17A antagonist in the treatment of ankylosing spondylitis or radiographic axial spondyloarthritis in patients previously untreated with biological disease-modifying anti-rheumatic drugs (COAST-V): 16 week results of a phase 3 randomised, double-blind, active-controlled and placebo-controlled trial. Lancet 392, 2441–2451 (2018).
Van der Heijde, D. et al. Limited radiographic progression and sustained reductions in MRI inflammation in patients with axial spondyloarthritis: 4-year imaging outcomes from the RAPID-axSpA phase III randomised trial. Ann. Rheum. Dis. 77, 699–705 (2018).
Van der Heijde, D. et al. Tofacitinib in patients with ankylosing spondylitis: a phase II, 16-week, randomised, placebo-controlled, dose-ranging study. Ann. Rheum. Dis. 76, 1430–1437 (2017).
Panwar, J. et al. Spondyloarthritis Research Consortium of Canada scoring system for sacroiliitis in juvenile spondyloarthritis/enthesitis-related arthritis: a reliability, validity, and responsiveness study. J. Rheumatol. 46, 636–644 (2019).
Maksymowych, W. P. Clinical and MRI responses to etanercept in early non-radiographic axial spondyloarthritis: 48-week results from the EMBARK study. Ann. Rheum. Dis. 75, 1328–1335 (2016).
Maksymowych, M. et al. Validation of online calibration modules for the Spondyloarthritis Research Consortium of Canada MRI scores based on real-time experiential learning [abstract 586]. Arthritis Rheumatol. 69 (Suppl. 10), 833–834 (2017).
Weiss, P. et al. Feasibility and reliability of the spondyloarthritis research consortium of canada sacroiliac joint structural score in children. J. Rheumatol. 45, 1411–1417 (2018).
Maksymowych, W. P. et al. Validation of the Spondyloarthritis Research Consortium of Canada magnetic resonance imaging spinal inflammation index: is it necessary to score the entire spine? Arthritis Care Res. 57, 501–507 (2007).
Maksymowych, W. P. et al. Low-dose Infliximab (3 mg/kg) significantly reduces spinal inflammation on magnetic resonance imaging in patients with ankylosing spondylitis: a randomized placebo-controlled study. J. Rheumatol. 37, 1728–1734 (2010).
Krabbe, S. et al. Inflammatory and structural changes in vertebral bodies and posterior elements of the spine in axial spondyloarthritis: construct validity, responsiveness and discriminatory ability of the anatomy-based CANDEN scoring system in a randomised placebo-controlled trial. RMD Open 4, e000624 (2018).
Gezmis, E. et al. Diagnosis of early sacroiliitis in seronegative spondyloarthropathies by DWI and correlation of clinical and laboratory findings with ADC values. Eur. J. Radiol. 82, 2316–2321 (2013).
Vendhan, K. et al. A diffusion-based quantification technique for assessment of sacroiliitis in adolescents with enthesitis-related arthritis. Br. J. Radiol. 89, 20150775 (2016).
Beltran, L. S., Samim, M., Gyftopoulos, S., Bruno, M. T. & Petchprapa, C. N. Does the addition of DWI to fluid-sensitive conventional MRI of the sacroiliac joints improve the diagnosis of sacroiliitis? AJR Am. J. Roentgenol. 210, 1309–1316 (2018).
Boy, F. N. et al. The role of multi-parametric MR imaging in the detection of early inflammatory sacroiliitis according to ASAS criteria. Eur. J. Radiol. 83, 989–996 (2014).
Bray, T. J. et al. Diffusion-weighted imaging is a sensitive biomarker of response to biologic therapy in enthesitis-related arthritis. Rheumatology 56, 399–407 (2017).
Diekhoff, T. et al. Improved detection of erosions in the sacroiliac joints on MRI with volumetric interpolated breath-hold examination (VIBE): results from the SIMACT study. Ann. Rheum. Dis. 77, 1585–1589 (2018).
Idolazzi, L. et al. 18F-fluoride PET/CT for detection of axial involvement in ankylosing spondylitis: correlation with disease activity. Ann. Nucl. Med. 30, 430–434 (2016).
Lee, S. G. et al. Assessment of bone synthetic activity in inflammatory lesions and syndesmophytes in patients with ankylosing spondylitis: the potential role of 18F-fluoride positron emission tomography-magnetic resonance imaging. Clin. Exp. Rheumatol. 33, 90–97 (2015).
Bruijnen, S. T. G. et al. Bone formation in ankylosing spondylitis during anti-tumour necrosis factor therapy imaged by 18F-fluoride positron emission tomography. Rheumatology 57, 631–638 (2018).
Ouichka, R. et al. Performance of 18F-sodium fluoride positron emission tomography with computed tomography to assess inflammatory and structural sacroiliitis on magnetic resonance imaging in axial spondyloarthritis. Clin. Exp. Rheumatol. 37, 19–25 (2019).
Park, E. K. et al. Baseline increased 18Ffluoride uptake lesions at vertebral corners on positron emission tomography predict new syndesmophyte development in ankylosing spondylitis: a 2-year longitudinal study. Rheumatol. Int. 37, 765–773 (2017).
Buchbender, C. et al. Hybrid 18F-labeled fluoride positron emission tomography/magnetic resonance (MR) imaging of the sacroiliac joints and the spine in patients with axial spondyloarthritis: a pilot study exploring the link of MR bone pathologies and increased osteoblastic activity. J. Rheumatol. 42, 1631–1637 (2015).
Raynal, M. et al. Performance of 18F-sodium fluoride positron emission tomography with computed tomography to assess inflammatory and structural sacroiliitis on magnetic resonance imaging and computed tomography, respectively, in axial spondyloarthritis. Arthritis Res. Ther. 21, 119 (2019).
Bruijnen, S. T. G. et al. Bone formation in ankylosing spondylitis during anti-tumour necrosis factor therapy imaged by 18F-fluoride positron emission tomography. Rheumatology 57, 631–638 (2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
W.P.M. is Chief Medical Officer of CaRE Arthritis and has received research and/or educational grants from Abbvie, Janssen, Novartis, Pfizer and UCB, and consulting fees and/or honoraria from Abbvie, Boehringer, Celgene, Galapagos, Janssen, Lilly, Novartis, Pfizer and UCB.
Additional information
Peer review information
Nature Reviews Rheumatology thanks A. Deodhar and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
CaRE Arthritis imaging portal: http://www.carearthritis.com/MRI.php
Rights and permissions
About this article
Cite this article
Maksymowych, W.P. The role of imaging in the diagnosis and management of axial spondyloarthritis. Nat Rev Rheumatol 15, 657–672 (2019). https://doi.org/10.1038/s41584-019-0309-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-019-0309-4
This article is cited by
-
The bone marrow side of axial spondyloarthritis
Nature Reviews Rheumatology (2023)
-
The correlations between C-reactive protein and MRI-detected inflammation in patients with axial spondyloarthritis: a systematic review and meta-analysis
Clinical Rheumatology (2023)
-
A novel structure preserving generative adversarial network for CT to MR modality translation of spine
Neural Computing and Applications (2023)
-
Can radiomics replace the SPARCC scoring system in evaluating bone marrow edema of sacroiliac joints in patients with axial spondyloarthritis?
Clinical Rheumatology (2023)
-
Comparison of zero echo time MRI with T1-weighted fast spin echo for the recognition of sacroiliac joint structural lesions using CT as the reference standard
European Radiology (2022)