Abstract
Estrogens play a physiologically important role in the brain, but controversies exist regarding the association between Alzheimer’s disease (AD) and estrogens. Estrogen-related factors were comprehensively examined in frontal lobe tissues from autopsied AD patients, and compared with controls. Concentrations of estrogens, expression of estrogen receptors (ERs), and estrogen-metabolizing enzymes (EMEs) which are important for determining the peripheral estrogen concentrations, were examined using liquid chromatography tandem mass spectrometry, immunohistochemistry, and quantitative real-time PCR, respectively. Body mass index (BMI), known to correlate with the serum estrogen concentrations, was also taken into consideration. There were no significant differences in estrogen concentrations or each EME level between the two groups in both the cortex and white matter, whereas glial nuclear ER-β expression was significantly lower in white matter from the AD group than the control group (Allred score, 3.2 ± 0.3 and 6.5 ± 0.3, respectively. P < 0.0001). Estrogen concentrations were found to closely correlate with BMI, particularly in controls. ER-β loss in the white matter from the AD group suggests the necessity of studying the effects of estrogens on glias as well as neurons in the etiology of AD. The correlation between BMI and estrogen concentrations in the frontal lobe suggests the importance of non-brain sources of estrogens.
Similar content being viewed by others
Introduction
Estrogens play a physiologically important role in various organs, and deficiency/excess of estrogens has been reported to cause various diseases. In the nervous system, estrogens have been reported to maintain homeostasis by protecting neurons against damage, organizing sex-specific neuronal networks, or regulating bioenergetic systems1,2,3. Further, estrogens have been suggested to inhibit amyloid β (Aβ) deposition by favorably regulating β-secretase (BACE1) or neprilysin, which generate and degrade Aβ, respectively4,5,6,7. Epidemiologically, the risk of Alzheimer’s disease (AD) is higher in women than men, and its incidence in women increases after menopause8. Levels of estradiol (E2), a representative and the most potent estrogen, in postmenopausal women reportedly decrease to levels lower than those in men9. In some studies at the end of the 1990s, the risk of AD was reported to significantly decrease in women treated with estrogen replacement therapy10. These observations suggest a protective effect of estrogens against AD; however, in Women’s Health Initiative (WHI) hormone therapy trials, where the effect of external female hormone use was tested against a placebo, the risk of AD was reported to increase in women treated with hormone (estrogen-progesterone combined) replacement therapy, raising controversies in this study area11. Generally, in postmenopausal women, where estrogens from the ovary markedly decrease, peripheral estrogen synthesis via estrogen-metabolizing enzymes (EMEs) is important as the source of estrogens12. EMEs include aromatase, steroid sulfatase (STS), estrogen sulfotransferase (EST), and some types of 17β-hydroxysteroid dehydrogenase (HSD). Aromatase converts circulating androgens from the adrenal gland or ovary into estrogens. STS hydrolyzes biologically inactive estrogen sulfates to produce active estrogens. HSD type 1 (HSD-1) catalyzes the 17β-reduction of a biologically weak estrogen, estrone (E1), converting into E2. EST and HSD type 2 (HSD-2) play opposite roles to STS and HSD-1, respectively (Fig. 1)12. Examining EMEs as well as estrogen levels in the tissue of interest is important for studying the relationship between estrogens and diseases in postmenopausal women12. Further, examination of the expression of estrogen receptors (ERs) in the target tissues is necessary in this study area, because estrogens cannot exert their function in the absence of ERs even if estrogen concentrations are high enough8, 12. Although many studies have examined the association between estrogens and AD, few studies have systematically examined estrogen-related factors (e.g. estrogen concentrations, EMEs expression, or ERs expression) in the brain tissues of AD patients. We address this in the present study using the frontal lobe tissues (cortex and white matter, separately) from AD patients and controls. In AD, Aβ deposition or neurofibrillary tangles are typically found in the hippocampus or cerebral cortex, and these are the most frequently studied regions. The frontal lobe is the most suitable for the study of AD using frozen materials for the following reasons: the frontal cortex is frequently affected in AD; the volume of the frontal lobe is large, and separate sample collection from cortex and white matter is straightforward compared with the hippocampus or other cerebral lobes. The white matter, mainly composed of neuronal axons and glias, has been less frequently studied than the cortex in AD; however, we also focused on this, because glias play important roles in maintaining homeostasis of the brain, and increasing evidence has shown their importance in the etiology of AD13, 14. Concentrations of E1, E2, and androstenedione (Adione), a representative androgen, were examined in the frontal lobe (cortex and white matter, separately) from AD patients and controls, using liquid chromatography tandem mass spectrometry (LC-MS/MS), the most accurate method for measuring sex steroid concentrations. In addition, expressions of EMEs and two types of ERs, ER-α and ER-β (wild type, ER-β1), were examined by quantitative real-time PCR and immunohistochemistry, respectively. Further, the associations between body mass index (BMI) and steroid concentrations in the frontal lobe were examined, because aromatase is abundantly contained in the fatty tissue, and BMI is reportedly important as a determinant of serum estrogen levels in postmenopausal women15.
Estrogen-metabolizing enzymes. Aromatase, STS, and HSD-1 act in a direction producing more active estrogens, whereas EST and HSD-2 act in the opposite direction. Adione, androstenedione; E1, estrone; E1S, estrone sulfate; E2, estradiol; STS, steroid sulfatase; EST, estrone sulfotransferase; HSD-1, 17β-hydroxysteroid dehydrogenase type 1; HSD-2, 17β-hydroxysteroid dehydrogenase type 2.
Results
Sex steroids concentrations in the frontal lobe and their association with BMI
There was no significant difference in each sex steroid concentration between AD and control groups both in the cortex and white matter (Table 1). BMI in the AD group was lower than that in the control group although statistical significance was not achieved (16.1 vs. 19.3, P = 0.0752). The association between each sex steroid concentration and BMI is shown in Fig. 2. Concentrations of E1 and E2 correlated positively with BMI in both the cortex and white matter, with particularly high correlation coefficient in controls (Table 2). The correlation coefficient was lower in the AD group than in controls, with the slope among AD patients being larger for BMI ≥ 17.5 than for BMI < 17.5. Concentrations of E1 and E2 were significantly lower in subjects with BMI < 17.5 than in those with BMI ≥ 17.5 both in the cortex and white matter (Fig. 2 and Table 3). Adione concentration did not present such a clear association with BMI (Fig. 2, Table 2 and Table 3).
Expression of EMEs in the frontal lobe
Transcript levels of aromatase, STS, EST, and HSD-1 did not significantly differ between AD and control groups both in either the cortex or white matter. HSD-2 mRNA was undetectable in most samples (Table 4). There was no significant association between each EME mRNA level and any sex steroid concentration. There was no significant association between BMI and each EME mRNA level, either (data not shown).
Expression of ERs in the frontal lobe
The Allred score16,17,18 was adopted for assessing expression of ERs, because it is an objective and easy method to semiquantatively estimate ERs in a large area of target tissue. In the white matter, glial nuclear ER-β1 staining was clearly observed for most of the controls, but this was not true for AD patients (Fig. 3c,d). The Allred score for ER-β1 in the white matter was significantly higher in controls than in AD (6.5 ± 0.3 and 3.2 ± 0.3, respectively. P < 0.0001). As for ER-α, nuclear staining was not seen in the white matter irrespective of the antibodies used (Fig. 3g,h). In the cortex, the nuclear staining was insufficient to be evaluated irrespective of the ER type or disease status (Fig. 3a,b,e,f).
Discussion
Estrogen-related factors were systematically examined in the frontal lobe of AD and control groups. There was no significant difference in any of the sex steroid concentrations or expression levels of EMEs between the two groups in either the cortex or white matter, whereas nuclear ER-β1 expression was significantly lower in the white matter from the AD group than in that from controls.
Few studies have systematically examined estrogen-related factors in human brain tissue. Yue et al. reported a lower E2 concentration as well as lower aromatase mRNA level in the frontal cortex of AD patients compared with controls. Together with experimental evidence from animal models, they concluded that decrease of aromatase and consequent E2 reduction causes AD19. These observations by Yue et al. were not reproduced in the present study, although the reason is not known.
The cortex is a representative region affected in AD; however, in the present study, the only factor that was clearly different between the AD and control groups was the decrease of glial nuclear ER-β1 expression (considered to be estrogen-bound ER-β1, as described below) in the white matter from AD patients. White matter is an environment for neurons to interact with glias via neuronal axons. Glias are considered important regulators of neuronal survival/death, because they play important roles in trophic support or maintaining metabolic and ionic homeostasis in neurons, or maintaining synaptic transmission, etc. Recently, Ma et al. reported in vitro and in vivo observations suggesting that estrogens play a neuroprotective role against brain ischemia-reperfusion injury by activating glial cells via ER-β20. The participation of glias in the etiology of AD has also attracted attention recently13, 14. Furthermore, the importance of ER-β has been suggested in the prevention of Aβ accumulation and plaque formation, and the selective activation of ER-β has been suggested as a promising treatment for AD7, 8, 21. Taking our findings together with these reports, we assume that estrogen-bound ER-β in glias in the white matter exerts a neuroprotective action against AD development through glial-axonal interaction. Nuclear ER-β loss in glias, suggesting neuroprotective dysfunction, may lead to the neuronal loss observed in the AD cortex. The possibility that ER-β loss in glias is the result, not the cause, of AD cannot be ruled out at present. In vitro and in vivo studies taking not only neuronal cells but also glial cells into account are needed to clarify the importance of glial ER-β loss in the etiology of AD. Although there have been limited number of studies immunohistochemically examining ER (ER-α and/or ER-β) expression in the brain from AD patients, no consensus has been obtained thus far whether ERs decrease or increase in AD22,23,24,25,26,27. This is at least partly because of the diversity of the study settings. The brain is composed of many parts and various cell types having different functions. It is no wonder that the results are different among studies according to the parts or cell types examined. Results may also differ according to which of the intracellular structures are examined. Cytoplasmic staining has been estimated in most studies: however, we did not evaluate cytoplasm, because it was difficult to appropriately and differentially evaluate cytoplasm for the variety of cell types present in the frontal lobe. Although the importance of ERs in cytoplasm or cell membrane has recently been recognized, ERs are primarily nuclear receptors and nuclear staining is the first in line to be estimated8, 16,17,18. Like other nuclear receptors, ER localizes to the nucleus on binding with estrogens; therefore, nuclear ER expression suggests that the ER is estrogen-bound and activated/functional. A group from the Netherlands examined nuclear ER expression in AD patients’ brains22, 26, 27. Increased nuclear ER-α and ER-β expression was reported in the neurons of the nucleus basalis of Meynert in AD patients26. Higher ER-α nuclear expression in AD patients than in controls was reported in the infundibular nucleus of the hypothalamus22. To our knowledge, the present study is the first to have focused on nuclear ER expression in the white matter of the frontal lobe, and to have shown a decrease in ER-β expression in AD patients. Differences in immunohistochemical methods may also cause different results. We used a method established in clinical practice/study for breast cancer using robust antibodies16,17,18; however, most studies in the nervous system used other antibodies. In the present study, two kinds of antibodies were tried for ER-α, but neither yielded nuclear staining.
One of the most novel and important findings in the present study is that estrogen concentrations exhibited strongly positive correlation with BMI but not with aromatase mRNA level in the frontal tissue from controls. This suggests that non-brain sources are important as determinants of estrogen concentrations in the frontal lobe. BMI is known to positively correlate with serum estrogen levels in postmenopausal women, because aromatase is abundantly contained in the adipose tissue15. Estrogens and androgens have a steroidal structure and are highly lipophilic. Serum steroids may easily transfer and accumulate in the brain which is abundant in lipid, resulting in a close correlation between serum and frontal steroids concentrations, finally leading to a correlation between BMI and frontal estrogen concentrations. Interestingly, the correlation coefficient between BMI and frontal estrogen concentrations observed in the control group (0.714–0.815, Table 2) is higher than that reported for serum estrogens (0.52–0.60)15, suggesting a major effect of BMI on frontal estrogens. The lower correlation coefficient in AD than in the control group may reflect a rapid BMI decrease in AD patients, which has been reported in many studies28,29,30. The causal relationship between BMI and AD is not fully understood, but the following hypotheses have been suggested: eating disorders in AD (or pre-AD) patients may cause BMI decrease, or decreased BMI resulting in estrogen reduction may cause AD. In the present study, BMI 17.5 seems to be somewhat of a threshold for maintaining estrogen concentrations in the frontal lobe. This association between AD and BMI decrease may lead to a vicious cycle for the maintenance of brain homeostasis mediated by estrogens.
The concentrations of E1 and Adione in the frontal lobe observed in the present study were several times higher, while that for E2 was several score times higher, than those in the postmenopausal serum reported in other studies31. The higher concentration of E1 and Adione in the frontal tissue than the serum can be mostly ascribed to their lipophilia, but this is unlikely for E2′s remarkably higher concentration. In the present analyses of EMEs, HSD-2 was undetectable in most cases, while HSD-1 was detectable for almost all cases. The relative dominance of HSD-1 over HSD-2 would accelerate the conversion of E1 into E2, leading to marked condensation of E2 in the frontal tissue (Fig. 1)12. Such a high level of estrogens concentrations in the frontal lobe, together with distinct nuclear ER-β1 expression in glias among controls, may add further evidence for the physiologically important role of the estrogen signaling system in the brain. A compensatory mechanism, or lack thereof, to impairment of the estrogen signaling system may determine the risk of AD3.
In conclusion, in a systematic analysis examining estrogen-related factors for the cortex and white matter of the frontal lobe from AD and control groups, the only remarkable difference between the two groups was a marked decrease of glial nuclear ER-β1 expression in the white matter from the AD group, suggesting the necessity of studying the effect of estrogens on glias as well as neurons in the etiology of AD. Frontal estrogen concentrations were closely correlated with BMI, suggesting that extremely low BMI is not favorable for maintaining estrogen concentrations in the brain.
Materials and Methods
Patients
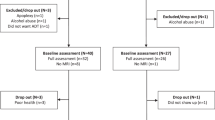
Brain tissues (convex of the second frontal gyrus) were obtained from 13 pathologically confirmed Alzheimer’s disease patients and 12 age adjusted controls. All of them were Japanese women autopsied at the Tokyo Metropolitan Geriatric Hospital between 1999 and 2006. The study protocol was approved by the ethics committee of Tokyo Metropolitan Geriatric Hospital and Fujita Health University. All methods were performed in accordance with the relevant guidelines and regulations. Written informed consent, including consent to use in medical studies, was obtained from the bereaved family of each patient prior to autopsy.
Quantitative Analysis by LC-MS/MS
Concentrations of E1, E2 and Adione were measured using LC-MS/MS at ASKA Pharma Medical Co., Ltd (Kawasaki, Japan). Briefly, weighed brain tissues (40–200 mg) were homogenized, and each internal standard (100 pg of each internal standard, E1-13C4, E2-13C4, and Adione-d7) and ethanol were added, and then shaken at 50 °C for 2 hrs. The obtained extracts were applied to a Bond C18 cartridge column (Varian, Harbor City, CA), and the steroid fraction was eluted with 80% acetonitrile. The steroids fraction was loaded onto a mixed-mode cartridge (Oasis MAX, Waters, Milford, MA) to separate the neutral and phenol fractions containing androgens and estrogens, respectively. Estrogens were measured by LC-MS/MS, an API-5000 triple stage quadrupole mass spectrometer (Appied Biosystems, Foster City, CA) connected to an LC-20AD pump and SI HTC autosampler (Shimadzu, Kyoto, Japan), and electrospray ionization ion source devices, using the column, a Xterra MS C18 (2.1 mm × 100 mm I.D.3.5 μm; Waters) at 40 °C. Adione was measured by an LC-MS/MS instrument, API-4000 (Appied Biosystems) equipped with an ESI ion source and an Agilent 1100 HPLC system (Agilent Technologies, Santa Clara, CA) with an HTC PAL auto-sampler (CTC Analytical, Zwingen, Switzerland), using the column, Cadenza CD-C18 (150 mm × 3 mm I.D., 3 μm; Imtakt, Kyoto, Japan) at 40 °C.
Determination of EMEs mRNA level
Frozen brain tissues (10–50 mg) were smashed to fine powders by Micro Smash MS-100R (TOMY SEIKO Co., Tokyo, Japan) with stainless beads, SUB-55 and zirconia beads, ZB-10 (TOMY SEIKO Co.) at 4 °C and extracted with 1 ml of TRIzol Reagent (Invitrogen, Carlsbad, CA) to prepare total RNA according to manufacturers’ instructions. The cDNA was synthesized from the total RNA by SuperScript II reverse transcriptase (Invitrogen) using random hexamers (Applied Biosystems). The resulting cDNA was subjected to quantitative PCR reactions using TaqMan probes (Applied Biosystems). Transcript levels of EMEs were determined using the following TaqMan Gene Expression Assays (TaqMan probes): Hs00903413_m1 (aromatase), Hs00165853_m1 (STS), Hs00193690 (EST), Hs00166219_m1 (HSD-1), Hs00157993_m1 (HSD-2), and Hs99999903_m1 (β-actin). The fluorescence generated by cleavage of the TaqMan probes during PCR was analyzed by a 7300 Real Time PCR system (Applied Biosystems). Briefly, a total of 20 μl reaction mixture consisting of 10 μl TaqMan Gene Expression Master Mix (Applied Biosystems), 1 μl Primer/TaqMan probe, and the cDNA was amplified by PCR (40 cycles of 95 °C for 10 sec and 60 °C for 30 sec). The level of each EME transcript was calculated from the respective PCR cycle threshold (Ct) value by ΔΔCt method.
Immunohistochemical examination of ERs status
Immunostaining was performed for sections of formalin-fixed and paraffin-embedded tissue according to the methods used for breast cancer samples as described elsewhere18. ER-α and ER-β1 were detected by anti-ER-α mouse monoclonal antibodies (clone 1D5; Dako, Glostrup, Denmark and clone 6F11; Novocastra, Newcastle upon Tyne, England) and anti-ER-β1 mouse monoclonal antibody (clone PPG5/10; Dako) specific for ER-β1, respectively16,17,18. As there is no standard method for assessing ER expression in the nervous system, the Allred score routinely used in clinical practice for breast cancer was adopted for evaluation. Briefly, nuclear immunoreactivity for ER-α and ER-β1 is estimated independently by summing the percentage score, PS, and intensity score, IS, of positively stained cells (PS: 0%, 0; <1%, 1; <10%, 2; <33%, 3; <67%, 4; ≥67%, 5. IS: weak, 1; medium, 2; strong, 3)17.
Statistical analysis
The t-test was used to compare concentrations of each steroid hormone, BMI, mRNA level of each EME, or Allred score for each ER, between AD and control groups. In all instances, the statistical software JMP 12.0.1 (SAS Institute, Cary, NC) was used. P < 0.05, when necessary dividing by the number of factors examined (Bonferroni adjustment), was considered significant.
References
Arevalo, M. A., Azcoitia, I. & Garcia-Segura, L. The neuroprotective actions of oestradiol and oestrogen receptors. Nat. Rev. Neurosci. 16, 17–29 (2015).
Harada, N., Wakatsuki, T., Aste, N., Yoshimura, N. & Honda, S. I. Functional analysis of neurosteroidal oestrogen using gene-disrupted and transgenic mice. J. Neuroendocrinol 21, 365–369 (2009).
Rettberg, J. R., Yao, J. & Brinton, R. D. Estrogen: a master regulator of bioenergetic systems in the brain and body. Front. Neuroendocrinol. 35, 8–30 (2014).
Agostinho, P., Pliássova, A., Oliveira, C. R. & Cunha, R. A. Localization and Trafficking of Amyloid-β Protein Precursor and Secretases: Impact on Alzheimer’s Disease. J. Alzheimer’s Dis. 45, 329–347 (2015).
Hüll, M., Berger, M. & Heneka, M. Disease-modifying therapies in Alzheimer’s disease: how far have we come? Drugs 66, 2075–2093 (2006).
Li, R. et al. Brain endogenous estrogen levels determine responses to estrogen replacement therapy via regulation of BACE1 and NEP in female Alzheimer’s transgenic mice. Mol. Neurobiol. 47, 857–867 (2013).
Nilsson, S., Koehler, K. F. & Gustafsson, J. Development of subtype-selective oestrogen receptor-based therapeutics. Nat Rev Drug Discov 10, 778–792 (2011).
Zhao, L., Woody, S. K. & Chhibber, A. Estrogen receptor beta in Alzheimer’s disease: From mechanisms to therapeutics. Ageing Res. Rev. 24, 178–190 (2015).
Khosla, S., Melton, L. J., Atkinson, E. J. & O’Fallon, W. M. Relationship of serum sex steroid levels to longitudinal changes in bone density in young versus elderly men. J. Clin. Endocrinol. Metab. 86, 3555–3561 (2001).
Tang, M. X. et al. Effect of oestrogen during menopause on risk and age at onset of Alzheimer’s disease. Lancet 348, 429–432 (1996).
Shumaker, S. A. et al. Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA 289, 2651–2662 (2003).
Honma, N., Hosoi, T., Arai, T. & Takubo, K. Estrogen and cancers of the colorectum, breast, and lung in postmenopausal women. Pathol. Int. 65, 451–459 (2015).
Dhandapani, K. M. & Brann, D. W. Role of astrocytes in estrogen-mediated neuroprotection. Exp. Gerontol. 42, 70–75 (2007).
Struble, R. G., Nathan, B. P., Cady, C., Cheng, X. & McAsey, M. Estradiol regulation of astroglia and apolipoprotein E: an important role in neuronal regeneration. Exp. Gerontol. 42, 54–63 (2007).
Mahabir, S. et al. Usefulness of body mass index as a sufficient adiposity measurement for sex hormone concentration associations in postmenopausal women. Cancer Epidemiol. Biomarkers Prev. 15, 2502–2507 (2006).
Hammond, M. E. et al. American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J. Clin. Oncol. 28, 2784–2795 (2010).
Harvey, J. M., Clark, G. M., Osborne, C. K. & Allred, D. C. Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J. Clin. Oncol. 17, 1474–1481 (1999).
Honma, N. et al. Clinical importance of estrogen receptor-beta evaluation in breast cancer patients treated with adjuvant tamoxifen therapy. J. Clin. Oncol. 26, 3727–3734 (2008).
Yue, X. et al. Brain estrogen deficiency accelerates Abeta plaque formation in an Alzheimer’s disease animal model. Proc. Natl. Acad. Sci. USA 102, 19198–19203 (2005).
Ma, Y. et al. Estrogen replacement therapy-induced neuroprotection against brain ischemia-reperfusion injury involves the activation of astrocytes via estrogen receptor beta. Sci. Rep. 6, 21467 (2016).
Zhao, L. et al. 17beta-Estradiol regulates insulin-degrading enzyme expression via an ERbeta/PI3-K pathway in hippocampus: relevance to Alzheimer’s prevention. Neurobiol. Aging 32, 1949–1963 (2011).
Hestiantoro, A. & Swaab, D. F. Changes in estrogen receptor-alpha and -beta in the infundibular nucleus of the human hypothalamus are related to the occurrence of Alzheimer’s disease neuropathology. J. Clin. Endocrinol. Metab. 89, 1912–1925 (2004).
Hong-Goka, B. & Chang, F. L. Estrogen receptors alpha and beta in choroid plexus epithelial cells in Alzheimer’s disease. Neurosci. Lett. 360, 113–116 (2004).
Lu, Y. P., Zeng, M., Swaab, D. F., Ravid, R. & Zhou, J. N. Colocalization and alteration of estrogen receptor-alpha and -beta in the hippocampus in Alzheimer’s disease. Hum. Pathol. 35, 275–280 (2004).
Savaskan, E., Olivieri, G., Meier, F., Ravid, R. & Müller-Spahn, F. Hippocampal estrogen beta-receptor immunoreactivity is increased in Alzheimer’s disease. Brain Res. 908, 113–119 (2001).
Ishunina, T. A. & Swaab, D. F. Increased expression of estrogen receptor alpha and beta in the nucleus basalis of Meynert in Alzheimer’s disease. Neurobiol. Aging 22, 417–426 (2001).
Ishunina, T. A., van Heerikhuize, J. J., Ravid, R. & Swaab, D. F. Estrogen receptors and metabolic activity in the human tuberomamillary nucleus: changes in relation to sex, aging and Alzheimer’s disease. Brain Res. 988, 84–96 (2003).
Buchman, A. S. et al. Change in body mass index and risk of incident Alzheimer disease. Neurology 65, 892–897 (2005).
Qizilbash, N. et al. BMI and risk of dementia in two million people over two decades: a retrospective cohort study. Lancet Diabetes Endocrinol 3, 431–436 (2015).
Buchman, A. S., Schneider, J. A., Wilson, R. S., Bienias, J. L. & Bennett, D. A. Body mass index in older persons is associated with Alzheimer disease pathology. Neurology 67, 1949–1954 (2006).
Honma, N. et al. Sex steroid hormones in pairs of tumor and serum from breast cancer patients and pathobiological role of androstene-3β, 17β-diol. Cancer Sci 102, 1848–1854 (2011).
Acknowledgements
This work was supported by MEXT/JSPS KAKENHI Grant Number 19590376, 25461398, and JP 16H06277; the Japanese Foundation for Multidisciplinary Treatment of Cancer; and the Mitsui Sumitomo Insurance Welfare Foundation. We thank Dr. Tomio Arai and all the staff of Department of Pathology, Tokyo Metropolitan Geriatric Hospital and Department of Neuropathology, Tokyo Metropolitan Institute of Gerontology for their great help in pathological works. We appreciate the kind help of Dr. Shigeaki Kato, from the Research Institute of Innovative Medicine Jyoban Hospital of the Tokiwakai Group, with the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
N.Ho., S.S., and N.Ha. designed the study, Y.S. and S.Mu. collected samples and performed pathological diagnosis, N.Ho., M.T., N.Y., and N.Ha. acquired data, N.Ho. and N.Ha. wrote the main manuscript text, N.Ho., S.S. and S.Mo., S.Mu., and N.Ha. acquired research grant. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Honma, N., Saji, S., Mikami, T. et al. Estrogen-Related Factors in the Frontal Lobe of Alzheimer’s Disease Patients and Importance of Body Mass Index. Sci Rep 7, 726 (2017). https://doi.org/10.1038/s41598-017-00815-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-00815-3
This article is cited by
-
Estrogen concentration and estrogen receptor-β expression in postmenopausal colon cancer considering patient/tumor background
Journal of Cancer Research and Clinical Oncology (2022)
-
Estrogen receptor beta (ESR2) gene polymorphism and susceptibility to dementia
Acta Neurologica Belgica (2021)
-
Estrogenic hormones receptors in Alzheimer’s disease
Molecular Biology Reports (2021)
-
Estrogen Signaling in Alzheimer’s Disease: Molecular Insights and Therapeutic Targets for Alzheimer’s Dementia
Molecular Neurobiology (2020)
-
Characteristics of adverse events of endocrine therapies among older patients with breast cancer
Supportive Care in Cancer (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.