Summary
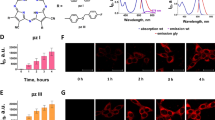
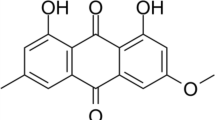
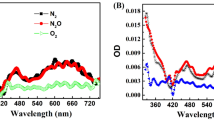
The mechanism of cell death induction by dimethyl tetrahydroxyhelianthrone (DTHe), a new second-generation photodynamic sensitizer, is analysed in human leukaemic cell lines in comparison with the structurally related hypericin. DTHe has a broad range of light spectrum absorption that enables effective utilization of polychromatic light. Photosensitization of HL-60 cells with low doses of DTHe (0.65 μM DTHe and 7.2 J cm–2 light energy) induced rapid apoptosis of ≥90% of the cells. At doses ≥2 μM, dying cells assumed morphological necrosis with perinucleolar condensation of chromatin in HL-60 and K-562 cell lines. Although nuclear fragmentation that is characteristic to apoptosis was prevented, DNA digestion to oligonucleosomes proceeded unhindered. Such incomplete apoptosis was more prevalent with the related analogue hypericin throughout most doses of photosensitization. Despite hypericin being a stronger photosensitizer, DTHe exhibited advantageous phototoxic properties to tumour cells, initiating apoptosis at concentrations about threefold lower than hypericin. Photosensitization of the cells induced dissociation of the nuclear envelope, releasing lamins into the cytosol. DTHe also differed from hypericin in effects exerted on the nuclear lamina, causing release of an 86-kDa lamin protein into the cytosol that was unique to DTHe. Within the nucleus, nuclear envelope lamin B underwent covalent polymerization, which did not affect apoptotic nuclear fragmentation at low doses of DTHe. At higher doses, polymerization may have been extensive enough to prevent nuclear collapse. Hut-78, CD4+ cells were resistant to the photodynamically activated apoptotic pathway. Beyond the tolerated levels of photodynamic damage, these cells died exclusively via necrosis. Hut-78 cells overexpress Bcl-XL as well as a truncated Bcl-XLtr isoform that could contribute to the observed resistance to apoptosis.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Agostinis, P., Vandenbogaerde, A., Donella-Deana, A., Pinna, L. A., Lee, K., Goris, J., Merlevede, W., Vandenheede, J. R. & de Witte, P. (1995). Photosensitized inhibition of growth factor-regulated protein kinases by hypericin. Biochem Pharmacol 49: 1615–1622.
Agostinis, P., Donella-Deana, A., Cuveele, J., Vandenbogaerde, A., Sarno, S. & Merlevede, W. (1996). A comparative analysis of the photosensitized inhibition of growth-factor regulated protein kinases by hypericin derivatives. Biochem Biophys Res Commun 220: 613–617.
Anker, L., Gopalakrishna, R., Jones, K. D., Antel, J. P., Apuzzo, M. L. J. & Couldwell, W. T. (1995). Hypericin in adjuvant brain tumor therapy. Drugs Future 20: 511–517.
Boise, L. H., Gonzalez-Garcia, M., Postema, C. E., Ding, L., Lindsten, T., Turka, L. A., Mao, X., Nunez, G. & Thompson, C. B. (1993). Bcl-X, a bcl-2-related gene that functions as a dominant regulator of apoptotic cell death. Cell 74: 597–608.
Carpenter, S. & Kraus, G. A. (1991). Photosensitization is required for inactivation of equine infectious anemia virus by hypericin. Photochem Photobiol 53: 169–174.
Chattopadhyay, S. V., Kumar, C. V. & Das, P. K. (1984). Laser flash photolytic determination of triplet yields via singlet oxygen generation. J Photochem 24: 1–9.
Chung, P. S., Saxton, R. E., Paiva, M. B., Rhee, C. K., Soudant, J., Mathey, A., Foote, C. & Castro, D. J. (1994). Hypericin uptake in rabbits and nude mice transplanted with human squamous cell carcinoma: study of a new sensitizer for laser phototherapy. Laryngoscope 104: 1471–1476.
Degar, S., Prince, A. M., Pascual, D., Lavie, G., Levin, B., Mazur, Y., Lavie, D., Ehrlich, L. S., Carter, C. & Meruelo, D. (1992). Inactivation of the human immunodeficiency virus by hypericin: Evidence for photochemical alterations of p24 and a block in uncoating. AIDS Res and Human Retroviruses 8: 1929–1936.
Degar, S., Lavie, G. & Meruelo, D. (1993). Photodynamic inactivation of Radiation Leukemia virus produced from hypericin treated cells. Virology 197: 796–800.
Diwu, Z. (1995). Novel therapeutic and diagnostic applications of hypocrellins and hypericin. Photochem Photobiol 61: 529–539.
Diwu, Z. & Lown, J. W. (1990). Hypocrellins and their use in photosensitization. Photochem Photobiol 52: 606–616.
Diwu, Z. & Lown, J. W. (1993). Photosensitization with anticancer agents 17. EPR studies of photodynamic action of hypericin: formation of semiquinone radical and activated oxygen species on illumination. Free Radical Biol Med 14: 209–215.
Dougherty, T. J. (1983). Hematoporphyrin as a photosensitizer of tumors. Photochem Photobiol 38: 377–379.
Evans, S., Matthews, W., Perry, R., Flaker, D., Norton, J. & Pass, H. I. (1990). Effect of photodynamic therapy on tumor necrosis factor production by murine macrophages. J Natl Cancer Inst 82: 34–39.
Gomer, C. I. (1991). Preclinical examination of first and second generation photosensitizers used in photodynamic therapy. Photochem Photobiol 61: 529–539.
Granville, D. J., Levy, J. G. & Hunt, D. W. C. (1997). Photodynamic therapy induces caspase-3 activation in HL-60 cells. Cell Death Differ 4: 623–626.
Grossweiner, L. I. (1994). Photodynamic therapy. In The Science of Phototherapy, Chapter 8. CRC Press: Boca Raton, FL
Hadjur, C., Jeunet, A. & Jardon, P. (1994). Photosensitization by hypericin: ESR evidence for singlet oxygen and superoxide anion radicals formation in an in vitro model. J Photochem Photobiol B Biol 26: 67–74.
Hadjur, C., Richard, M. J., Parat, M. O., Favier, A. & Jardon, P. (1995). Photodynamically induced cytotoxicity of hypericin dye on human fibroblast cell line MRC5. J Photochem Photobiol 27: 139–146.
Honigsmann, H., Tanew, A. & Wolff, K. (1987). Treatment of mycosis fungoides with PUVA. Photo Dermatol 4: 55–58.
Hudson, J. B., Harris, L. & Towers, G. H. N. (1993). The importance of light in the anti-HIV effect of hypericin. Antiviral Res 20: 173–178.
Jones, L. R. & Grossweiner, L. I. (1996). Effect of Photofrin R on in vivo skin reflectivity. J Photochem Photobiol B33: 153–156.
Kessel, D. (1984). Hematoporphyrin and HPD, photophysics, photochemistry and phototherapy. Photochem Photobiol 39: 851–859.
Kick, G., Messer, G., Goetz, A., Plewig, G. & Kind, P. (1995). Photodynamic therapy induces expression of interleukin 6 by activation of AP-1 but not NF-κB DNA binding. Cancer Res 55: 2373–2379.
Kick, G., Messer, G., Plewig, G., Kind, P. & Goetz, A. (1996). Strong and prolonged induction of c-jun and c-fos proto-oncogenes by photodynamic therapy. Br J Cancer 74: 30–36.
Kluck, R. M., Bossy-Wetzel, E., Green, D. R. & Newmeyer, D. D. (1997). The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275: 1132–1136.
Lavie, G., Valentine, F., Levin, B., Mazur, Y., Gallo, G., Lavie, D., Weiner, D. & Meruelo, D. (1989). Studies of the mechanisms of action of the antiretroviral agents hypericin and pseudohypericin. Proc Natl Acad Sci USA 86: 5963–5967.
Lavie, D., Freeman, D., Bock, H., Fleischer, J., van Kranenburg, K., Ittah, Y., Mazur, Y., Lavie, G., Liebes, L. & Meruelo, D. (1990). Hypericin, a potential anti-AIDS drug. In Trends in Medicinal Chemistry ‘90. Proceedings of the XIth International Symposium on Medicinal Chemistry, 2–7 September, Jerusalem, Israel, pp. 321–327.
Li, X., Traganos, F. & Darzynkiewicz, Z. (1994). Simultaneous analysis of DNA replication and apoptosis during treatment of HL-60 cells with camptothecin and hyperthermia and mitogen stimulation of human lymphocytes. Cancer Res 54: 4289–4293.
Lotem, J. & Sachs, L. (1995). Regulation of bcl-2, bcl-x and bax in the control of apoptosis by hematopoietic cytokines and dexamethasone. Cell Growth Difference 6: 647–653.
Lotem, J., Cragoe, E. J. & Sachs, L. (1991). Rescue from programmed cell death in leukemic and normal myeloid cells. Blood 78: 953–960.
McCaughan, Jr J. S. (1984). Photoradiation of malignant tumors presensitized with hematoporphyrin derivative. Prog Clin Biol Res 170: 805–827.
Meruelo, D., Lavie, G. & Lavie, D. (1988). Therapeutic agents with dramatic antiretroviral activity and little toxicity at effective doses: aromatic polycyclic diones hypericin and pseudohypericin. Proc Natl Acad Sci USA 85: 5230–5234.
Miller, G. G., Brown, K. & Ballangrud, A. M. (1997). Preclinical assessment of hypocrellins and hypocrellin B derivatives as sensitizers for photodynamic therapy of cancer: progress update. Photochem Photobiol 65: 714–722.
Mossman, T. (1983). Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytoxicity assays. J Immunogenet 21: 235
Murray, N. R., Burns, D. J. & Fields, A. P. (1994). Presence of a β|| protein kinase C-selective nuclear membrane activation factor in human leukemia cells. J Biol Chem 269: 21385–21390.
Noodt, B. B., Berg, K., Stokke, T., Peng, Q. & Nesland, J. M. (1996). Apoptosis and necrosis induced with light and 5-aminolaevulinic acid-derived protoporphyrin IX. Br J Cancer 74: 22–29.
Orenstein, A., Kostenich, G., Roitman, L., Tsur, H., Katanik, D., Kopolovic, J., Ehrenberg, B. & Malik, Z. (1996). Photodynamic therapy of malignant lesions of the skin mediated by topical application of 5-aminolevulinic acid in combination with DMSO and EDTA. Lasers Life Sci 7: 1–9.
Pace, N. (1942). The etiology of hypericism, a photosensitivity produced by St. Johns wort. Am J Physiol 136: 650–656.
Peter, M., Nakagawa, J., Doree, M., Labbe, J. C. & Nigg, E. A. (1990). In vitro disassembly of the nuclear lamina and M phase-specific phosphorylation of lamins by cdc2 kinase. Cell 61: 591–602.
Senthil, V., Longworth JW Ghiron, C. A. & Grossweiner, L. I. (1992). Photosensitization of aqueous model systems by hypericin. Biochim Biophys Acta 1115: 192–200.
Takahashi, I., Nakanishi, S., Kobayashi, E., Nakano, H., Suzuki, K. & Tanaoki, T. (1989). Hypericin and pseudohypericin specifically inhibit protein kinase C: possible relation to their antiretroviral activity. Biochem. Biophys. Res Commun 165: 1207–1212.
Takahashi, A., Alnemri, E. S., Lazebnik, Y. A., Fernandes-Alnemri, T., Litwack, G., Moir, R. D., Goldman, R. D., Poilier, G. G., Kaufmann, S. H. & Earnshaw, W. C. (1996). Cleavage of lamin A by Mch2α but not CPP32: Multiple interleukin 1β-converting enzyme-related proteases with distinct substrate specificities. Proc Nat Acad Sci USA 93: 8395–8400.
Tang, J., Colacino, J. M., Larsen, S. H. & Spitzer, W. (1990). Virucidal activity of hypericin against enveloped and non-enveloped DNA and RNA viruses. Antiviral Res 13: 313–326.
Thomas, C., MacGill, R. S., Miller, G. C. & Pardini, R. S. (1992a). Photoactivation of hypericin generates singlet oxygen in mitochondria and inhibits succinoxidase. Photochem Photobiol 55: 47–53.
Thomas, C. & Pardini, R. S. (1992b). Oxygen dependence of hypericin-induced phototoxicity to EMT6 mouse mammary carcinoma cells. Photochem Photobiol 55: 831–837.
Weiner, L. & Mazur, Y. (1992). EPR studies of hypericin. Photogeneration of free radicals and superoxide. J Chem Soc Perkin Trans 2: 1439–1442.
Yang, J., Liu, X., Bhalla, K., Kim, C. N., Ibrado, A. M., Cai, J., Peng, T. I., Jones, D. P. & Wang, X. (1997). Prevention of apoptosis by Bcl-2: Release of cytochrome c from mitochondria blocked. Science 275: 1129–1132.
Zhang, W., Lawa, R. E., Hintona, D. R., Su, Y. & Cauldwell, W. T. (1995). Growth inhibition and apoptosis in human neuroblastoma SK-N-SH cells induced by hypericin, a potent inhibitor of protein kinase C. Cancer Lett 96: 31–35.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Lavie, G., Kaplinsky, C., Toren, A. et al. A photodynamic pathway to apoptosis and necrosis induced by dimethyl tetrahydroxyhelianthrone and hypericin in leukaemic cells: possible relevance to photodynamic therapy. Br J Cancer 79, 423–432 (1999). https://doi.org/10.1038/sj.bjc.6690066
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690066
Keywords
This article is cited by
-
Photodithazine photodynamic effect on viability of 9L/lacZ gliosarcoma cell line
Lasers in Medical Science (2017)
-
Hypericin-photodynamic therapy leads to interleukin-6 secretion by HepG2 cells and their apoptosis via recruitment of BH3 interacting-domain death agonist and caspases
Cell Death & Disease (2013)
-
Impact of photodynamic inactivation (PDI) using the photosensitizer chlorin e6 on viability, apoptosis, and proliferation of human keratocytes in vitro
Graefe's Archive for Clinical and Experimental Ophthalmology (2013)
-
Effect and mechanism of 5-aminolevulinic acid-mediated photodynamic therapy in esophageal cancer
Lasers in Medical Science (2011)
-
p38 MAPK plays an essential role in apoptosis induced by photoactivation of a novel ethylene glycol porphyrin derivative
Oncogene (2008)