Summary
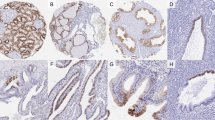
The expression of laminin and fibronectin isoforms varies with cellular maturation and differentiation and these differences may well influence cellular processes such as adhesion and motility. The basement membrane (BM) of fetal oral squamous epithelium contains the laminin chains, α2, α3, α5, β1, β2, β3, γ1 and γ2. The BM of adult normal oral squamous epithelium comprises the laminin chains, α3, α5, β1, β3, γ1 and γ2. A re-expression of the laminin α2 and β2 chains could be shown in adult hyperproliferative, dysplastic and carcinomatous lesions. In dysplasia and oral squamous cell carcinoma (OSCC), multifocal breaks of the BM are present as indicated by laminin chain antibodies. These breaks correlate to malignancy grade in their extent. Moreover, in the invasion front the α3 and γ2 chain of laminin-5 can immunohistochemically be found outside the BM within the cytoplasm of budding carcinoma cells and in the adjacent stroma. The correlation between the morphological pattern of invasive tumour clusters and a laminin-5 immunostaining in the adjacent stroma may suggest, first, that a laminin-5 deposition outside the BM is an immunohistochemical marker for invasion and second, that OSCC invasion is guided by the laminin-5 matrix. Expression of oncofetal fibronectins (IIICS de novo glycosylated fibronectin and ED-B fibronectin) could be demonstrated throughout the stromal compartment. However, the ED-B fibronectin synthesizing cells (RNA/RNA in situ hybridization) are confined to small stroma areas and to single stroma and inflammatory cells in the invasion front. A correlation of the number of ED-B fibronectin synthesizing cells to malignancy grade could not be seen. ED-B fibronectin mRNA-positive cells seem to be concentrated in areas of fibrous stroma recruitment with a linear alignment of stromal fibro-/myofibroblasts (desmoplasia). Double staining experiments (ED-B fibronectin in situ hybridization and α-smooth muscle actin immunohistochemistry) indicated that the stroma myofibroblasts are a preferential source of ED-B fibronectin. In conclusion, in OSCC, a fetal extracellular matrix conversion is demonstrable. Tumour cells (laminin α2 and β2 chain) and recruited stromal myofibroblasts (oncofetal ED-B fibronectin) contribute to the fetal extracellular matrix milieu.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Barnes, JL, Torres, E, Mitchell, RJ & Peters, JH (1995). Expression of alternatively spliced fibronectin variants during remodelling in proliferative glomerulonephritis. Am J Pathol 147: 1361–1371.
Berndt, A, Hyckel, P, Könnecker, A, Katenkamp, D & Kosmehl, H (1997). Oral squamous cell carcinoma invasion is associated with a laminin-5 matrix re-organisation but independent of basement membrane and hemidesmosome formation. Clues from an in vitro invasion model. Inv Metast 17: 251–258.
Berndt, A, Kosmehl, H, Mandel, U, Gabler, U, Luo, X, Celeda, D, Zardi, L & Katenkamp, D (1995). TGFβ and bFGF synthesis and localization in Dupuytren’s disease (nodular palmar fibromatosis) relative to cellular activity, myofibroblast phenotype and oncofetal variants of fibronectin. Histochem J 27: 1041–1020.
Borsi, L, Carnemolla, B, Castellani, P, Rosellini, C, Vecchio, D, Allemani, G, Chang, SE, Taylor-Papadimitriou, Pande, H & Zardi, L (1987). Monoclonal antibodies in the analysis of fibronectin isoforms generated by alternative splicing of mRNA precursors in normal and transformed human cells. J Cell Biol 104: 595–600.
Borsi, L, Castellani, P, Risso, AM, Leprini, A & Zardi, L (1990). Transforming growth factor-β regulates the splicing pattern of fibronectin messenger RNA precursor. FEBS 261: 175–178.
Brouty-Boye, D & Magnien, V (1994). Myofibroblast and concurrent ED-B fibronectin phenotype in human stromal cells cultured from non-malignant and malignant breast tissue. Eur J Cancer 30A: 66–73.
Bryne, M, Koppang, HS, Lilleng, R & Kjaerheim, A (1992). Malignancy grading of the deep invasive margins of oral squamous cell carcinoma has high prognostic value. J Pathol 166: 375–381.
Burton-Wurster, N, Lust, G & Macleod, JN (1997). Cartilage fibronectin isoforms: in search of functions for a special population of matrix glycoproteins. Matrix Biol 15: 441–454.
Carnemolla, B, Balza, E, Siri, A, Zardi, L, Nicotra, MR, Bigotti, A & Natali, PG (1989). A tumor-associated fibronectin isoform generated by alternative splicing of messenger RNA precursors. J Cell Biol 108: 1139–1148.
Carnemolla, B, Neri, D, Castellani, P, Leprini, A, Neri, G, Pini, A, Winter, G & Zardi, L (1996). Phage antibodies with pan-species recognition of the oncofetal angiogenesis marker fibronectin ED-B domain. Int J Cancer 68: 397–405.
Castellani, P, Viale, G, Dorcaratto, A, Nicolo, G, Kaczmarek, J, Querze, G & Zardi, L (1994). The fibronectin isoform containing the ED-B oncofetal domain: a marker of angiogenesis. Int J Cancer 59: 612–618.
Chen, W & Culp, LA (1996). Adhesion mediated by fibronectin’s alternatively spliced Edb (EIIIB) and its neighbouring type III repeats. Exp Cell Res 223: 9–19.
Downer, CS, Watt, FM & Speight, PM (1993). Loss of α6 and β4 integrin subunits coincides with loss of basement membrane components in oral squamous cell carcinomas. J Pathol 171: 183–190.
Durkin, ME, Loechel, F, Mattei, MG, Gilpin, BJ, Albrechtsen, R & Wewer, UM (1997). Tissue-specific expression of the human laminin alpha5-chain, and mapping of the gene to human chromosome 20q13.2–13.3 and to distal mouse chromosome 2 near the locus for the ragged (Ra) mutation. FEBS Lett 411: 296–300.
Ekblom, M, Klein, G, Mugrauer, G, Fecker, L, Deutzmann, R, Timpl, R & Ekblom, P (1990). Transient and locally restricted expression of laminin A chain mRNA by developing epithelial cells during kidney organogenesis. Cell 60: 337–346.
Engvall, E, Earwicker, D, Haaparanta, T, Ruoslahti, E & Sanes, JR (1990). Distribution and isolation of four laminin variants; tissue restricted distribution of heterotrimers assembled from five different subunits. Cell Regul 1: 731–740.
Harada, T, Shinohara, M, Nakamura, S & Oka, M (1994). An immunohistochemical study of the extracellular matrix in oral squamous cell carcinoma and its association with invasive and metastatic potential. Virchows Arch 424: 257–266.
Hashimoto-Uoshima, M, Yan, YZ, Schneider, G & Aukhil, I (1997). The alternative spliced domains EIIIB and EIIIA of human fibronectin affect cell adhesion and spreading. J Cell Sci 110: 2271–2280.
Hori, H, Kanamori, T, Mizuta, T, Yamaguchi, N, Liu, Y & Nagai, Y (1994). Human laminin M chain: epitope analysis of its monoclonal antibodies by immunoscreening of cDNA clones and tissue expression. J Biochem 116: 1212–1219.
Hruban, RH, Kuhajda, FP & Mann, RB (1987). Acute myelofibrosis. Immunohistochemical study of four cases and comparison with acute megakaryocytic leukemia. Am J Clin Pathol 88: 578–588.
Hsi, BL & Yeh, C-JG (1986). Monoclonal antibodies to human amnion. J Reprod Immunol 9: 11–21.
Iivanainen, A, Vuolteenaho, R, Sainio, K, Eddy, R, Shows, TB, Sariola, H & Tryggvason, K (1994). The human laminin β2 chain (s-laminin): structure, expression in fetal tissues and chromosomal assignment of the LAMB2 gene. Matrix Biol 14: 489–497.
Kaczmarek, J, Castellani, P, Nicolo, G, Spina, B, Allemanni, G & Zardi, L (1994). Distribution of oncofetal fibronectin isoforms in normal, hyperplastic and neoplastic human breast tissues. Int J Cancer 58: 11–16.
Kainulainen, T, Autio-Harmainen, H, Oikarinen, A, Salo, S, Tryggvason, K & Salo, T (1997). Altered distribution and synthesis of laminin-5 (Kalinin) in oral lichen planus, epithelial dysplasias and squamous cell carcinomas. Br J Dermatol 136: 331–336.
Kikkawa, Y, Sanzen, N & Sekiguchi, K (1998). Isolation and characterization of laminin-10/11 secreted by human lung carcinoma cells. Laminin-10/11 mediates cell adhesion through integrin alpha3 beta1. J Biol Chem 273: 15854–15859.
Klein, G, Ekblom, M, Fecker, L, Timpl, R & Ekblom, P (1990). Differential expression of laminin A and B chains during development of embryonic mouse organs. Development 110: 823–837.
Kobayashi, Y, Nakajima, T & Saku, T (1995). Loss of basement membranes in the invading front of O-1N, hamster squamous cell carcinoma with high potential of lymph node metastasis: An immunohistochemical study for laminin and type IV collagen. Pathol Int 45: 327–334.
Korang, K, Christiano, AM, Uitto, J & Mauviel, A (1995). Differential cytokine modulation of the genes LAMA3, LAMB3, and LAMC2, encoding the constitutive polypeptides, α3, β3, and γ2, of human laminin 5 in epidermal keratinocytes. FEBS Lett 368: 556–558.
Kornblihtt, AR, Pesce, CG, Alonso, CR, Cramer, P, Srebrow, A, Werbajh, S & Muro, AF (1996). The fibronectin gene as a model for splicing and transcription studies. FASEB J 10: 248–257.
Kosmehl, H, Berndt, A, Katenkamp, D, Hyckel, P, Stiller, KD, Gabler, U, Langbein, L & Reh, T (1995a). Integrin receptors and their relationship to cellular proliferation and differentiation of oral squamous cell carcinoma. A quantitative immunohistochemical study. J Oral Pathol Med 24: 343–348.
Kosmehl, H, Berndt, A, Katenkamp, D, Mandel, U, Bohle, R, Gabler, U & Celeda, D (1995b). Differential expression of fibronectin splice variants, oncofetal glycosylated fibronectin and laminin isoforms in nodular palmar fibromatosis. Path Res Pract 191: 1105–1113.
Kosmehl, H, Berndt, A & Katenkamp, D (1996). Molecular variants of fibronectin and laminin: structure, physiological occurrence and histopathological aspects. Virchows Arch 429: 311–322.
Leivo, I & Engvall, E (1988). Merosin, a protein specific for basement membranes of Schwann cells, striated muscle, and trophoblast, is expressed late in nerve and muscle development. Proc Natl Acad Sci USA 85: 1544–1548.
Loridon-Rosa, B, Vielh, P, Matsuura, H, Clausen, H, Cuadrado, C & Burtin, P (1990). Distribution of oncofetal fibronectin in human mammary tumors: immunofluorescence study on histological sections. Cancer Res 50: 1608–1612.
Mandel, U, Therkildsen, MH, Reibel, J, Sweeney, Matsuura, H, Hakamori, S-I, Dabelsteen, E & Clausenn, H (1992). Cancer-associated changes in glxcosylation of fibronectin. Immunohistological localization of oncofetal fibronectin defined by monoclonal antibodies. APMIS 100: 817–826.
Mandel, U, Gaggero, B, Reibel, J, Therkildsen, MH, Dabelsteen, E & Clausen, H (1994). Oncofetal fibronectins in oral carcinomas: correlation of two different types. APMIS 102: 695–702.
Marinkovich, PM, Lunstrum, GP, Keene, DR & Burgeson, RE (1992). The dermal-epidermal junction of human skin contains a novel laminin variant. J Cell Biol 119: 695–703.
Matsuura, H & Hakamori, S (1985). The oncofetal domain of fibronectin defined by monoclonal antibody FDC-6: its presence in fibronectins from fetal and tumor tissues and its absence in those from normal adult tissues and plasma. Proc Natl Acad Sci USA 82: 6517–6521.
Miner, JH & Sanes, JR (1994). Collagen IV α3, α4 and α5 chains in rodent basal laminae: sequence, distribution, association with laminins, and development switches. J Cell Biol 127: 879–891.
Miner, JH, Patton, BL, Lentz, SI, Gilbert, DJ, Snider, WD, Jenkins, NA, Copeland, NG & Sanes, J (1997). The laminin α chains: expression, developmental transitions, and chromosomal locations of α1–5, identification of heterotrimeric laminins 8–11, and cloning of a novel α3 isoform. J Cell Bio 137: 685–701.
Nickeleit, V, Zagachin, L, Nishikawa, K, Peters, JH, Hynes, RO & Colvin, RB (1995). Embryonic fibronectin isoforms are synthesised in crescents in experimental autoimmune glomerulonephritis. Am J Pathol 147: 965–978.
Odermatt, BF, Lang, AB, Rüttner, JR, Winterhalter, KH & Trüeb, B (1984). Monoclonal antibodies to human type IV collagen: useful reagents to demonstrate the heterotrimeric nature of the molecule. Proc Natl Acad Sci USA 81: 7343–7347.
O’Toole, EA, Marinkovich, MP, Hoeffler, WK, Furthmayr, H & Woodley, DT (1997). Laminin-5 inhibits human keratinocyte migration. Exp Cell Res 233: 330–339.
Pujuguet, P, Hammann, A, Moutet, M, Samuel, JL, Martin, F & Martin, M (1996). Expression of fibronectin ED-A+ and ED-B+ isoforms by human and experimental colorectal cancer. Am J Pathol 148: 579–592.
Pyke, C, Roemer, J, Kallunki, P, Lund, LR, Ralfkiaer, E, Danoe, K & Tryggvason, K (1994). The γ2 chain of kalinin/laminin-5 is preferentially expressed in invading malignant cells in human cancers. Am J Pathol 145: 782–791.
Rousselle, P, Lunstrum, GP, Keene, DR & Burgeson, RE (1991). Kalinin: an epithelium-specific basement membrane adhesion molecule that is a component of anchoring filaments. J Cell Biol 114: 567–576.
Rousselle, P, Golbik, R, van-der-Rest, M & Aumailley, M (1995). Structural requirement for cell adhesion to kalinin (laminin-5). J Biol Chem 270: 13766–13770.
Ryan, M, Christiano, AM, Engvall, E, Wewer, U, Miner, JH, Sanes, JR & Burgeson, RE (1996). The functions of laminins: lessons from in vivo studies. Matrix Biol 15: 369–381.
Sanes, JR & Chiu, AY (1983). The basal lamina of the neuromuscular junction. Cold Spring Harbor Symp Quant Biol 48: 776–678.
Skalli, O, Ropraz, P, Trzeciak, A, Benzonana, G, Gillesen, D & Gabbiani, G (1986). A monoclonal antibody against alpha-smooth muscle actin: a new probe for smooth muscle differentiation. J Cell Biol 103: 2787–2796.
Sollberg, S, Peltonen, J & Uitto, J (1992). Differential expression of laminin isoforms and β4 integrin epitopes in the basement membrane zone of normal human skin and basal cell carcinomas. J Invest Dermatol 98: 864–970.
Sordat, I, Bosman, FT, Dorta, G, Rousselle, P, Aberdam, D, Blum, AL & Sordat, B (1998). Differential expression of laminin-5 subunits and integrin receptors in human colorectal neoplasia. J Pathol 185: 44–52.
Squarzoni, S, Villanova, M, Sabatelli, P, Malandrini, A, Toti, P, Pini, A, Merlini, L, Guazzi, GC & Maraldi, NM (1997). Intracellular detection of the laminin α2 chain in skin by electron microscopy immunocytochemistry: comparison between normal and laminin α2 chain deficient subjects. Neuromusc Disord 7: 91–98.
Strassburger, S, Berndt, A, Hyckel, P, Katenkamp, D & Kosmehl, H (1998). Differential expression of laminin chains in the human major salivary gland. Histochem J 30: 81–88.
Tani, T, Lumme, A, Linnala, A, Kivilaakso, E, Kiviluoto, T, Burgeson, RE, Kangas, L, Leivo, I & Virtanen, I (1997). Pancreatic carcinomas deposit laminin-5, preferably adhere to laminin-5, and migrate on the newly deposited basement membrane. Am J Pathol 151: 1289–1302.
Thorup, AK, Dabelsteen, E, Schou, S, Gil, SG, Carter, WG & Reibel, J (1997). Differential expression of integrins and laminin-5 in normal oral epithelia. APMIS 105: 519–530.
Tiger, CF, Champliaud, MF, Pedrosa-Domellof, F, Thornell, LE, Ekblom, P & Gullberg, D (1997). Presence of laminin alpha 5 chain and lack of laminin alpha 1 chain during human muscle development and in muscular dystrophies. J Biol Chem 272, (45): 28590–28595.
Ueno, S, Osugi, Y, Nishimura, A, Shinoda, Y, Mushimoto, K & Shirasu, R (1997). Ultrastructural localization of collagen type IV and laminin expression in the epithelial basement membrane of oral carcinomas. Med Electron Microsc 30: 37–42.
Uriel, J (1979). Retrodifferentiation and the fetal patterns of the gene expression in cancer. Adv Canc Res 29: 127–174.
Wayner, EA, Gil, SG, Murphy, GF, Mark, SW & Carter, WG (1993). Epiligrin, a component of epithelial basement membranes is an adhesive ligand for α3β1 positive T lymphocytes. J Cell Biol 121: 1141–1152.
Wewer, U, Albrechtsen, R, Manthorpe, M, Varon, S, Engvall, E & Ruoslahti, E (1983). Human laminin isolated in a nearly intact, biologically active form from placenta by limited proteolysis. J Biol Chem 258: 12654–12660.
Wewer, U, Wayner, EA, Hoffstrom, BG, Lan, F, Mayer-Nielsen, B, Engvall, E & Albrechtsen, R (1994). Selective assembly of laminin variants by human carcinoma cells. Lab Invest 71: 719–730.
Williamson, RA, Henry, MD, Daniels, KJ, Hrstka, RF, Lee, JC, Sunada, Y, Ibraghimov-Beskrovnaya, O & Campbell, KP (1997). Dystroglycan is essential for early embryonic development: disruption of Reichert’s membrane in Dag-1 null mice. Hum Mol Genet 6: 831–841.
Xia, P & Culp, LA (1995). Adhesion activity in fibronectin’s alternatively spliced domain Eda (EIIIA): complementary to plasma fibronectin functions. Exp Cell Res 217: 517–527.
Zardi, L, Carnemolla, B, Siri, A, Santi, L & Accolla, RS (1980). Somatic cell hybrids producing antibodies specific to human fibronectin. Int J Cancer 25: 325–329.
Zhang, K & Kramer, RH (1996). Laminin-5 deposition promotes keratinocyte motility. Exp Cell Res 227: 309–322.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Kosmehl, H., Berndt, A., Strassburger, S. et al. Distribution of laminin and fibronectin isoforms in oral mucosa and oral squamous cell carcinoma. Br J Cancer 81, 1071–1079 (1999). https://doi.org/10.1038/sj.bjc.6690809
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690809
Keywords
This article is cited by
-
EMID2 is a novel biotherapeutic for aggressive cancers identified by in vivo screening
Journal of Experimental & Clinical Cancer Research (2024)
-
Genomic landscape and clonal architecture of mouse oral squamous cell carcinomas dictate tumour ecology
Nature Communications (2020)
-
Regulation of FN1 degradation by the p62/SQSTM1-dependent autophagy–lysosome pathway in HNSCC
International Journal of Oral Science (2020)
-
Expression of fibronectin in esophageal squamous cell carcinoma and its role in migration
BMC Cancer (2018)
-
Fibronectin-guided migration of carcinoma collectives
Nature Communications (2017)