Abstract
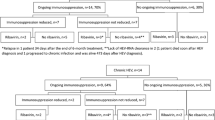
Fourty-four patients who underwent allogeneic bone marrow transplantation (alloBMT) were studied for hepatitis B virus (HBV)-related complications. The mean follow-up period was 15.3 months. Positivity for HBV surface antigen (HBsAg) was observed in 10 patients (22.7%) throughout the study. Four of the 10 patients were HBsAg carriers before alloBMT, while the remaining six became HBsAg(+) after alloBMT. During the follow-up period (from 6 months to 45 months), an elevation in serum ALT activity was observed in the four carriers when immunosuppression was reduced or withdrawn. All of the four HBsAg carriers developed hepatitis, but none of them died of liver failure due to HBV. Only one death due to GVHD and diabetic ketoacidosis was observed in this group. Two of the four carriers received marrow from anti-HBs positive donors and one of them cleared HBsAg from his serum via adoptive immunity 8 months after transplantation. The remaining six patients acquired HBV after alloBMT, but we were unable to demonstrate the source of HBV. Five of them had a moderate increase in serum ALT activity while the other patient had a normal ALT. Two patients seroconverted to anti-HBs spontaneously. Two patients died during the follow-up, one due to intracranial hemorrhage and the other due to GVHD and accompanying pulmonary infection. The rest of the study group (34 patients) remained HBsAg(−) throughout the study. Two of them had an HBsAg(+) donor, but neither developed HBV infection in their follow-up period. The acquisition rate of HBV infection was relatively low in recipients who were positive for anti-HBs compared to those who were negative for anti-HBs (8 vs 19%). Anti-HBs positivity remained for a longer period in recipients who received marrow from anti-HBs positive donors compared to those recipients who had anti-HBs negative donors (median 12 vs 3 months). We think that HBV is a frequent cause of liver dysfunction in alloBMT patients where HBV infection is endemic. Whether the disease is in the form of reactivation of HBsAg-positive recipients, or is acquired from unknown sources in recipients who never had contact with the virus, the course of the disease is not fatal. Silent serologic changes can be demonstrated if viral serologic markers are sought serially. Among them, the disappearance of serum anti-HBs may be important as it increases the risk of HBV contamination in recipients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Üstün, C., Koç, H., Karayalçın, S. et al. Hepatitis B virus infection in allogeneic bone marrow transplantation. Bone Marrow Transplant 20, 289–296 (1997). https://doi.org/10.1038/sj.bmt.1700885
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1700885
Keywords
This article is cited by
-
Clinical Implications and Management of Chronic Occult Hepatitis B Virus Infection
Current Hepatology Reports (2017)
-
Occult HBV infection in the oncohematological setting
Infection (2016)
-
Safety of hematopoietic stem cell transplantation from hepatitis B core antibodies-positive donors with low/undetectable viremia in HBV-naïve children
European Journal of Clinical Microbiology & Infectious Diseases (2014)
-
Hepatitis B reactivation after chemotherapy: two decades of clinical research
Hepatology International (2008)
-
Infections among allogeneic bone marrow transplant recipients in India
Bone Marrow Transplantation (2004)