Abstract
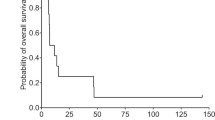
To improve the effectiveness of herpes simplex virus (HSV) thymidine kinase/ganciclovir (HSV-tk/GCV) suicide gene therapy, the replication-defective HSV vector TOIκB expressing both HSV-TK and a mutant form of the NF-κB inhibitor IκBα (IκBαM) was developed. TOIκB was constructed by recombining the IκBαM gene into the UL41 locus of a replication-defective lacZ expression vector, TOZ.1. Expression of IκBαM was confirmed by Western blotting, and the ability of the mutant protein to inhibit NF-κB nuclear translocation was examined by electrophoretic mobility shift assay. In human glioblastoma U-87MG cells, the p50/p50 dimer of NF-κB was already translocated to the nucleus without receptor-dependent signaling by TNF-α. Following infection with TOIκB, nuclear translocation of NF-κB in U-87MG cells was significantly inhibited and caspase-3 activity increased compared with TOZ.1-infected cells. The cytotoxicity of TOIκB for U-87MG cells was investigated by colorimetric MTT assay. At an MOI of 3, TOIκB infection killed 85% of the cells compared to 20% killed by TOZ.1 infection. In the presence of GCV, these numbers increased to 95–100% for TOIκB and 80–85% for TOZ.1. TOIκB neurotoxicity measured on cultured murine neurons was relatively low and similar to that of TOZ.1. The survival of nude mice implanted into the brain with U-87MG tumor cells was markedly prolonged by intratumoral TOIκB injection and GCV administration. Survival of TOIκB+GCV group was significantly longer (P<.02, Wilcoxon test) than for the control groups (TOZ.1 or TOIκB only, PBS or PBS+GCV). These results suggest that IκBαM expression may be a safe enhancement of replication-defective HSV-based suicide gene therapy in vitro and in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Dirks P, Bernstein M, Muler PJ, et al. The value of reoperation for recurrent glioblastoma. Can J Surg. 1993;36: 271–275.
Culver KM, Ram Z, Walbridge S, et al. In vivo gene transfer with retroviral vector-producing cells for treatment of experimental brain tumors. Science. 1992;256:1550–1552.
Barba D, Hardin J, Ray J, et al. Thymidine kinase-mediated killing of rat brain tumors. J Neurosurg. 1993;79:729–735.
Barba D, Hardin J, Sadelain M, et al. Development of antitumor immunity for following thymidine kinase-mediated killing of experimental brain tumors. Proc Natl Acad Sci USA. 1994;91:4348–4352.
Ram Z, Culver KW, Oshiro EM, et al. Therapy of malignant brain tumors by intratumoral implantation of retroviral vector-producing cells. Nat Med. 1997;12:1354–1361.
Okada H, Miyamura K, Itoh T, et al. Gene therapy against an experimental glioma using adeno-associated virus vectors. Gene Ther. 1996;3:957–964.
Chen SH, Shine HD, Goodman JC, et al. Gene therapy for brain tumors: regression of experimental gliomas by adenovirus-mediated gene transfer in vivo. Proc Natl Acad Sci USA. 1994;91:3054–3057.
Perez-Cruet MJ, Trask TW, Chen SH, et al. Adenovirus-mediated gene therapy of experimental gliomas. J Neurosci Res. 1994;39:506–511.
Vincent AJ, Vogels R, Someren GV, et al. Herpes simplex virus thymidine kinase gene therapy for rat malignant brain tumors. Hum Gene Ther. 1996;7:197–205.
Vincent AJ, Esandi MC, Avezaat CJ, et al. Preclinical testing of recombinant adenoviral herpes simplex virus-thymidine kinase gene therapy for central nervous system malignancies. Neurosurgery. 1997;41:442–452.
Boviatsis EJ, Park JS, Sena-Esteves M, et al. Long-term survival of rats harboring brain neoplasms treated with ganciclovir and a herpes simplex virus vector that retains an intact thymidine kinase gene. Cancer Res. 1994;54:5745–5751.
Moriuchi S, Oligino T, Krisky D, et al. Enhanced tumor cell killing in the presence of ganciclovir by herpes simplex virus type 1 vector-directed coexpression of human tumor necrosis factor-alpha and herpes simplex virus thymidine kinase. Cancer Res. 1998;58:5731–5737.
Beg AA, Baltimore D . An essential role for NF-κB in preventing TNF-α-induced cell death. Science. 1996;274: 782–784.
Wang CY, Mayo MW, Baldwin Jr AS . TNF- and cancer therapy-induced apoptosis: potentiation by inhibition of NF-κB. Science. 1996;274:784–787.
Antwerp DJV, Martin SJ, Kafri T, et al. Supression of TNF-α-induced apoptosis by NF-κB. Science. 1996;274: 787–789.
Baeuerle PA . The inducible transcription activator NF-κB: regulation by distinct protein subunits. Biochem Biophys Acta. 1991;1072:63–80.
Blank V, Lourilsky P, Israel A . NF-κB and related proteins: Rel/dorsal homologies meet ankyrin-like repeats. Trends Biochem Sci. 1992;17:135–140.
Wu M, Lee H, Bellas RE, et al. Inhibition of NF-κB/Rel induces apoptosis of murine B cells. EMBO J. 1996;15: 4682–4690.
Rayet B, Gelinas C . Aberrant rel/nfkb genes and activity in human cancer. Oncogene. 1999;18:6938–6947.
Krisky DM, Marconi PC, Oligino T, et al. Rapid method for construction of recombinant HSV gene transfer vectors. Gene Ther. 1997;4:1120–1125.
Marconi P, Krisky D, Oligino T, et al. Replication-defective herpes simplex virus vectors for gene transfer in vivo. Proc Natl Acad Sci USA. 1996;93:11319–11320.
Samaniego LA, Neiderhiser L, DeLuca NA . Persistence and expression of the herpes simplex virus genome in the absence of immediate-early proteins. J Virol. 1998;72: 3307–3320.
Preston CM, Mabbs R, Nicholl MJ . Construction and characterization of herpes simplex virus type 1 mutants with conditional defects in immediate early gene expression. Virology. 1997;229:228–239.
Dignam JD, Lebovitz RM, Roeder RG . Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 1983;11:1475–1489.
Urban MB, Baeuerle PA . The 65-kD subunit of NF-kB is a receptor for IkB and a modulator of DNA-binding specificity. Genes Dev. 1990;4:1975–1984.
Mosmann T . Rapid calorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55–63.
Brewer GJ, Torricelli JR, Evege EK, et al. Optimized survival of hippocampal neurons in B27-supplemented Neurobasal, a new serum-free medium combination. J Neurosci Res. 1993;35:567–576.
Lelong I H, Petegnief V, Rebel G . Neuronal cells mature faster on polyethyleneimine coated plates than on polylysine coated plates. J Neurosci Res. 1992;32:562–568.
Kashiwagi Y, Nakamura Y, Miyamae Y, et al. Pulse exposure of cultured rat neurons to aluminium-maltol affected the axonal transport system. Neurosci Lett. 1998;252:5–8.
Rainov NG . A phase III clinical evaluation of herpes simplex virus type 1 thymidine kinase and ganciclovir gene therapy as an adjuvant to surgical resection and radiation in adults with previously untreated glioblastoma multiforme. Human Gene Ther. 2000;11:2389–2401.
Wang C, Cusack JC, Liu R, et al. Control of inducible chemoresistance; enhanced anti-tumor therapy through increased apoptosis by inhibition of NF-κB. Nat Med. 1999;5:412–417.
Samejima Y, Meruelo D . ‘Bystander killing’ induces apoptosis and is inhibited by forskolin. Gene Ther. 1995;2: 50–58.
Hobbs WE, DeLuca NA . Perturbation of cell cycle progression and cellular gene expression as a function of herpes simplex virus ICP0. J Virol. 1999;73:8245–8255.
Beltinger C, Fulda S, Kammertoens T, et al. Herpes simplex virus thymidine kinase/ganciclovir-induced apoptosis involves ligand-independent death receptor aggregation and activation of caspases. Proc Natl Acad Sci USA. 1999;96: 8699–8704.
Marconi P, Tamura M, Moriuchi S, et al. Connexin 43-enhanced suicide gene therapy using herpesviral vectors. Mol Ther. 2000;1:71–81.
Nasu Y, Bangma CH, Hull GW, et al. Combination gene therapy with adenoviral vector-mediated HSV-tk+GCV and IL-12 in an orthotopic mouse model for prostate cancer. Prostate Cancer Prostatic Dis. 2001;4:44–55.
Niranjan A, Moriuchi S, Lunsford D, et al. Effective treatment of experimental glioblastoma by HSV vector-mediated TNFα and HSV-tk gene transfer in combination with radiosurgery and ganciclovir administration. Mol Ther. 2000;2:114–120.
Jarosinski KW, Whitney LW, Massa PT . Specific deficiency in nuclear factor-κB activation in neurons of the central nervous system. Lab Invest. 2001;81:1275–1288.
Chen XP, Li J, Mata M, et al. Herpes simplex virus type 1 ICP0 protein does not accumulate in the nucleus of primary neurons in culture. J Virol. 2000;74:10132–10141.
Acknowledgements
This work was supported by NIH Grant GM34534 to JCG, by GenVec, Inc. (Rockville, MD), and by a Grant-in-Aid for Scientific Research to SM (No. 12671357) from the Ministry of Education, Science and Culture, Ministry of Health and Human Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moriuchi, S., Glorioso, J., Maruno, M. et al. Combination gene therapy for glioblastoma involving herpes simplex virus vector-mediated codelivery of mutant IκBα and HSV thymidine kinase. Cancer Gene Ther 12, 487–496 (2005). https://doi.org/10.1038/sj.cgt.7700816
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700816
Keywords
This article is cited by
-
Synergistic effects of eukaryotic coexpression plasmid carrying LKB1 and FUS1 genes on lung cancer in vitro and in vivo
Journal of Cancer Research and Clinical Oncology (2014)
-
Evaluation and optimization of the administration of a selectively replicating herpes simplex viral vector to the brain by convection-enhanced delivery
Cancer Gene Therapy (2011)
-
An NF-κB p65-cIAP2 link is necessary for mediating resistance to TNF-α induced cell death in gliomas
Journal of Neuro-Oncology (2011)
-
Side populations of glioblastoma cells are less sensitive to HSV-TK/GCV suicide gene therapy system than the non-side population
In Vitro Cellular & Developmental Biology - Animal (2010)
-
Herpes simplex virus 1 (HSV-1) for cancer treatment
Cancer Gene Therapy (2006)