Abstract
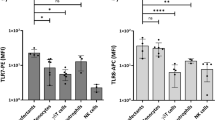
Weak immunogenicity of chronic lymphocytic leukemia (CLL) cells may contribute to disease progression and inhibit effective immunotherapy. Accordingly, agents that enhance the immunogenicity of CLL cells may be useful in immunotherapeutic approaches to this disease. Since Toll-like receptors (TLRs) are major regulators of innate immunity and initiation of adaptive immunity, we studied the effects of viral pathogen associated molecular pattern agonists (that are recognized by TLRs) on the costimulatory phenotype and function of CLL cells. CLL cells (especially those with high endogenous expression of CD38) responded to TLR7-activating imidazoquinolines and guanosine analogs by increasing costimulatory molecule expression, producing inflammatory cytokines, and becoming more sensitive to killing by cytotoxic effectors. Additional activation of protein kinase C pathways increased the ability to stimulate T-cell proliferation, blocked phosphorylation of the transcription factor, signal transducer and activator of transcription (STAT)3, and resulted in the acquisition of a dendritic cell surface phenotype by TLR7-activated CLL cells. Normal B cells also responded to TLR7 activation by increasing costimulatory molecule expression and cytokine production. These findings suggest a potential role for TLR7 agonists in CLL immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Spaner DE, Hammond C, Mena J, Foden C, Deabreu A . A phase I/II trial of oxidized autologous tumor vaccines during the ‘watch and wait’ phase of chronic lymphocytic leukemia. Cancer Immunol Immunother 2005; 54: 635–646.
Zinkernagel RM . Immunity against solid tumors? Int J Cancer 2001; 93: 1–5.
Spaner D . Amplifying cancer vaccine responses by modifying pathogenic gene programs in tumor cells. J Leukoc Biol 2004; 76: 338–351.
Kawai T, Akira S . Pathogen recognition with Toll-like receptors. Curr Opin Immunol 2005; 17: 338–344.
Decker T, Schneller F, Sparwasser T, Tretter T, Lipford GB, Wagner H et al. Immunostimulatory CpG-oligonucleotides cause proliferation, cytokine production, and an immunogenic phenotype in CLL B cells. Blood 2000; 95: 999–1006.
Hemmi H, Kaisho T, Takeuchi O, Sato S, Tomizawa H, Takeda K et al. Small anti-viral compounds activate immune cells via the TLR7-signaling pathway. Nat Immunol 2002; 3: 196–200.
Lee J, Chuang TH, Redecke V, She L, Pitha PM, Carson DA et al. Molecular basis for the immunostimulatory activity of guanine nucleoside analogs: activation of TLR7. Proc Natl Acad Sci USA 2003; 100: 6646–6651.
Shanafelt TD, Geyer SM, Kay NE . Prognosis at diagnosis: integrating molecular biologic insights into clinical practice for CLL patients. Blood 2004; 103: 1202–1210.
Spaner D, Miller R, Mena J, Grossman L, Sorrenti V, Shi Y . Regression of skin deposits in a CLL patient treated with the TLR7/8 agonist, Imiquimod. Leuk Lymphoma 2005; 46: 935–939.
Darlak K, Miller RB, Stack MS, Spatola AF, Gray RD . Thiol-based inhibitors of mammalian collagenase. J Biol Chem 1990; 265: 5199–5205.
Bueno C, Rodriguez-Caballero A, Garcia-Montero A, Orfao A . A new method for detecting TNFα-secreting cells using direct-immunofluorescence. J Immunol Methods 2002; 264: 77–87.
Hammond C, Shi Y, Mena J, Tomic J, Cervi D, He L et al. Effect of serum and antioxidants on the immunogenicity of PKC-activated CLL cells. J Immunother 2005; 28: 28–39.
Wallgren A, Festin R, Gidlof C, Dohlsten M, Kalland T, Totterman TH . Efficient killing of B-CLL cells by superantigen-directed T cells. Blood 1993; 82: 1230–1238.
Gitelson E, Hammond C, Mena J, Lorenzo M, Berinstein N, Spaner D . CLL-reactive T cells during tumor progression and after autologous tumor vaccines. Clin Cancer Res 2003; 9: 1656–1665.
Auphan N, DiDonato JA, Helmberg A, Karin M . Immunosuppression by glucocorticoids: inhibition of NFκB activity through induction of IκB synthesis. Science 1995; 270: 286–290.
Lee JC, Kassis S, Kumar S, Badger A, Adams JL . p38 mitogen-activated protein kinase inhibitors – mechanisms and therapeutic potentials. Pharmacol Ther 1999; 82: 389–397.
Li L, Cousart S, Hu J, McCall CE . Characterization of interleukin-1 receptor-associated kinase in normal and endotoxin-tolerant cells. J Biol Chem 2000; 275: 23340–23345.
Gamero AM, Young HA, Wiltrout RH . Inactivation of Stat3 in tumor cells: releasing a brake on immune responses against cancer? Cancer Cell 2004; 5: 111–112.
Sengupta TK, Talbot ES, Scherle PA, Ivashkiv LB . Rapid inhibition of IL6 signaling and Stat3 activation mediated by MAP kinases. Proc Natl Acad Sci USA 1998; 95: 11107–11112.
Gorden KB, Gorski KS, Gibson SJ, Kedl RM, Tomai MA, Alkan SS et al. Synthetic agonists reveal differences between human TLR7 and TLR8. J Immunol 2005; 174: 1259–1268.
Lecoeur H, Fevrier M, Garcia S, Riviere Y, Gougeon ML . A novel flow cytometric assay for characterization of cell-mediated cytotoxicity. J Immunol Methods 2001; 253: 177–187.
Bekeredjian-Ding IB, Wagner M, Schnurr M, Endres S, Hartmann G . Plasmacytoid DCs control TLR7 sensitivity of naive B cells via type I IFN. J Immunol 2005; 174: 4043–4050.
Fan H, Cook JA . Mechanisms of endotoxin tolerance. J Endotoxin Res 2004; 10: 71–84.
Tosi P, Zinzani PL, Pellacani A, Ottaviani E, Magagnoli M, Tura S . Loxoribine affects fludarabine activity on freshly isolated B-CLL cells. Leuk Lymphoma 1997; 26: 343–348.
Acknowledgements
Supported by grants from the Leukemia Research Fund of Canada and the Ontario Cancer Research Network (OCRN) (to DS).RM and MT are employed by a company whose (potential) product was studied in this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information is available on the Leukemia website (http://www.nature.com/leu/).
Supplementary information
Rights and permissions
About this article
Cite this article
Spaner, D., Shi, Y., White, D. et al. Immunomodulatory effects of Toll-like receptor-7 activation on chronic lymphocytic leukemia cells. Leukemia 20, 286–295 (2006). https://doi.org/10.1038/sj.leu.2404061
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404061
Keywords
This article is cited by
-
In-vitro Modulation of mTOR-HIF-1α Axis by TLR7/8 Agonist (Resiquimod) in B-Chronic Lymphocytic Leukemia
Indian Journal of Hematology and Blood Transfusion (2023)
-
Harnessing cancer immunotherapy during the unexploited immediate perioperative period
Nature Reviews Clinical Oncology (2020)
-
The Expression of Toll-Like Receptors in Patients with B-Cell Chronic Lymphocytic Leukemia
Archivum Immunologiae et Therapiae Experimentalis (2016)
-
Local administration of a novel Toll-like receptor 7 agonist in combination with doxorubicin induces durable tumouricidal effects in a murine model of T cell lymphoma
Journal of Hematology & Oncology (2015)
-
Toll-like receptors in the pathogenesis of human B cell malignancies
Journal of Hematology & Oncology (2014)