Abstract
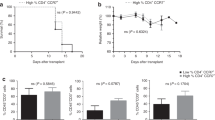
CC-chemokine receptor 7 (CCR7), a chemokine receptor required for transmigration into lymphoid organs, is only expressed by naive and central memory T cells. T cells with a capacity of homing into lymphoid organs can initiate acute graft-versus-host disease (GVHD) in mice and respond vigorously in vitro to alloantigens in humans, but their impact on clinical outcomes is unknown. We evaluated prospectively the distribution of naive, central memory and CCR7neg memory T-cell subsets in 39 bone marrow and 23 granulocyte colony-stimulating factor-mobilized peripheral blood stem cell allografts and investigated their impact on patient outcomes. Ranges of the relative proportions of CCR7+ cells within CD4+ and CD8+ T-cell populations were broad, but did not differ between the two sources of allografts. By multivariate analysis, high percentage of donor-derived CD4+CCR7+ T cells (>73.5%) significantly correlated with incidence, earliness of onset and severity of acute GVHD, conferring the highest adjusted hazard ratio (HR=3.9; 95% confidence interval 1.4–10.8; P=0.008) without interfering in other clinical events, especially chronic GVHD and relapse. Determination of the percentage of CD4+CCR7+ T cells in the graft provides a predictive indicator of acute GVHD. Partial depletion of this subset may reduce the risk of acute GVHD while preserving immunotherapeutic effects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Barrett AJ, Rezvani K, Solomon S, Dickinson AM, Wang XN, Stark G et al. New developments in allotransplant immunology. Hematology (Am Soc Hematol Educ Program) 2003, 350–371.
Beilhack A, Schulz S, Baker J, Beilhack GF, Wieland CB, Herman EI et al. In vivo analyses of early events in acute graft-versus-host disease reveal sequential infiltration of T-cell subsets. Blood 2005; 106: 1113–1122.
Ferrara JL, Reddy P . Pathophysiology of graft-versus-host disease. Semin Hematol 2006; 43: 3–10.
Winsock CA, Panoskaltsis-Mortari A, Blazar BR, Serody JS . Leukocyte migration and graft-versus-host disease. Blood 2005; 105: 4191–4199.
Beckmann JH, Yan S, Luhrs H, Heid B, Skubich S, Forster R et al. Prolongation of allograft survival in ccr7-deficient mice. Transplantation 2004; 77: 1809–1814.
Hopken UE, Droese J, Li JP, Joergensen J, Breitfeld D, Zerwes HG et al. The chemokine receptor CCR7 controls lymph node-dependent cytotoxic T cell priming in alloimmune responses. Eur J Immunol 2004; 34: 461–470.
Sasaki M, Hasegawa H, Kohno M, Inoue A, Ito MR, Fujita S . Antagonist of secondary lymphoid-tissue chemokine (CCR ligand 21) prevents the development of chronic graft-versus-host disease in mice. J Immunol 2003; 170: 588–596.
Dutt S, Ermann J, Tseng D, Liu YP, George TI, Fathman CG et al. L-selectin and beta7 integrin on donor CD4T cells are required for the early migration to host mesenteric lymph nodes and acute colitis of graft-versus-host disease. Blood 2005; 106: 4009–4015.
Li B, New JY, Tay YK, Goh E, Yap EH, Chan SH et al. Delaying acute graft-versus-host disease in mouse bone marrow transplantation by treating donor cells with antibodies directed at L-selectin and alpha4-integrin prior to infusion. Scand J Immunol 2004; 59: 464–468.
Li B, New JY, Yap EH, Lu J, Chan SH, Hu H . Blocking L-selectin and alpha4-integrin changes donor cell homing pattern and ameliorates murine acute graft versus host disease. Eur J Immunol 2001; 31: 617–624.
Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A . Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999; 401: 708–712.
Weninger W, Crowley MA, Manjunath N, von Andrian UH . Migratory properties of naive, effector, and memory CD8(+) T cells. J Exp Med 2001; 194: 953–966.
Anderson BE, McNiff J, Yan J, Doyle H, Mamula M, Shlomchik MJ et al. Memory CD4+ T cells do not induce graft-versus-host disease. J Clin Invest 2003; 112: 101–108.
Chen BJ, Cui X, Sempowski GD, Liu C, Chao NJ . Transfer of allogeneic CD62L- memory T cells without graft-versus-host disease. Blood 2004; 103: 1534–1541.
Adams AB, Williams MA, Jones TR, Shirasugi N, Durham MM, Kaech SM et al. Heterologous immunity provides a potent barrier to transplantation tolerance. J Clin Invest 2003; 111: 1887–1895.
Foster AE, Marangolo M, Sartor MM, Alexander SI, Hu M, Bradstock KF et al. Human CD62L− memory T cells are less responsive to alloantigen stimulation than CD62L+ naive T cells: potential for adoptive immunotherapy and allodepletion. Blood 2004; 104: 2403–2409.
Bondanza A, Valtolina V, Magnani Z, Ponzoni M, Fleischhauer K, Bonyhadi M et al. Suicide gene therapy of graft-versus-host disease induced by central memory human T lymphocytes. Blood 2006; 107: 1828–1836.
Appay V, Dunbar PR, Callan M, Klenerman P, Gillespie GM, Papagno L et al. Memory CD8+ T cells vary in differentiation phenotype in different persistent virus infections. Nat Med 2002; 8: 379–385.
van Lier RA, ten Berge IJ, Gamadia LE . Human CD8(+) T-cell differentiation in response to viruses. Nat Rev Immunol 2003; 3: 931–939.
Harari A, Vallelian F, Pantaleo G . Phenotypic heterogeneity of antigen-specific CD4T cells under different conditions of antigen persistence and antigen load. Eur J Immunol 2004; 34: 3525–3533.
Saule P, Trauet J, Dutriez V, Lekeux V, Dessaint JP, Labalette M . Accumulation of memory T cells from childhood to old age: central and effector memory cells in CD4(+) versus effector memory and terminally differentiated memory cells in CD8(+) compartment. Mech Ageing Dev 2006; 127: 274–281.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant 1995; 15: 825–828.
Kaplan E, Meier P . Non parametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Peto R, Peto J . Asymptotically efficient rank invariant test procedures. J R Stat Soc 1972; 135: 185–206.
Cox D . Regression models and life tables (with discussion). J R Stat Soc 1972; 34: 187–220.
Hopken U, Droeses J, Joergensen J, Breitfeld D, Zerwes HML . The chemokine receptor CCR7 controls lymph node-dependent cytotoxic T cell priming in alloimmune responses. J Immunol 2004; 34: 461–470.
Sallusto F, Geginat J, Lanzavecchia A . Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol 2004; 22: 745–763.
Ottinger HD, Beelen DW, Scheulen B, Schaefer UW, Grosse-Wilde H . Improved immune reconstitution after allotransplantation of peripheral blood stem cells instead of bone marrow. Blood 1996; 88: 2775–2779.
Storek J, Dawson MA, Storer B, Stevens-Ayers T, Maloney DG, Marr KA et al. Immune reconstitution after allogeneic marrow transplantation compared with blood stem cell transplantation. Blood 2001; 97: 3380–3389.
Robinet E, Lapierre V, Tayebi H, Kuentz M, Blaise D, Tiberghien P . Blood versus marrow hematopoietic allogeneic graft. Transfus Apheresis Sci 2003; 29: 53–59.
Tayebi H, Kuttler F, Saas P, Lienard A, Petracca B, Lapierre V et al. Effect of granulocyte colony-stimulating factor mobilization on phenotypical and functional properties of immune cells. Exp Hematol 2001; 29: 458–470.
Chen SH, Li X, Huang XJ . Effect of recombinant human granulocyte colony-stimulating factor on T-lymphocyte function and the mechanism of this effect. Int J Hematol 2004; 79: 178–184.
Xystrakis E, Bernard I, Dejean AS, Alsaati T, Druet P, Saoudi A . Alloreactive CD4T lymphocytes responsible for acute and chronic graft-versus-host disease are contained within the CD45RChigh but not the CD45RClow subset. Eur J Immunol 2004; 34: 408–417.
Rocha B, Tanchot C . Towards a cellular definition of CD8+ T-cell memory: the role of CD4+ T-cell help in CD8+ T-cell responses. Curr Opin Immunol 2004; 16: 259–263.
Yu XZ, Albert MH, Anasetti C . Alloantigen affinity and CD4 help determine severity of graft-versus-host disease mediated by CD8 donor T cells. J Immunol 2006; 176: 3383–3390.
Blaise D, Kuentz M, Fortanier C, Bourhis JH, Milpied N, Sutton L et al. Randomized trial of bone marrow versus lenograstim-primed blood cell allogeneic transplantation in patients with early-stage leukemia: a report from the Societe Francaise de Greffe de Moelle. J Clin Oncol 2000; 18: 537–546.
Guardiola P, Runde V, Bacigalupo A, Ruutu T, Locatelli F, Boogaerts MA et al. Retrospective comparison of bone marrow and granulocyte colony-stimulating factor-mobilized peripheral blood progenitor cells for allogeneic stem cell transplantation using HLA identical sibling donors in myelodysplastic syndromes. Blood 2002; 99: 4370–4378.
Atkinson K, Farrelly H, Cooley M, O’Flaherty E, Downs K, Biggs J . Human marrow T cell dose correlates with severity of subsequent acute graft-versus-host disease. Bone Marrow Transplant 1987; 2: 51–57.
Kernan NA, Collins NH, Juliano L, Cartagena T, Dupont B, O’Reilly RJ . Clonable T lymphocytes in T cell-depleted bone marrow transplants correlate with development of graft-v-host disease. Blood 1986; 68: 770–773.
Urbano-Ispizua A, Rozman C, Pimentel P, Solano C, de la Rubia J, Brunet S et al. The number of donor CD3(+) cells is the most important factor for graft failure after allogeneic transplantation of CD34(+) selected cells from peripheral blood from HLA-identical siblings. Blood 2001; 97: 383–387.
Aversa F, Tabilio A, Velardi A, Cunningham I, Terenzi A, Falzetti F et al. Treatment of high-risk acute leukemia with T-cell-depleted stem cells from related donors with one fully mismatched HLA haplotype. N Engl J Med 1998; 339: 1186–1193.
Maus MV, Kovacs B, Kwok WW, Nepom GT, Schlienger K, Riley JL et al. Extensive replicative capacity of human central memory T cells. J Immunol 2004; 172: 6675–6683.
Higman MA, Vogelsang GB . Chronic graft versus host disease. Br J Haematol 2004; 125: 435–454.
Horwitz ME, Sullivan KM . Chronic graft-versus-host disease. Blood Rev 2006; 20: 15–27.
Kansu E . The pathophysiology of chronic graft-versus-host disease. Int J Hematol 2004; 79: 209–215.
Mohty M, Kuentz M, Michallet M, Bourhis JH, Milpied N, Sutton L et al. Chronic graft-versus-host disease after allogeneic blood stem cell transplantation: long-term results of a randomized study. Blood 2002; 100: 3128–3134.
Marleau AM, Sarvetnick N . T cell homeostasis in tolerance and immunity. J Leukocyte Biol 2005; 78: 575–584.
van den Brink MR, Moore E, Ferrara JL, Burakoff SJ . Graft-versus-host-disease-associated thymic damage results in the appearance of T cell clones with anti-host reactivity. Transplantation 2000; 69: 446–449.
Weinberg K, Blazar BR, Wagner JE, Agura E, Hill BJ, Smogorzewska M et al. Factors affecting thymic function after allogeneic hematopoietic stem cell transplantation. Blood 2001; 97: 1458–1466.
Teshima T, Reddy P, Liu C, Williams D, Cooke KR, Ferrara JL . Impaired thymic negative selection causes autoimmune graft-versus-host disease. Blood 2003; 102: 429–435.
Hakim FT, Memon SA, Cepeda R, Jones EC, Chow CK, Kasten-Sportes C et al. Age-dependent incidence, time course, and consequences of thymic renewal in adults. J Clin Invest 2005; 115: 930–939.
Zorn E, Kim HT, Lee SJ, Floyd BH, Litsa D, Arumugarajah S et al. Reduced frequency of FOXP3+ CD4+CD25+ regulatory T cells in patients with chronic graft-versus-host disease. Blood 2005; 106: 2903–2911.
Pavletic SZ, Carter SL, Kernan NA, Henslee-Downey J, Mendizabal AM, Papadopoulos E et al. Influence of T-cell depletion on chronic graft-versus-host disease: results of a multicenter randomized trial in unrelated marrow donor transplantation. Blood 2005; 106: 3308–3313.
Slavin S, Nagler A, Naparstek E, Kapelushnik Y, Aker M, Cividalli G et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998; 91: 756–763.
Zecca M, Prete A, Rondelli R, Lanino E, Balduzzi A, Messina C et al. Chronic graft-versus-host disease in children: incidence, risk factors, and impact on outcome. Blood 2002; 100: 1192–1200.
Baron F, Maris MB, Storer BE, Sandmaier BM, Panse JP, Chauncey TR et al. High doses of transplanted CD34+ cells are associated with rapid T-cell engraftment and lessened risk of graft rejection, but not more graft-versus-host disease after nonmyeloablative conditioning and unrelated hematopoietic cell transplantation. Leukemia 2005; 19: 822–828.
Fowler DH . Shared biology of GVHD and GVT effects: potential methods of separation. Crit Rev Oncol Hematol 2006; 57: 225–244.
Rezvani K, Grube M, Brenchley JM, Sconocchia G, Fujiwara H, Price DA et al. Functional leukemia-associated antigen-specific memory CD8+ T cells exist in healthy individuals and in patients with chronic myelogenous leukemia before and after stem cell transplantation. Blood 2003; 102: 2892–2900.
Verdijk RM, Kloosterman A, Pool J, van de Keur M, Naipal AM, van Halteren AG et al. Pregnancy induces minor histocompatibility antigen-specific cytotoxic T cells: implications for stem cell transplantation and immunotherapy. Blood 2004; 103: 1961–1964.
Acknowledgements
We thank Dr Léonardo Magro, Dr Stéphane Darré, the hematology fellows and other senior physicians. We are especially grateful to Sandrine Apaza-Lopez, Jacques Trauet, Véronique Lekeux, Virginie Dutriez and staffs involved in the care of the BMT unit. We also thank all the donors and patients who agreed to be enrolled in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yakoub-Agha, I., Saule, P., Depil, S. et al. A high proportion of donor CD4+ T cells expressing the lymph node-homing chemokine receptor CCR7 increases incidence and severity of acute graft-versus-host disease in patients undergoing allogeneic stem cell transplantation for hematological malignancy. Leukemia 20, 1557–1565 (2006). https://doi.org/10.1038/sj.leu.2404308
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404308
Keywords
This article is cited by
-
Evaluation of therapeutic targeting of CCR7 in acute graft-versus-host disease
Bone Marrow Transplantation (2020)
-
Effect of bone marrow CD34+cells and T-cell subsets on clinical outcomes after myeloablative allogeneic hematopoietic cell transplantation
Bone Marrow Transplantation (2019)
-
Donor-derived CD4+/CCR7+ T-cell impact on acute GVHD incidence following haplo-HCT after reduced intensity conditioning and posttransplant cyclophosphamide
Bone Marrow Transplantation (2019)
-
Pre-transplant donor CD4− invariant NKT cell expansion capacity predicts the occurrence of acute graft-versus-host disease
Leukemia (2017)
-
A high migratory capacity of donor T-cells in response to the lymph node homing receptor CCR7 increases the incidence and severity of GvHD
Bone Marrow Transplantation (2017)