Abstract
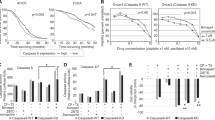
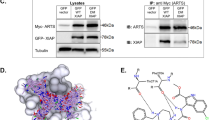
Maspin, a novel serine protease inhibitor (serpin), suppresses the growth and metastasis of breast tumor in vivo. However, the underlying molecular mechanism is unclear. In the current study, we report the first evidence that endogenous maspin expression in mammary carcinoma cells MDA-MB-435 enhanced staurosporine (STS)-induced apoptosis as judged by the increased fragmentation of DNA, increased proteolytic inactivation of poly-[ADP-ribose]-polymerase (PARP), as well as the increased activation of caspase-8 and caspase-3. In parallel, recombinant maspin did not directly regulate the proteolytic activities of either caspase-3 or caspase-8 in vitro. Consistent with this result, maspin expressing normal mammary epithelial cells underwent more rapid STS-induced apoptosis as compared to breast carcinoma cells. Interestingly, maspin transfectant cells did not undergo spontaneous apoptosis in the absence of STS. Moreover, neither purified maspin protein added from outside nor endogenous maspin secreted to the cell culture media sensitized cells to STS-induced apoptosis. To investigate the structural determinants of maspin in its apoptosis-sensitizing effect, MDA-MB-435 cells were also transfected with maspin/PAI-1 and PAI-1/maspin chimeric constructs resulting from swapping the N-terminal and the C-terminal domains between maspin and PAI-1 (plasminogen activator inhibitor type 1). The resulting stable transfectant clones expressing maspin/PAI-1 and PAI-1/maspin, respectively, did not undergo spontaneous apoptosis, and were similarly inhibited as maspin transfectant cells in motility assay. Interestingly, however, expression of both maspin/PAI-1 and PAI-1/maspin in MDA-MB-435 cells failed to sensitize these cells to STS-induced apoptosis. Taken together, our evidence provides new insights into the complex molecular mechanisms of maspin that may suppress breast tumor progression not only at the step of invasion and motility, but also by regulating tumor cell apoptosis. The sensitizing effect of maspin on apoptosis is to be contrasted by the pro-survival effect of several other serpins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Andreasen PA, Egelund R, Petersen HH . 2000 Cell Mol. Life Sci. 57: 25–40
Band V, Sager R . 1989 Proc. Natl. Acad. Sci. USA 86: 1249–1253
Biliran Jr H, Sheng S . 2001 Cancer Res. 61: 8676–8682
Biswas DK, Dai S, Cruz A, Weiser B, Graner E, Pardee AB . 2001 Proc. Natl. Acad. Sci. USA 98: 10386–10391
Blasi F . 1999 Thromb. Haemost. 82: 298–304
Boulares AH, Yakovlev AG, Ivanova V, Stoica BA, Wang G, Iyer S, Smulson M . 1999 J. Biol. Chem. 274: 22932–22940
Bromme HJ, Holtz J . 1996 Mol Cell Biochem 163–164: 261–275
Budihardjo I, Oliver H, Lutter M, Luo X, Wang X . 1999 Annu. Rev. Cell Dev. Biol. 15: 269–290
Chen CY, Juo P, Liou JS, Li CQ, Yu Q, Blenis J, Faller DV . 2001 Cell Growth Differ. 12: 297–306
D'Amours D, Germain M, Orth K, Dixit VM, Poirier GG . 1998 Radiat. Res. 150: 3–10
Dickinson JL, Bates EJ, Ferrante A, Antalis TM . 1995 J. Biol. Chem. 270: 27894–27904
Dickinson JL, Norris BJ, Jensen PH, Antalis TM . 1998 Cell Death Differ. 5: 163–171
Gomez-Angelats M, Bortner CD, Cidlowski JA . 2000 J. Biol. Chem. 275: 19609–19619
Hall AK . 1995 Cell Mol. Biol. Res. 41: 167–180
Hopkins PC, Whisstock J . 1994 Science 265: 1893–1894
Jacobsen MD, Weil M, Raff MC . 1996 J. Cell Biol. 133: 1041–1051
Kanamori H, Krieg S, Mao C, Di Pippo VA, Wang S, Zajchowski DA, Shapiro DJ . 2000 J. Biol. Chem. 275: 5867–5873
Kwaan HC, Wang J, Svoboda K, Declerck PJ . 2000 Br. J Cancer 82: 1702–1708
Lawrence DA . 1997 Adv. Exp. Med. Biol. 425: 99–108
Lee DH, Goldberg AL . 1998 Trends Cell Biol. 8: 397–403
Li JJ, Colburn NH, Oberley LW . 1998 Carcinogenesis 19: 833–839
Los M, Van de Craen M, Penning LC, Schenk H, Westendorp M, Baeuerle PA, Droge W, Krammer PH, Fiers W, Schulze-Osthoff K . 1995 Nature 375: 81–83
McGowen R, Biliran Jr H, Sager R, Sheng S . 2000 Cancer Res. 60: 4771–4778
Miura M, Friedlander RM, Yuan J . 1995 Proc. Natl. Acad. Sci. USA 92: 8318–8322
Miura M, Zhu H, Rotello R, Hartwieg EA, Yuan J . 1993 Cell 75: 653–660
Ossowski L, Aguirre-Ghiso JA . 2000 Curr. Opin. Cell. Biol. 12: 613–620
Palombella VJ, Rando OJ, Goldberg AL, Maniatis T . 1994 Cell 78: 773–785
Pemberton PA, Tipton AR, Pavloff N, Smith J, Erickson JR, Mouchabeck ZM, Kiefer MC . 1997 J. Histochem. Cytochem. 45: 1697–1706
Potempa J, Korzus E, Travis J . 1994 J. Biol. Chem. 269: 15957–15960
Sager R, Sheng S, Pemberton P, Hendrix MJ . 1996 Curr. Top. Microbiol. Immunol. 213: 51–64
Schleef RR, Chuang TL . 2000 J. Biol. Chem. 275: 26385–26389
Seftor RE, Seftor EA, Sheng S, Pemberton PA, Sager R, Hendrix MJ . 1998 Cancer Res. 58: 5681–5685
Shao ZM, Nguyen M, Alpaugh ML, O'Connell JT, Barsky SH . 1998 Exp. Cell Res. 241: 394–403
Sheng S, Carey J, Seftor EA, Dias L, Hendrix MJ, Sager R . 1996 Proc. Natl. Acad. Sci. USA 93: 11669–11674
Sheng S, Pemberton PA, Sager R . 1994 J. Biol. Chem. 269: 30988–30993
Sheng S, Truong B, Frederickson D, Wu R, Pardee AB, Sager R . 1998 Proc. Natl. Acad. Sci. USA 95: 499–504
Tamaoki T, Nomoto H, Takahashi I, Kato Y, Morimoto M, Tomita F . 1986 Biochem. Biophys. Res. Commun. 135: 397–402
Tang D, Lahti JM, Kidd VJ . 2000 J. Biol. Chem. 275: 9303–9307
Travis J, Guzdek A, Potempa J, Watorek W . 1990 Biol. Chem. Hoppe Seyler 371: Suppl) 3–11
Vercammen D, Beyaert R, Denecker G, Goossens V, Van Loo G, Declercq W, Grooten J, Fiers W, Vandenabeele P . 1998 J. Exp. Med. 187: 1477–1485
Yu FX, Lin SC, Morrison-Bogorad M, Atkinson MA, Yin HL . 1993 J. Biol. Chem. 268: 502–509
Zhang M, Sheng S, Maass N, Sager R . 1997 Mol. Med. 3: 49–59
Zhang M, Shi Y, Magit D, Furth PA, Sager R . 2000 Oncogene 19: 6053–6058
Zhang M, Magit D, Botteri F, Shi HY, He K, Li M, Furth P, Sager R . 1999 Dev. Biol. 215: 278–287
Zou Z, Anisowicz A, Hendrix MJ, Thor A, Neveu M, Sheng S, Rafidi K, Seftor E, Sager R . 1994 Science 263: 526–529
Acknowledgements
This work was supported in part by a Ruth Sager Memorial Fund (to S Sheng) and a DOD grant BC0996974 (to S Sheng). The authors wish to thank Mr Hector Biliran Jr and Dr Shuping Yin for critical review and proofreading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, N., Meng, Y., Zhang, S. et al. Maspin sensitizes breast carcinoma cells to induced apoptosis. Oncogene 21, 4089–4098 (2002). https://doi.org/10.1038/sj.onc.1205507
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205507
Keywords
This article is cited by
-
Tackling tumor heterogeneity and phenotypic plasticity in cancer precision medicine: our experience and a literature review
Cancer and Metastasis Reviews (2018)
-
Maspin enhances cisplatin chemosensitivity in bladder cancer T24 and 5637 cells and correlates with prognosis of muscle-invasive bladder cancer patients receiving cisplatin based neoadjuvant chemotherapy
Journal of Experimental & Clinical Cancer Research (2016)
-
A phase II trial of ganetespib, a heat shock protein 90 Hsp90) inhibitor, in patients with docetaxel-pretreated metastatic castrate-resistant prostate cancer (CRPC)-a prostate cancer clinical trials consortium (PCCTC) study
Investigational New Drugs (2016)
-
Association between SNPs in Serpin gene family and risk of esophageal squamous cell carcinoma
Tumor Biology (2015)
-
Nuclear location of tumor suppressor protein maspin inhibits proliferation of breast cancer cells without affecting proliferation of normal epithelial cells
BMC Cancer (2014)