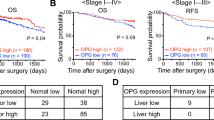
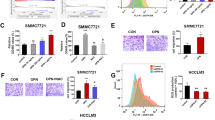
Abstract
Tumor cell invasion is a primary event in the metastatic progression of hepatocellular carcinoma (HCC). Our recent results indicate a concordant elevated expression of osteopontin (OPN) and matrix metalloproteinase-9 (MMP-9) in primary metastatic HCC. This study hypothesizes an MMP-9-directed cleavage of OPN that biologically contributes to HCC metastasis. We found that MMP-9 cleaved OPN into specific fragments in vitro, of which three could be identified by Edman degradation amino-acid sequencing. One of these fragments (OPN-5 kDa, residues 167–210) induced low-metastatic HCC cellular invasion via CD44 receptors, which was effectively blocked by the addition of small peptides within the region of OPN-5 kDa. Increased expression of an OPN splice variant (OPN-c) was associated with clinical metastatic HCC. Overexpression of OPN-c with physiological levels of MMP-9 enhanced cellular invasion and coincided with elevated OPN-5 kDa levels. Our data suggest that an alternative splicing event (OPN-c) promotes extracellular cleavage of OPN by MMP-9, thus releasing a distinct region of OPN (OPN-5 kDa) that is essential for HCC cellular invasion and appears to correlate with metastatic potential. The findings of this study may help to improve advanced-stage HCC prognosis and suggest the utility of small peptides for novel therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Abbreviations
- HCC:
-
hepatocellular carcinoma
- MMP-9:
-
matrix metalloproteinase-9
- OPN:
-
osteopontin
References
Agnihotri R, Crawford HC, Haro H, Matrisian LM, Havrda MC, Liaw L . (2001). Osteopontin, a novel substrate for matrix metalloproteinase-3 (stromelysin-1) and matrix metalloproteinase-7 (matrilysin). J Biol Chem 276: 28261–28267.
Ashida K, Nakatsukasa H, Higashi T, Ohguchi S, Hino N, Nouso K et al. (1996). Cellular distribution of 92-kd type IV collagenase/gelatinase B in human hepatocellular carcinoma. Am J Pathol 149: 1803–1811.
Blum HE . (2005). Liver cancer. Eur J Gastroenterol Hepatol 17: 475–476.
Butler WT . (1995). Structural and functional domains of osteopontin. Ann NY Acad Sci 760: 6–11.
Coppola D, Szabo M, Boulware D, Muraca P, Alsarraj M, Chambers AF et al. (2004). Correlation of osteopontin protein expression and pathological stage across a wide variety of tumor histologies. Clin Cancer Res 10: 184–190.
Denhardt DT, Noda M, O’Regan AW, Pavlin D, Berman JS . (2001). Osteopontin as a means to cope with environmental insults: regulation of inflammation, tissue remodeling, and cell survival. J Clin Invest 107: 1055–1061.
Endo K, Terada T . (2000). Protein expression of CD44 (standard and variant isoforms) in hepatocellular carcinoma: relationships with tumor grade, clinicopathologic parameters, p53 expression, and patient survival. J Hepatol 32: 78–84.
Fedarko NS, Jain A, Karadag A, Van Eman MR, Fisher LW . (2001). Elevated serum bone sialoprotein and osteopontin in colon, breast, prostate, and lung cancer. Clin Cancer Res 7: 4060–4066.
Franco D, Capussotti L, Smadja C, Bouzari H, Meakins J, Kemeny F et al. (1990). Resection of hepatocellular carcinomas. Results in 72 European patients with cirrhosis. Gastroenterology 98: 733–738.
Harada N, Mizoi T, Kinouchi M, Hoshi K, Ishii S, Shiiba K et al. (2001). Introduction of antisense CD44S CDNA down-regulates expression of overall CD44 isoforms and inhibits tumor growth and metastasis in highly metastatic colon carcinoma cells. Int J Cancer 91: 67–75.
He B, Mirza M, Weber GF . (2006). An osteopontin splice variant induces anchorage independence in human breast cancer cells. Oncogene 25: 2192–2202.
Hotte SJ, Winquist EW, Stitt L, Wilson SM, Chambers AF . (2002). Plasma osteopontin: associations with survival and metastasis to bone in men with hormone-refractory prostate carcinoma. Cancer 95: 506–512.
Kaartinen MT, Pirhonen A, Linnala-Kankkunen A, Maenpaa PH . (1999). Cross-linking of osteopontin by tissue transglutaminase increases its collagen binding properties. J Biol Chem 274: 1729–1735.
Kaneyoshi T, Nakatsukasa H, Higashi T, Fujiwara K, Naito I, Nouso K et al. (2001). Actual invasive potential of human hepatocellular carcinoma revealed by in situ gelatin zymography. Clin Cancer Res 7: 4027–4032.
Lesley J, Hyman R . (1998). CD44 structure and function. Front Biosci 3: d616–d630.
Lin LI, Ke YF, Ko YC, Lin JK . (1998). Curcumin inhibits SK-Hep-1 hepatocellular carcinoma cell invasion in vitro and suppresses matrix metalloproteinase-9 secretion. Oncology 55: 349–353.
Matsumura Y, Tarin D . (1992). Significance of CD44 gene products for cancer diagnosis and disease evaluation. Lancet 340: 1053–1058.
McKenna GJ, Chen Y, Smith RM, Meneghetti A, Ong C, McMaster R et al. (2002). A role for matrix metalloproteinases and tumor host interaction in hepatocellular carcinomas. Am J Surg 183: 588–594.
Pan HW, Ou YH, Peng SY, Liu SH, Lai PL, Lee PH et al. (2003). Overexpression of osteopontin is associated with intrahepatic metastasis, early recurrence, and poorer prognosis of surgically resected hepatocellular carcinoma. Cancer 98: 119–127.
Ponta H, Sherman L, Herrlich PA . (2003). CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol 4: 33–45.
Prince CW, Dickie D, Krumdieck CL . (1991). Osteopontin, a substrate for transglutaminase and factor XIII activity. Biochem Biophys Res Commun 177: 1205–1210.
Rittling SR, Chambers AF . (2004). Role of osteopontin in tumour progression. Br J Cancer 90: 1877–1881.
Rittling SR, Chen Y, Feng F, Wu Y . (2002). Tumor-derived osteopontin is soluble, not matrix associated. J Biol Chem 277: 9175–9182.
Rudzki Z, Jothy S . (1997). CD44 and the adhesion of neoplastic cells. Mol Pathol 50: 57–71.
Saitoh Y, Kuratsu J, Takeshima H, Yamamoto S, Ushio Y . (1995). Expression of osteopontin in human glioma. Its correlation with the malignancy. Lab Invest 72: 55–63.
Senger DR, Perruzzi CA, Gracey CF, Papadopoulos A, Tenen DG . (1988). Secreted phosphoproteins associated with neoplastic transformation: close homology with plasma proteins cleaved during blood coagulation. Cancer Res 48: 5770–5774.
Si MS, Amersi F, Golish SR, Ortiz JA, Zaky J, Finklestein D et al. (2003). Prevalence of metastases in hepatocellular carcinoma: risk factors and impact on survival. Am Surg 69: 879–885.
Singhal H, Bautista DS, Tonkin KS, O’Malley FP, Tuck AB, Chambers AF et al. (1997). Elevated plasma osteopontin in metastatic breast cancer associated with increased tumor burden and decreased survival. Clin Cancer Res 3: 605–611.
Sorensen ES, Rasmussen LK, Moller L, Jensen PH, Hojrup P, Petersen TE . (1994). Localization of transglutaminase-reactive glutamine residues in bovine osteopontin. Biochem J 304 (Part 1): 13–16.
Takahashi K, Takahashi F, Tanabe KK, Takahashi H, Fukuchi Y . (1998). The carboxyl-terminal fragment of osteopontin suppresses arginine–glycine–asparatic acid-dependent cell adhesion. Biochem Mol Biol Int 46: 1081–1092.
Theret N, Musso O, Turlin B, Lotrian D, Bioulac-Sage P, Campion JP et al. (2001). Increased extracellular matrix remodeling is associated with tumor progression in human hepatocellular carcinomas. Hepatology 34: 82–88.
Turpeenniemi-Hujanen T . (2005). Gelatinases (MMP-2 and -9) and their natural inhibitors as prognostic indicators in solid cancers. Biochimie 87: 287–297.
van Kempen LC, Coussens LM . (2002). MMP9 potentiates pulmonary metastasis formation. Cancer Cell 2: 251–252.
Wai PY, Kuo PC . (2004). The role of Osteopontin in tumor metastasis. J Surg Res 121: 228–241.
Wang X . (2005). Osteopontin overexpression in hepatocellular carcinoma is a predictor of survival after liver transplantation. Hepatology 42: 391A.
Weber GF . (2001). The metastasis gene osteopontin: a candidate target for cancer therapy. Biochim Biophys Acta 1552: 61–85.
Weber GF, Ashkar S, Cantor H . (1997). Interaction between CD44 and osteopontin as a potential basis for metastasis formation. Proc Assoc Am Physicians 109: 1–9.
Weber GF, Zawaideh S, Hikita S, Kumar VA, Cantor H, Ashkar S . (2002). Phosphorylation-dependent interaction of osteopontin with its receptors regulates macrophage migration and activation. J Leukoc Biol 72: 752–761.
Ye QH, Qin LX, Forgues M, He P, Kim JW, Peng AC et al. (2003). Predicting hepatitis B virus-positive metastatic hepatocellular carcinomas using gene expression profiling and supervised machine learning. Nat Med 9: 416–423.
Acknowledgements
This work was supported by intramural funding at the Center for Cancer Research, National Cancer Institute, National Institutes of Health, Bethesda, MD, USA. The authors thank Dominic Esposito of the SAIC-Frederick Protein Expression Lab, Frederick, MD, USA for help with cloning of the OPN expression constructs. We also thank Curtis Harris and Anuradha Budhu for critical comments and the NIH Fellows Editorial Board for editorial review of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Takafuji, V., Forgues, M., Unsworth, E. et al. An osteopontin fragment is essential for tumor cell invasion in hepatocellular carcinoma. Oncogene 26, 6361–6371 (2007). https://doi.org/10.1038/sj.onc.1210463
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210463
Keywords
This article is cited by
-
Meta-analysis of Osteopontin splice variants in cancer
BMC Cancer (2023)
-
Structural insights into regulation of CCN protein activities and functions
Journal of Cell Communication and Signaling (2023)
-
Osteopontin promotes hepatocellular carcinoma progression through inducing JAK2/STAT3/NOX1-mediated ROS production
Cell Death & Disease (2022)
-
Integrated analysis reveals critical glycolytic regulators in hepatocellular carcinoma
Cell Communication and Signaling (2020)
-
Osteopontin binds ICOSL promoting tumor metastasis
Communications Biology (2020)