Abstract
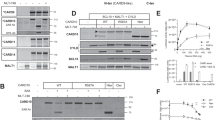
Marked reduction in apoptosis is a hallmark of early colon tumour growth and the vast majority of these tumours exhibit a loss of expression of the glycoprotein carcinoembryonic-antigen-related cell adhesion molecule 1 (CEACAM1). We recently reported that the CEACAM1 functions as a mediator of apoptosis implicating this cell surface protein in early tumour development. However, the mechanistic involvement of CEACAM1 in cell death pathways is unclear. Here, we show that apoptosis triggers cleavage of the long form of CEACAM1 (CEACAM1-4L) at intracellular and extracellular sites in Jurkat cells and HEK293 cells. Signalling through CEACAM1 leads to caspase activation including caspase-1 and -3 and also involves non-caspase proteases. Moreover, we provide evidence that the naturally occurring CEACAM family member CEA is an inducer of CEACAM1-mediated apoptosis in HT29 colon cancer cells, an effect that depends on the abundance of CEACAM1 on the cell surface. Together, our results demonstrate that the CEACAM1-dependent cell death pathway involves dual cleavage of CEACAM1 and caspase activation and can be activated by CEA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Baranov V, Yeung MM, Hammarstrom S . (1994). Expression of carcinoembryonic antigen and nonspecific cross-reacting 50-kDa antigen in human normal and cancerous colon mucosa: comparative ultrastructural study with monoclonal antibodies. Cancer Res 54: 3305–3314.
Barnett TR, Kretschmer A, Austen DA, Goebel SJ, Hart JT, Elting JJ et al. (1989). Carcinoembryonic antigens: alternative splicing accounts for the multiple mRNAs that code for novel members of the carcinoembryonic antigen family. J Cell Biol 108: 267–276.
Brummer J, Neumaier M, Gopfert C, Wagener C . (1995). Association of pp60c-src with biliary glycoprotein (CD66a), an adhesion molecule of the carcinoembryonic antigen family downregulated in colorectal carcinomas. Oncogene 11: 1649–1655.
Charbonneau J, Stanners CP . (1999). Role of carbohydrate structures in CEA-mediated intercellular adhesion. Cell Adhes Commun 7: 233–244.
Edlund M, Blikstad I, Obrink B . (1996). Calmodulin binds to specific sequences in the cytoplasmic domain of C-CAM and down-regulates C-CAM self-association. J Biol Chem 271: 1393–1399.
Edlund M, Wikstrom K, Toomik R, Ek P, Obrink B . (1998). Characterization of protein kinase C-mediated phosphorylation of the short cytoplasmic domain isoform of C-CAM. FEBS Lett 425: 166–170.
Fournes B, Sadekova S, Turbide C, Letourneau S, Beauchemin N . (2001). The CEACAM1-L Ser503 residue is crucial for inhibition of colon cancer cell tumorigenicity. Oncogene 20: 219–230.
Frangsmyr L, Baranov V, Hammarstrom S . (1999). Four carcinoembryonic antigen subfamily members, CEA, NCA, BGP and CGM2, selectively expressed in the normal human colonic epithelium, are integral components of the fuzzy coat. Tumour Biol 20: 277–292.
Frangsmyr L, Baranov V, Prall F, Yeung MM, Wagener C, Hammarstrom S . (1995). Cell- and region-specific expression of biliary glycoprotein and its messenger RNA in normal human colonic mucosa. Cancer Res 55: 2963–2967.
Hammarstrom S . (1999). The carcinoembryonic antigen (CEA) family: structures, suggested functions and expression in normal and malignant tissues. Semin Cancer Biol 9: 67–81.
Hefta SA, Hefta LJ, Lee TD, Paxton RJ, Shively JE . (1988). Carcinoembryonic antigen is anchored to membranes by covalent attachment to a glycosylphosphatidylinositol moiety: identification of the ethanolamine linkage site. Proc Natl Acad Sci USA 85: 4648–4652.
Hilbi H, Moss JE, Hersh D, Chen Y, Arondel J, Banerjee S et al. (1998). Shigella-induced apoptosis is dependent on caspase-1 which binds to IpaB. J Biol Chem 273: 32895–32900.
Hinoda Y, Neumaier M, Hefta SA, Drzeniek Z, Wagener C, Shively L et al. (1988). Molecular cloning of a cDNA coding biliary glycoprotein I: primary structure of a glycoprotein immunologically crossreactive with carcinoembryonic antigen. Proc Natl Acad Sci USA 85: 6959–6963.
Houde C, Roy S, Leung N, Nicholson DW, Beauchemin N . (2003). The cell adhesion molecule CEACAM1-L is a substrate of caspase-3-mediated cleavage in apoptotic mouse intestinal cells. J Biol Chem 278: 16929–16935.
Huang J, Hardy JD, Sun Y, Shively JE . (1999). Essential role of biliary glycoprotein (CD66a) in morphogenesis of the human mammary epithelial cell line MCF10F. J Cell Sci 112: 4193–4205.
Huber M, Izzi L, Grondin P, Houde C, Kunath T, Veillette A et al. (1999). The carboxyl-terminal region of biliary glycoprotein controls its tyrosine phosphorylation and association with protein-tyrosine phosphatases SHP-1 and SHP-2 in epithelial cells. J Biol Chem 274: 335–344.
Kirshner J, Chen CJ, Liu P, Huang J, Shively JE . (2003). CEACAM1-4S, a cell-cell adhesion molecule, mediates apoptosis and reverts mammary carcinoma cells to a normal morphogenic phenotype in a 3D culture. Proc Natl Acad Sci USA 100: 521–526.
Kunath T, Ordonez-Garcia C, Turbide C, Beauchemin N . (1995). Inhibition of colonic tumor cell growth by biliary glycoprotein. Oncogene 11: 2375–2382.
Liu K, Abrams SI . (2003). Coordinate regulation of IFN consensus sequence-binding protein and caspase-1 in the sensitization of human colon carcinoma cells to Fas-mediated apoptosis by IFN-gamma. J Immunol 170: 6329–6337.
Neumaier M, Paululat S, Chan A, Matthaes P, Wagener C . (1993a). Biliary glycoprotein, a potential human cell adhesion molecule, is down-regulated in colorectal carcinomas. Proc Natl Acad Sci USA 90: 10744–10748.
Neumaier M, Paululat S, Chan A, Matthaes P, Wagener C, Gaida FJ et al. (1993b). Biliary glycoprotein, a potential human cell adhesion molecule, is down-regulated in colorectal carcinomas. Proc Natl Acad Sci USA 90: 10744–10748.
Nittka S, Gunther J, Ebisch C, Erbersdobler A, Neumaier M . (2004). The human tumor suppressor CEACAM1 modulates apoptosis and is implicated in early colorectal tumorigenesis. Oncogene 23: 9306–9313.
Nollau P, Prall F, Helmchen U, Wagener C, Neumaier M . (1997a). Dysregulation of carcinoembryonic antigen group members CGM2, CD66a (biliary glycoprotein), and nonspecific cross-reacting antigen in colorectal carcinomas. Comparative analysis by northern blot and in situ hybridization. Am J Pathol 151: 521–530.
Nollau P, Scheller H, Kona-Horstmann M, Rohde S, Hagenmuller F, Wagener C et al. (1997b). Expression of CD66a (human C-CAM) and other members of the carcinoembryonic antigen gene family of adhesion molecules in human colorectal adenomas. Cancer Res 57: 2354–2357.
Obrink B . (1997). CEA adhesion molecules: multifunctional proteins with signal-regulatory properties. Curr Opin Cell Biol 9: 616–626.
Paxton RJ, Mooser G, Pande H, Lee TD, Shively JE . (1987). Sequence analysis of carcinoembryonic antigen: identification of glycosylation sites and homology with the immunoglobulin supergene family. Proc Natl Acad Sci USA 84: 920–924.
Roncucci L, Stamp D, Medline A, Cullen JB, Bruce WR . (1991). Identification and quantification of aberrant crypt foci and microadenomas in the human colon. Hum Pathol 22: 287–294.
Singer BB, Scheffrahn I, Obrink B . (2000). The tumor growth-inhibiting cell adhesion molecule CEACAM1 (C-CAM) is differently expressed in proliferating and quiescent epithelial cells and regulates cell proliferation. Cancer Res 60: 1236–1244.
Vancompernolle K, Van Herreweghe F, Pynaert G, Van de Craen M, De Vos K, Totty N et al. (1998). Atractyloside-induced release of cathepsin B, a protease with caspase-processing activity. FEBS Lett 438: 150–158.
Watt SM, Teixeira AM, Zhou GQ, Doyonnas R, Zhang Y, Grunert F et al. (2001). Homophilic adhesion of human CEACAM1 involves N-terminal domain interactions: structural analysis of the binding site. Blood 98: 1469–1479.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Nittka, S., Böhm, C., Zentgraf, H. et al. The CEACAM1-mediated apoptosis pathway is activated by CEA and triggers dual cleavage of CEACAM1. Oncogene 27, 3721–3728 (2008). https://doi.org/10.1038/sj.onc.1211033
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1211033
Keywords
This article is cited by
-
TIM-3 drives temporal differences in restimulation-induced cell death sensitivity in effector CD8+ T cells in conjunction with CEACAM1
Cell Death & Disease (2021)
-
Elevated serum CEA levels are associated with the explosive progression of lung adenocarcinoma harboring EGFR mutations
BMC Cancer (2017)
-
Blood-based lung cancer biomarkers identified through proteomic discovery in cancer tissues, cell lines and conditioned medium
Clinical Proteomics (2015)
-
Clinical and experimental studies regarding the expression and diagnostic value of carcinoembryonic antigen-related cell adhesion molecule 1 in non-small-cell lung cancer
BMC Cancer (2013)
-
CEA is an Independent Prognostic Indicator that is Associated with Reduced Survival and Liver Metastases in SCLC
Cell Biochemistry and Biophysics (2011)