Abstract
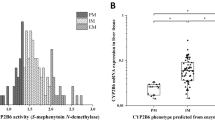
We investigated whether polymorphisms in the CYP2D6 and CYP2C19 genes influence the metabolic ratios and enantiomeric S/R ratios of venlafaxine (VEN) and its metabolites O-desmethylvenlafaxine (ODV), N-desmethylvenlafaxine (NDV) and N,O-didesmethylvenlafaxine (DDV) in blood from forensic autopsy cases. In all, 94 postmortem cases found positive for VEN during toxicological screening were included. The CYP2D6 genotype was shown to significantly influence the ODV/VEN (P=0.003), DDV/NDV (P=0.010) and DDV/ODV (P=0.034) ratios. The DDV/ODV (P=0.013) and DDV/VEN (P=0.021) ratios were significantly influenced by the CYP2C19 genotype. The S/R ratios of VEN were significantly influenced by both CYP2D6 and CYP2C19 genotypes. CYP2D6 poor metabolizers (PMs) had lower S/R VEN ratios and CYP2C19 PMs had high S/R ratios of VEN in comparison. Our results show that the CYP2D6 genotype influences the O-demethylation whereas CYP2C19 influences the N-demethylation of VEN and its metabolites. In addition, we show a stereoselective metabolism where CYP2D6 favours the R-enantiomer whereas CYP2C19 favours the S-enantiomer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ingelman-Sundberg M, Sim SC, Gomez A, Rodriguez-Antona C . Influence of cytochrome P450 polymorphisms on drug therapies: pharmacogenetic, pharmacoepigenetic and clinical aspects. Pharmacol Ther 2007; 116: 496–526.
Bertilsson L, Dahl ML, Dalen P, Al-Shurbaji A . Molecular genetics of CYP2D6: clinical relevance with focus on psychotropic drugs. Br J Clin Pharmacol 2002; 53: 111–122.
Goldstein JA . Clinical relevance of genetic polymorphisms in the human CYP2C subfamily. Br J Clin Pharmacol 2001; 52: 349–355.
Kirchheiner J, Seeringer A . Clinical implications of pharmacogenetics of cytochrome P450 drug metabolizing enzymes. Biochim Biophys Acta 2007; 1770: 489–494.
Holmgren P, Carlsson B, Zackrisson AL, Lindblom B, Dahl ML, Scordo MG et al. Enantioselective analysis of citalopram and its metabolites in postmortem blood and genotyping for CYD2D6 and CYP2C19. J Anal Toxicol 2004; 28: 94–104.
Kingback M, Karlsson L, Zackrisson AL, Carlsson B, Josefsson M, Bengtsson F et al. Influence of CYP2D6 genotype on the disposition of the enantiomers of venlafaxine and its major metabolites in postmortem femoral blood. Forensic Sci Int 2012; 214: 124–134.
Koski A, Sistonen J, Ojanpera I, Gergov M, Vuori E, Sajantila A . CYP2D6 and CYP2C19 genotypes and amitriptyline metabolite ratios in a series of medicolegal autopsies. Forensic Sci Int 2006; 158: 177–183.
Levo A, Koski A, Ojanpera I, Vuori E, Sajantila A . Post-mortem SNP analysis of CYP2D6 gene reveals correlation between genotype and opioid drug (tramadol) metabolite ratios in blood. Forensic Sci Int 2003; 135: 9–15.
Zackrisson AL, Holmgren P, Gladh AB, Ahlner J, Lindblom B . Fatal intoxication cases: cytochrome P450 2D6 and 2C19 genotype distributions. Eur J Clin Pharmacol 2004; 60: 547–552.
Zackrisson AL, Lindblom B, Ahlner J . High frequency of occurrence of CYP2D6 gene duplication/multiduplication indicating ultrarapid metabolism among suicide cases. Clin Pharmacol Ther 2010; 88: 354–359.
Garcia-Quetglas E, Azanza JR, Sadaba B, Munoz MJ, Gil I, Campanero MA . Pharmacokinetics of tramadol enantiomers and their respective phase I metabolites in relation to CYP2D6 phenotype. Pharmacol Res 2007; 55: 122–130.
Dahl ML, Tybring G, Elwin CE, Alm C, Andreasson K, Gyllenpalm M et al. Stereoselective disposition of mianserin is related to debrisoquin hydroxylation polymorphism. Clin Pharmacol Ther 1994; 56: 176–183.
Begre S, von Bardeleben U, Ladewig D, Jaquet-Rochat S, Cosendai-Savary L, Golay KP et al. Paroxetine increases steady-state concentrations of (R)-methadone in CYP2D6 extensive but not poor metabolizers. J Clin Psychopharmacol 2002; 22: 211–215.
Ellingrod VL, Perry PJ . Venlafaxine: a heterocyclic antidepressant. Am J Hosp Pharm 1994; 51: 3033–3046.
Holliday SM, Benfield P . Venlafaxine. A review of its pharmacology and therapeutic potential in depression. Drugs 1995; 49: 280–294.
Muth EA, Haskins JT, Moyer JA, Husbands GE, Nielsen ST, Sigg EB . Antidepressant biochemical profile of the novel bicyclic compound Wy-45030, an ethyl cyclohexanol derivative. Biochem Pharmacol 1986; 35: 4493–4497.
Fogelman SM, Schmider J, Venkatakrishnan K, von Moltke LL, Harmatz JS, Shader RI et al. O- and N-demethylation of venlafaxine in vitro by human liver microsomes and by microsomes from cDNA-transfected cells: effect of metabolic inhibitors and SSRI antidepressants. Neuropsychopharmacology 1999; 20: 480–490.
Otton SV, Ball SE, Cheung SW, Inaba T, Rudolph RL, Sellers EM . Venlafaxine oxidation in vitro is catalysed by CYP2D6. Br J Clin Pharmacol 1996; 41: 149–156.
Sangkuhl K, Stingl JC, Turpeinen M, Altman RB, Klein TE . PharmGKB summary: venlafaxine pathway. Pharmacogenet Genomics 2014; 24: 62–72.
Lessard E, Yessine MA, Hamelin BA, O'Hara G, LeBlanc J, Turgeon J . Influence of CYP2D6 activity on the disposition and cardiovascular toxicity of the antidepressant agent venlafaxine in humans. Pharmacogenetics 1999; 9: 435–443.
McAlpine DE, Biernacka JM, Mrazek DA, O'Kane DJ, Stevens SR, Langman LJ et al. Effect of cytochrome P450 enzyme polymorphisms on pharmacokinetics of venlafaxine. Ther Drug Monit 2011; 33: 14–20.
Eap CB, Lessard E, Baumann P, Brawand-Amey M, Yessine M-A, ÒHara G et al. Role of CYP2D6 in the stereoselective disposition of venlafaxine in humans. Pharmacogenetics 2003; 13: 39–47.
Hermann M, Hendset M, Fosaas K, Hjerpset M, Refsum H . Serum concentrations of venlafaxine and its metabolites O-desmethylvenlafaxine and N-desmethylvenlafaxine in heterozygous carriers of the CYP2D6*3, *4 or *5 allele. Eur J Clin Pharmacol 2008; 64: 483–487.
Shams ME, Arneth B, Hiemke C, Dragicevic A, Muller MJ, Kaiser R et al. CYP2D6 polymorphism and clinical effect of the antidepressant venlafaxine. J Clin Pharm Ther 2006; 31: 493–502.
van der Weide J, van Baalen-Benedek EH, Kootstra-Ros JE . Metabolic ratios of psychotropics as indication of cytochrome P450 2D6/2C19 genotype. Ther Drug Monit 2005; 27: 478–483.
Veefkind AH, Haffmans PM, Hoencamp E . Venlafaxine serum levels and CYP2D6 genotype. Ther Drug Monit 2000; 22: 202–208.
Kingback M, Josefsson M, Karlsson L, Ahlner J, Bengtsson F, Kugelberg FC et al. Stereoselective determination of venlafaxine and its three demethylated metabolites in human plasma and whole blood by liquid chromatography with electrospray tandem mass spectrometric detection and solid phase extraction. J Pharm Biomed Anal 2010; 53: 583–590.
Zackrisson AL, Lindblom B . Identification of CYP2D6 alleles by single nucleotide polymorphism analysis using pyrosequencing. Eur J Clin Pharmacol 2003; 59: 521–526.
Howell SR, Husbands GE, Scatina JA, Sisenwine SF . Metabolic disposition of 14C-venlafaxine in mouse, rat, dog, rhesus monkey and man. Xenobiotica 1993; 23: 349–359.
Sproule BA, Hazra M, Pollock BG . Desvenlafaxine succinate for major depressive disorder. Drugs Today (Barc) 2008; 44: 475–487.
Gerber JG, Rhodes RJ, Gal J . Stereoselective metabolism of methadone N-demethylation by cytochrome P4502B6 and 2C19. Chirality 2004; 16: 36–44.
Tybring G, Bottiger Y, Widen J, Bertilsson L . Enantioselective hydroxylation of omeprazole catalyzed by CYP2C19 in Swedish white subjects. Clin Pharmacol Ther 1997; 62: 129–137.
Smith SW . Chiral toxicology: it's the same thing...only different. Toxicol Sci 2009; 110: 4–30.
Jaffe PD, Batziris HP, van der Hoeven P, DeSilva D, McIntyre IM . A study involving venlafaxine overdoses: compararsion of fatal and therapeutic concentrations in postmortem specimens. J Forensic Sci 1999; 44: 193–196.
Baumann P, Hiemke C, Ulrich S, Eckermann G, Gaertner I, Gerlach M et al. The AGNP-TDM expert group consensus guidelines: therapeutic drug monitoring in psychiatry. Pharmacopsychiatry 2004; 37: 243–265.
Hiemke C, Baumann P, Bergemann N, Conca A, Dietmaier O, Egberts K et al. AGNP consensus guidelines for therapeutic drug monitoring in psychiatry: update 2011. Pharmacopsychiatry 2011; 44: 195–235.
Acknowledgements
This work was supported by grants from the Swedish Research Council, the Lions Research Foundation and the National Board of Forensic Medicine in Sweden.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website
Supplementary information
Rights and permissions
About this article
Cite this article
Karlsson, L., Zackrisson, AL., Josefsson, M. et al. Influence of CYP2D6 and CYP2C19 genotypes on venlafaxine metabolic ratios and stereoselective metabolism in forensic autopsy cases. Pharmacogenomics J 15, 165–171 (2015). https://doi.org/10.1038/tpj.2014.50
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2014.50
This article is cited by
-
Age of onset for increased dose-adjusted serum concentrations of antidepressants and association with sex and genotype: An observational study of 34,777 individuals
European Journal of Clinical Pharmacology (2024)
-
Pharmacokinetic correlates of venlafaxine: associated adverse reactions
European Archives of Psychiatry and Clinical Neuroscience (2019)
-
Full-gene haplotypes refine CYP2D6 metabolizer phenotype inferences
International Journal of Legal Medicine (2018)
-
Effects of the Proton Pump Inhibitors Omeprazole and Pantoprazole on the Cytochrome P450-Mediated Metabolism of Venlafaxine
Clinical Pharmacokinetics (2018)