Abstract
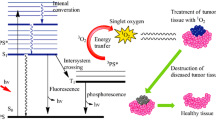
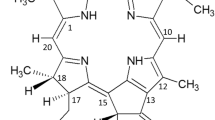
Photodynamic therapy (PDT) is an alternative anticancer treatment in which direct tumor-cell killing results from selective accumulation of photosensitizers in the tumor sites and phototoxicity occurs when light-activated photosensitizers transfer the energy to oxygen nearby to produce singlet oxygen. The objective of this study was to investigate the effects of PDT using chlorophyll derivatives such as pheophytin a (phe a), pheophytin b (phe b), pheophorbide a (pho a) and pheophorbide b (pho b) as the photosensitizers, and the 660 nm light-emitting diodes (LEDs) irradiation on human hepatocellular carcinoma cells (HuH-7). The drug concentration-dependent inhibition of HuH-7 cell viability was studied under LEDs irradiation (10 mW cm−2) at radiant exposure of 5.1 and 10.2 J cm−2 by MTT assay. Significant inhibition of the survival of HuH-7 cells (<10%) was observed when an irradiation dose of 10.2 J cm−2 combined with the concentration of 0.5 μg ml−1 of phe a, 0.125 μg ml−1 of pho a, 0.25 μg ml−1 of phe b, and 0.125 μg ml−1 of pho b were applied. The results from Annexin V—propidium iodide staining revealed that phe a, phe b, pho a and pho b could induce cell death in HuH-7 cells predominantly via a necrotic process. The results from immunoblot analyses exhibited that chlorophyll derivative-mediated PDT initiated cytochrome c release, caspase-9 and caspase-3 activation, followed by poly ADP-ribose polymerase (PARP) cleavage. Thus, apoptosis also occurred in HuH-7 cells after PDT treatment, and the execution of the apoptotic process may be initiated from the loss of mitochondrial function. Our findings demonstrate that both apoptosis and necrosis can be induced in HuH-7 cells after PDT using phe a, phe b, pho a and pho b and LEDs.
Similar content being viewed by others
References
I. H. McKillop, D. M. Moran, X. Jin, L. G. Koniaris, Molecular pathogenesis of hepatocellular carcinoma, J. Surg. Res., 2006, 136, 125–135.
H. E. Blum, Treatment of hepatocellular carcinoma, Best Pract. Res. Clin. Gastroenterol., 2005, 19, 129–145.
D. E. J. G. J. Dolmans, D. Fukumura, R. K. Jain, Photodynamic therapy for cancer, Nat. Rev. Cancer, 2003, 3, 380–387.
T. S. Mang, Lasers and light sources for PDT: past, present and future, Photodiagn. Photodyn. Ther., 2004, 1, 43–48.
M. Otake, M. Nishiwaki, Y. Kobayashi, S. Baba, E. Kohno, T. Kawasaki, Y. Fujise, H. Nakamura, Selective accumulation of ALA-induced PpIX and photodynamic effect in chemically induced hepatocellular carcinoma, Br. J. Cancer, 2003, 89, 730–736.
C. M. N. Yow, C. K. Wong, Z. Huang, R. J. Ho, Study of the efficacy and mechanism of ALA-mediated photodynamic therapy on human hepatocellular carcinoma cell, Liver Int., 2007, 27, 201–207.
V. Vonarx-Coinsman, M.-T. Foultier, L. X. de Brito, L. Morlet, A. Gouyette, T. Patrice, HepG2 human hepatocarcinoma cells: an experimental model for photosensitization by endogenous porphyrins, J. Photochem. Photobiol., 1995, 30, 201–208.
N. G. Egger, J. A. Schoenecker, Jr., W. K. Gourley, M. Motamedi, K. E. Anderson, S. A. Weinman, Photosensitization of experimental hepatocellular carcinoma with protoporhyrin synthesized from administered d-aminolevulinic acid: studies with cultured cells and implanted tumors, J. Hepatol., 1997, 26, 913–920.
J. Nakamura, H. Kajiwara, Photodynamic therapy using mono-l-aspartyl chlorin e6 for rabbit experimental hepatoma, J. Hepatobil. Pancreat. Surg., 1999, 6, 312–319.
M. Date, K. Fukuchi, Y. Namiki, A. Okumura, S. Morita, H. Takahashi, K. Ohura, Therapeutic effect of photodynamic therapy using PAD-S31 and diode laser on human liver cancer cells, Liver Int., 2004, 24, 142–148.
J. C. Lai, P.-C. Lo, D. K. P. Ng, W.-H. Ko, S. C. H. Leung, K.-P. Fung, W.-P. Fong, BAM-SiPc, a novel agent for photodynamic therapy, induces apoptosis in human hepatocarcinoma HepG2 by a direct mitochondrial action, Cancer Biol. Ther., 2006, 5, 413–418.
C.-Y. Hsu, C.-M. Yang, C.-M. Chen, P.-Y. Chao, S.-P. Hu, Effects of chlorophyll-related compounds on hydrogen peroxide induced DNA damage within human lymphocytes, J. Agric. Food Chem., 2005, 53, 2746–2750.
D.-Y. Xu, Research and development of photodynamic therapy photosensitizers in China, Photodiagn. Photodyn Ther., 2007, 4, 13–25.
C.-M. Yang, K.-W. Chang, M.-H. Yin, H.-M. Huang, Methods for the determination of the chlorophylls and their derivatives, Taiwania, 1998, 43, 116–122.
S. M. Srinivasula, M. Ahmad, T. Fernandes-Alnemri, E. S. Alnemri, Autoactivation of procaspase-9 by Apaf-1-mediated oligomerization, Mol. Cell, 1998, 1, 949–957.
H. Zou, R. Yang, J. Hao, J. Wang, C. Sun, S. W. Fesik, J. C. Wu, K. J. Tomaselli, R. C. Armstrong, Regulation of the Apaf-1/caspase-9 apoptosom by caspase-3 and XIAP, J. Biol. Chem., 278, 8091–8998.
M. R. Detty, S. L. Gibson, S. J. Wagner, Current clinical and preclinical photosensitizers for use in photodynamic therapy, J. Med. Chem., 2004, 47, 3897–3915.
W.-Y. Lee, D.-S. Lim, S.-H. Ko, Y.-J. Park, K.-S. Ryu, M.-Y. Ahn, Y.-R. Kim, D.-W. Lee, C.-W. Cho, Photoactivation of pheophorbide a induces a mitochondrial-mediated apoptosis in Jurkat leukemia cells, J. Photochem. Photobiol., 2004, 75, 119–126.
D.-S. Lim, S.-H. Ko, W.- Y. Lee, Silkworm-pheophorbide a mediated photodynamic therapy against B16F10 pigmented melanoma, J. Photochem. Photobiol., 2004, 74, 1–16.
R. K. Pandey, L. N. Goswami, Y. Chen, A. Gryshuk, J. R. Missert, A. Oseroff, T. J. Dougherty, Nature: a rich source for developing multifunctional agents. Tumor-imaging and photodynamic therapy, Lasers Surg. Med., 2006, 38, 445–467.
A. Brandis, O. Mazor, E. Neumark, V. Rosenbach-Belkin, Y. Salomon, A. Scherz, Novel water-soluble bacteriochlorophyll derivatives for vascular-targeted photodynamic therapy: synthesis, solubility, phototoxicity and the effect of serum proteins, Photochem. Photobiol., 2005, 81, 983–993.
Z. Huang, Q. Chen, D. Luck, J. Beckers, B. C. Wilson, N. Trncic, S. M. Larue, D. Blanc, F. W. Hetzel, Studies of a vascular-acting photosensitizer, Pd-bacteriopheophorbide, Lasers Surg. Med., 2005, 36, 390–397.
A. Hajri, S. Wack, C. Meyer, M. K. Smith, C. Leberquier, M. Kedinger, M. Aprahamian, In vitro and in vivo efficacy of Photofrin® and pheophorbide a, a bacteriochlorin, in photodynamic therapy of colonic cancer cells, Photchem. Photobiol., 2002, 75, 140–148.
P. M.-K. Tang, J. Y.-W. Chan, S. W.-N. Au, S.-K. Kong, S. K.-W. Tsui, M. M.-Y. Waye, T. C.-W. Mak, W.-P. Fong, K.-P. Fung, Pheophorbide a, an active compound isolated from Scutellaria barbata, possess photodynamic activities by inducing apoptosis in human hepatocellular carcinoma, Cancer Biol. Ther., 2006, 5, 1111–1116.
L. Wyld, M. W. R. Reed, N. J. Brown, Differential cell death response to photodynamic therapy is dependent on dose and cell type, Br. J. Cancer, 2001, 84, 1384–1386.
A. P. Castano, T. N. Demidova, M. R. Hamblin, Mechanisms in photodynamic therapy: part two-cellular signalling, cell metabolism and modes of cell death, Photodiagn. Photodyn. Ther., 2005, 2, 1–23.
Z. Jin, W. S. El-Deiry, Overview of cell death signaling pathways, Cancer Biol. Ther., 2005, 4, 139–163.
A. Vantieghem, Y. Xu, Z. Assefa, J. Piette, J. r. Vandenheede, W. Merlevede, P. A. M. de Witte, P. Agostinis, Phosphoyrlation of Bcl-2 in G2/M phase-arrested cells following photodynamic therapy with hypericin involves a CDK1-mediated signal and delays the onset of apoptosis, J. Biol. Chem., 2002, 277, 37718–37731.
D. Kessel, M. G. H. Vicente, J. J. Reiners, Jr., Initiation of apoptosis and autophagy by photodynamic therapy, Lasers Surg. Med., 2006, 38, 482–488.
J. Mikeš, J. Kleban, V. Sacková, V. Horváth, E. Jamborová, A. Vaculová, A. Kozubík, J. Hofmanová, P. Fedorocko, Necrosis predominates in the cell death of human colon adenocarcinoma HT-29 cells treated under variable conditions of photodynamic therapy with hypericin, Photochem. Photobiol. Sci., 2007, 6, 758–766.
Y.-J. Hsieh, C.-C. Wu, C.-J. Chang, J.-S. Yu, Subcellular localization of Photofrin determines the death phenotype of human epidermoid carcinoma A431 cells triggers by photodynamic therapy: when plasma membranes are the main targets, J. Cell. Physiol., 2003, 194, 363–375.
K. Plaetzer, T. Kiesslich, B. Krammer, P. Hammerl, Characterization of the cell death modes and the associated changes in cellular energy supply in response to AlPcS4-PDT, Photochem. Photobiol. Sci., 2002, 1, 172–177.
Z. Tong, G. Singh, A. J. Rainbow, The role of the p53 tumor suppressor in the response of human cells to photofrin-mediated photodynamic therapy, Photochem. Photobiol., 2000, 71, 201–210.
M. Barberi-Heyob, P.-O. Védrine, J.-L. Merlin, R. Millon, J. Abecassis, M.-F. Poupon, F. Guillemin, Wild-type p53 gene transfer into mutated p53 HT29 cells improves sensitivity to photodynamic therapy via induction of apoptosis, Int. J. Oncol., 2004, 24, 951–958.
V. Heinzelmann-Schwarz, A. Fedier, R. Hornung, H. Walt, U. Haller, D. Fink, Role of p53 and ATM in photodynamic therapy-induced apoptosis, Lasers Surg. Med., 2003, 33, 182–189.
G. Feng, N. Kaplowitz, Mechanism of staurosporine-induced apoptosis in murine hepatocytes, Am. J. Physiol. Gastrointest. Liver Physiol., 2002, 282, G825–G834.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, WT., Tsao, HW., Chen, YY. et al. A study on the photodynamic properties of chlorophyll derivatives using human hepatocellular carcinoma cells. Photochem Photobiol Sci 6, 1341–1348 (2007). https://doi.org/10.1039/b704539e
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/b704539e