Abstract
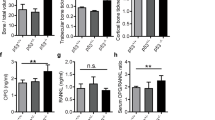
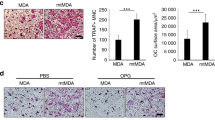
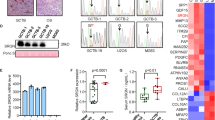
Local growth of osteosarcoma involves destruction of host bone by proteolytic mechanisms and/or host osteoclast activation. Osteoclast formation and activity are regulated by osteoblast-derived factors such as the osteoclast differentiating factor, receptor activator of NF-κB ligand (RANKL) and the inhibitor osteoprotegerin (OPG). We have investigated the in vitro effects of bisphosphonates on a clonal rat osteosarcoma cell line. The aminobisphosphonate pamidronate was added to UMR 106-01 cell cultures (10−8M to 10−4M up to 5 days). The non-aminobisphosphonate clodronate was administered for the same time periods (10−6M to 10−2M). Cell proliferation, apoptosis and mRNA expression was assessed. Both agents inhibited cell proliferation in a time- and dose-dependent manner. ELISA analysis demonstrated an increase in DNA fragmentation although there was no significant dose-related difference between the doses studied. Bisphosphonate-treated cultures had a greater subpopulation of cells exhibiting morphological changes of apoptosis. Expression of mRNA for osteopontin and RANKL was down-regulated by both agents, while the expression of mRNA for alkaline phosphatase, pro-α1(I) collagen and OPG was not altered. Out in vitro work suggests the bisphosphonates not only have direct effects on osteosarcoma cell growth and apoptosis, but also, by altering the relative expression of osteoclast-regulating factors, they may inhibit the activity of osteoclasts and their recruitment. © 2001 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Adami S (1997) Bisphosphonates in prostate carcinoma. Cancer 80: 1674–1679
Arends MJ, Morris RG and Wyllie AH (1990) Apoptosis. The role of the endonuclease. Am J Pathol 136: 593–608
Asou Y, Rittling SR, Yoshitake H, Denhardt DT and Noda M (1999) Osteopontin-deficient bone is defective in angiogenesis, osteoclast-recruitment and ectopic resorption. Proceedings from the 21st Annual Meeting of the American Society for Bone and Mineral Research,
Behrend EI, Craig AM, Wilson SM, Denhardt DT and Chambers AF (1994) Reduced malignancy of ras-transformed NIH 3T3 cells expressing antisense osteopontin RNA. Cancer Res 54: 832–837
Behrend EI, Craig AM, Wilson SM, Denhardt DT and Chambers AF (1995) Expression of antisense osteopontin RNA in metastatic mouse fibroblasts is associated with reduced malignancy. Ann N Y Acad Sci 760: 299–301
Bloomfield DJ (1998) Should bisphosphonates be part of the standard therapy of patients with multiple myeloma or bone metastases from other cancers? An evidence-based review. J Clin Oncol 16: 1218–1225
Boissier S, Magnetto S, Frappart L, Cuzin B, Ebetino FH, Delmas PD and Clezardin P (1997) Bisphosphonates inhibit prostate and breast carcinoma cell adhesion to unmineralized and mineralized bone extracellular matrices. Cancer Res 57: 3890–3894
Brown LF, Papadopoulos Sergiou A, Berse B, Manseau EJ, Tognazzi K, Perruzzi CA, Dvorak HF and Senger DR (1994) Osteopontin expression and distribution in human carcinomas. Am J Pathol 145: 610–623
Butle WT (1989) The nature and significance of osteopontin. Connect Tissue Res 23: 123–136
Chan YL, Gutell R, Noller HF and Wool IG (1984) The nucleotide sequence of a rat 18 S ribosomal ribonucleic acid gene and a proposal for the secondary structure of 18 S ribosomal ribonucleic acid. J Biol Chem 259: 224–230
Chikatsu N, Takeuchi Y, Tamura Y, Fukumoto S, Yano K, Tsuda E, Ogata E and Fujita T (2000) Interactions between cancer and bone marrow cells induce osteoclast differentiation factor expression and osteoclast-like cell formation in vitro. Biochem Biophys Res Commun 267: 632–637
Coleman RE and Purohit OP (1993) Osteoclast inhibition for the treatment of bone metastases. Cancer Treat Rev 19: 79–103
Craig AM, Nemir M, Mukherjee BB, Chambers AF and Denhardt DT (1988) Identification of the major phosphoprotein secreted by many rodent cell lines as 2ar/osteopontin: enhanced expression in H-ras-transformed 3T3 cells. Biochem Biophys Res Commun 157: 166–173
Denhardt DT and Guo X (1993) Osteopontin: a protein with diverse functions. Faseb J 7: 1475–1482
Diel IJ, Solomayer EF, Costa SD, Gollan C, Goerner R, Wallwiener D, Kaufmann M and Bastert G (1998) Reduction in new metastases in breast cancer with adjuvant clodronate treatment. N Engl J Med 339: 357–363
Dodds RA, Connor JR, James IE, Rykaczewski EL, Appelbaum E, Dul E and Gowen M (1995) Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone. J Bone Miner Res 10: 1666–1680
Evans CE and Braidman IP (1994) Effects of two novel bisphosphonates on bone cells in vitro. Bone Miner 26: 95–107
Flanagan AM and Chambers TJ (1989) Dichloromethylenebisphosphonate (C12MBP) inhibits bone resorption through injury to osteoclasts that resorb C12MBP-coated bone. Bone Miner 6: 33–43
Fleisch H (1993) New bisphosphonates in osteoporosis. Osteoporos Int 3 Suppl 2: S15–22
Galasko CS (1976) Mechanisms of bone destruction in the development of skeletal metastases. Nature 263: 507–508
García Moreno C, Serrano S, Nacher M, Farré M, A, DI, Mariñoso, M, A, DI Carbonell J, Mellibovsky L, Nogués X, Ballester J and Aubía J (1998) Effect of alendronate on cultured normal human osteoblasts. Bone 22: 233–239
Gardner HA, Berse B and Senger DR (1994) Specific reduction in osteopontin synthesis by antisense RNA inhibits the tumorigenicity of transformed Rat 1 fibroblasts. Oncogene 9: 2321–2326
Goziotis A, Sukhu B, Torontali M, Dowhaniuk M and Tenenbaum HC (1995) Effects of bisphosphonates APD and HEBP on bone metabolism in vitro. Bone 16: 317S–327S
Helfrich MH, Nesbitt SA, Dorey EL and Horton MA (1992) Rat osteoclasts adhere to a wide range of RGD (Arg-Gly-Asp) peptide-containing proteins, including the bone sialoproteins and fibronectin, via a beta 3 integrin. J Bone Miner Res 7: 335–343
Horwood NJ, Elliott J, Martin TJ and Gillespie MT (1998) Osteotropic agents regulate the expression of osteoclast differentiation factor and osteoprotegerin in osteoblastic stromal cells. Endocrinology 139: 4743–4746
Jilka RL, Weinstein RS, Bellido T, Parfitt AM and Manolagas SC (1998) Osteoblast programmed cell death (apoptosis): modulation by growth factors and cytokines. J Bone Miner Res 13: 793–802
Kartsogiannis V, Zhou H, Horwood NJ, Thomas RJ, Hards DK, Quinn JMW, Niforas P, Ng KW, Martin TJ and Gillespie MT (1999) Localization of RANKL (Receptor Activator of NFKB Ligand) mRNA and Protein in Skeletal and Extraskeletal Tissues. Bone 25: 525–534
Luckman SP, Hughes DE, Coxon FP, Graham R, Russell G and Rogers MJ (1998) Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of GTP-binding proteins, including Ras. J Bone Miner Res 13: 581–589
Majeska RJ, Port M and Einhorn TA (1993) Attachment to extracellular matrix molecules by cells differing in the expression of osteoblastic traits. J Bone Miner Res 8: 277–289
Martin TJ, Ingleton PM, Underwood JC, Michelangeli VP, Hunt NH and Melick RA (1976) Parathyroid hormone-responsive adenylate cyclase in induced transplantable osteogenic rat sarcoma. Nature 260: 436–438
Merry K, Dodds R, Littlewood A and Gowen M (1993) Expression of osteopontin mRNA by osteoclasts and osteoblasts in modelling adult human bone. J Cell Sci 104: 1013–1020
Oldberg A, Franzén A and Heinegård D (1986) Cloning and sequence analysis of rat bone sialoprotein (osteopontin) cDNA reveals an Arg-Gly-Asp cell-binding sequence. Proc Natl Acad Sci USA 83: 8819–8823
Orr FW, Sanchez-Sweatman OH, Kostenuik P and Singh G (1995) Tumor-bone interactions in skeletal metastasis. Clin Orthop, 19–33
Plotkin LI, Weinstein RS, Parfitt AM, Roberson PK, Manolagas SC and Bellido T (1999) Prevention of osteocyte and osteoblast apoptosis by bisphosphonates and calcitonin. J Clin Invest 104: 1363–1374
Reinholt FP, Hultenby K, Oldberg A and Heinegard D (1990) Osteopontin – a possible anchor of osteoclasts to bone. Proc Natl Acad Sci USA 87: 4473–4475
Rogers MJ, Gordon S, Benford HL, Coxon FP, Luckman SP, Monkkonen J and Frith JC (2000) Cellular and molecular mechanisms of action of bisphosphonates. Cancer 88: 2989–2994
Ross FP, Chappel J, Alvarez JI, Sander D, Butler WT, Farach-Carson MC, Mintz KA, Robey PG, Teitelbaum SL and Cheresh DA (1993) Interactions between the bone matrix proteins osteopontin and bone sialoprotein and the osteoclast integrin alpha v beta 3 potentiate bone resorption. J Biol Chem 268: 9901–9907.
Russell RG, Rogers MJ, Frith JC, Luckman SP, Coxon FP, Benford HL, Croucher PI, Shipman C and Fleisch HA (1999) The pharmacology of bisphosphonates and new insights into their mechanisms of action. J Bone Miner Res 14: 53–65
Sahni M, Guenther HL, Fleisch H, Collin P and Martin TJ (1993) Bisphosphonates act on rat bone resorption through the mediation of osteoblasts. J Clin Invest 91: 2004–2011
Sato M, Grasser W, Endo N, Akins R, Simmons H, Thompson DD, Golub E and Rodan GA (1991) Bisphosphonate action. Alendronate localization in rat bone and effects on osteoclast ultrastructure. J Clin Invest 88: 2095–2105
Senaratne SG, Pirianov G, Mansi JL, Arnett TR and Colston KW (2000) Bisphosphonates induce apoptosis in human breast cancer cell lines. Br J Cancer 82: 1459–1468
Shipman CM, Rogers MJ, Apperley JF, Russell RG and Croucher PI (1997) Bisphosphonates induce apoptosis in human myeloma cell lines: a novel anti-tumour activity. Br J Haematol 98: 665–672
Simonet WS, Lacey DL, Dunstan CR, Kelley M, Chang MS, Luthy R, Nguyen HQ, Wooden S, Bennett L, Boone T, Shimamoto G, DeRose M, Elliott R, Colombero A, Tan HL, Trail G, Sullivan J, Davy E, Bucay N, Renshaw-Gegg L, Hughes TM, Hill D, Pattison W, Campbell P and Boyle WJ (1997) Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89: 309–319
Sodek J, Chen J, Nagata T, Kasugai S, Todescan R Jr, Li IW and Kim RH (1995) Regulation of osteopontin expression in osteoblasts. Ann N Y Acad Sci 760: 223–241
Tan PL, Katz JM, Ames R, Caughey DE, Gray HD, Ibbertson HK and Watson JD (1988) Aminobisphosphonate inhibition of interleukin-1-induced bone resorption in mouse calvariae. Arthritis Rheum 31: 762–768
Thiébaud D, Leyvraz S, von Fliedner V, Perey L, Cornu P, Thiébaud S and Burckhardt P (1991) Treatment of bone metastases from breast cancer and myeloma with pamidronate. Eur J Cancer 27: 37–41
van der Pluijm G, Vloedgraven H, van Beek E, van der Wee- Pals L, C Lö and Papapoulos S (1996) Bisphosphonates inhibit the adhesion of breast cancer cells to bone matrices in vitro. J Clin Invest 98: 698–705
Vitté C, Fleisch H and Guenther HL (1996) Bisphosphonates induce osteoblasts to secrete an inhibitor of osteoclast-mediated resorption. Endocrinology 137: 2324–2333
Wong BR, Rho J, Arron J, Robinson E, Orlinick J, Chao M, Kalachikov S, Cayani E, Bartlett FS, 3rd Frankel WN, Lee SY and Choi Y (1997) TRANCE is a novel ligand of the tumor necrosis factor receptor family that activates c-Jun N-terminal kinase in T cells. J Biol Chem 272: 25190–25194
Yasuda H, Shima N, Nakagawa N, Mochizuki SI, Yano K, Fujise N, Sato Y, Goto M, Yamaguchi K, Kuriyama M, Kanno T, Murakami A, Tsuda E, Morinaga T and Higashio K (1998a) Identity of osteoclastogenesis inhibitory factor (OCIF) and osteoprotegerin (OPG): a mechanism by which OPG/OCIF inhibits osteoclastogenesis in vitro. Endocrinology 139: 1329–1337
Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, Tomoyasu A, Yano K, Goto M, Murakami A, Tsuda E, Morinaga T, Higashio K, Udagawa N, Takahashi N and Suda T (1998b) Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 95: 3597–3602
Yoshida H, Hayashi S, Kunisada T, Ogawa M, Nishikawa S, Okamura H, Sudo T, Shultz LD and Nishikawa S (1990) The murine mutation osteopetrosis is in the coding region of the macrophage colony stimulating factor gene. Nature 345: 442–444
Yu X, Scholler J and Foged NT (1996) Interaction between effects of parathyroid hormone and bisphosphonate on regulation of osteoclast activity by the osteoblast-like cell line UMR-106. Bone 19: 339–345
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Mackie, P., Fisher, J., Zhou, H. et al. Bisphosphonates regulate cell growth and gene expression in the UMR 106-01 clonal rat osteosarcoma cell line. Br J Cancer 84, 951–958 (2001). https://doi.org/10.1054/bjoc.2000.1679
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1679
Keywords
This article is cited by
-
The stimulatory impact of d-δ-Tocotrienol on the differentiation of murine MC3T3-E1 preosteoblasts
Molecular and Cellular Biochemistry (2019)
-
Bisphosphonates hinder osteoblastic/osteoclastic differentiation in the maxillary sinus mucosa-derived stem cells
Clinical Oral Investigations (2018)
-
Effect of low-level laser therapy on bisphosphonate-treated osteoblasts
Maxillofacial Plastic and Reconstructive Surgery (2016)
-
Role of osteopontin in osteosarcoma
Medical Oncology (2015)
-
Effect of compressive loading and incubation with clodronate on the RANKL/OPG system of human osteoblasts
Journal of Orofacial Orthopedics / Fortschritte der Kieferorthopädie (2015)