Abstract
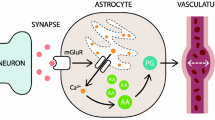
Neuronal-glial circuits underlie integrative processes in the nervous system. Function of glial syncytium is, to a very large extent, regulated by the intracellular calcium signaling system. Glial calcium signals are triggered by activation of multiple receptors, expressed in glial membrane, which regulate both Ca2+ entry and Ca2+ release from the endoplasmic reticulum. The endoplasmic reticulum also endows glial cells with intracellular excitable media, which is able to produce and maintain long-ranging signaling in a form of propagating Ca2+ waves. In pathological conditions, calcium signals regulate glial response to injury, which might have both protective and detrimental effects on the nervous tissue.
Similar content being viewed by others
Article PDF
References
Exner S . Entwurf zur physiologischen Erklärung der Psychischen Erscheinungen. Leipzig/Vienna: Deiticke; 1894.
Nedergaard M, Ransom B, Goldman SA . New roles for astrocytes: redefining the functional architecture of the brain. Trends Neurosci 2003; 26: 523–30.
Volterra A, Meldolesi J . Astrocytes, from brain glue to communication elements: the revolution continues. Nat Rev Neurosci 2005; 6: 626–40.
Bloom FE . Integration of wiring transmission and volume transmission. Prog Brain Res 2000; 125: 21–6.
Ridet I, Privat A . Volume transmission. Trends Neurosci 2000; 23: 58–9.
Sykova E, Chvatal A . Glial cells and volume transmission in the CNS. Neurochem Int 2000; 36: 397–409.
Dermietzel R . Gap junction wiring: a ‘new’ principle in cell-to-cell communication in the nervous system? Brain Res Brain Res Rev 1998; 26: 176–83.
Kettenmann H, Ransom BR Neuroglia. Oxford: OUP; 2005.
Berridge MJ . Neuronal calcium signaling. Neuron 1998; 21: 13–26.
Petersen OH, Michalak M, Verkhratsky A . Calcium signalling: past, present and future. Cell Calcium 2005; 38: 161–9.
Verkhratsky A . Calcium ions and integration in neural circuits. Acta Physiol 2006 ( in press).
Seifert G, Schilling K, Steinhauser C . Astrocyte dysfunction in neurological disorders: a molecular perspective. Nat Rev Neurosci 2006; 7: 194–206.
Berridge MJ . The endoplasmic reticulum: a multifunctional signaling organelle. Cell Calcium 2002; 32: 235–49.
Berridge MJ, Bootman MD, Roderick HL . Calcium signalling: dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol 2003; 4: 517–29.
Bootman MD, Petersen OH, Verkhratsky A . The endoplasmic reticulum is a focal point for co-ordination of cellular activity. Cell Calcium 2002; 32: 231–4.
Verkhratsky A . Physiology and pathophysiology of the calcium store in the endoplasmic reticulum of neurons. Physiol Rev 2005; 85: 201–79.
Vangheluwe P, Raeymaekers L, Dode L, Wuytack F . Modulating sarco(endo)plasmic reticulum Ca2+ ATPase 2 (SERCA2) activity: cell biological implications. Cell Calcium 2005; 38: 291–302.
Solovyova N, Veselovsky N, Toescu EC, Verkhratsky A . Ca2+ dynamics in the lumen of the endoplasmic reticulum in sensory neurons: direct visualization of Ca2+-induced Ca2+ release triggered by physiological Ca2+ entry. EMBO J 2002; 21: 622–30.
Solovyova N, Verkhratsky A . Monitoring of free calcium in the neuronal endoplasmic reticulum: an overview of modern approaches. J Neurosci Methods 2002; 122: 1–12.
Kiryushko DV, Savtchenko LP, Verkhratsky AN, Korogod SM . Theoretical estimation of the capacity of intracellular calcium stores in the Bergmann glial cell. Pflugers Arch 2002; 443: 643–51.
Hamilton SL . Ryanodine receptors. Cell Calcium 2005; 38: 253–60.
Galione A, Ruas M . NAADP receptors. Cell Calcium 2005; 38: 273–80.
Bezprozvanny I . The inositol 1,4,5-trisphosphate receptors. Cell Calcium 2005; 38: 261–72.
Burdakov D, Verkhratsky A . Biophysical re-equilibration of Ca2+ fluxes as a simple biologically plausible explanation for complex intracellular Ca2+ release patterns. FEBS Lett 2006; 380: 463–8.
Burdakov D, Petersen OH, Verkhratsky A . Intraluminal calcium as a primary regulator of endoplasmic reticulum function. Cell Calcium 2005; 38: 303–10.
Cornell Bell AH, Finkbeiner SM . Ca2+ waves in astrocytes. Cell Calcium 1991; 12: 185–204.
Cornell Bell AH, Finkbeiner SM, Cooper MS, Smith SJ . Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science 1990; 247: 470–3.
Deitmer JW, Verkhratsky AJ, Lohr C . Calcium signalling in glial cells. Cell Calcium 1998; 24: 405–16.
Verkhratsky A, Orkand RK, Kettenmann H . Glial calcium: homeostasis and signaling function. Physiol Rev 1998; 78: 99–141.
Bennett MR, Farnell L, Gibson WG . A quantitative model of purinergic junctional transmission of calcium waves in astrocyte networks. Biophys J 2005; 89: 2235–50.
Giaume C, Venance L . Intercellular calcium signaling and gap junctional communication in astrocytes. Glia 1998; 24: 50–64.
Anderson CM, Bergher JP, Swanson RA . ATP-induced ATP release from astrocytes. J Neurochem 2004; 88: 246–56.
Suadicani SO, Flores CE, Urban-Maldonado M, Beelitz M, Scemes E . Gap junction channels coordinate the propagation of intercellular Ca2+ signals generated by P2Y receptor activation. Glia 2004; 48: 217–29.
Perea G, Araque A . Glial calcium signaling and neuron-glia communication. Cell Calcium 2005; 38: 375–82.
Araque A, Parpura V, Sanzgiri RP, Haydon PG . Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci 1999; 22: 208–15.
Volterra A, Haydon P, Magistretti P Glia in synaptic transmission. Oxford: Oxford University Press; 2002.
Verkhratsky A, Kettenmann H . Calcium signalling in glial cells. Trends Neurosci 1996; 19: 346–52.
Kirischuk S, Tuschick S, Verkhratsky A, Kettenmann H . Calcium signalling in mouse Bergmann glial cells mediated by alpha 1-adrenoreceptors and H1 histamine receptors. Eur J Neurosci 1996; 8: 1198–208.
Kirischuk S, Matiash V, Kulik A, Voitenko N, Kostyuk P, Verkhratsky A, et al. Activation of P2-purino-, α1-adreno and H1-histamine receptors triggers cytoplasmic calcium signalling in cerebellar Purkinje neurons. Neuroscience 1996; 73: 643–7.
Verkhratsky A, Steinhauser C . Ion channels in glial cells. Brain Res Brain Res Rev 2000; 32: 380–412.
Lalo U, Pankratov Y, Kirchhoff F, North RA, Verkhratsky A . NMDA receptors mediate neuron-to-glia signaling in mouse cortical astrocytes. J Neurosci 2006; 26: 2673–83.
Steinhauser C, Gallo V . News on glutamate receptors in glial cells. Trends Neurosci 1996; 19: 339–45.
Seifert G, Steinhauser C . Ionotropic glutamate receptors in astrocytes. Prog Brain Res 2001; 132: 287–99.
Kirischuk S, Moller T, Voitenko N, Kettenmann H, Verkhratsky A . ATP-induced cytoplasmic calcium mobilization in Bergmann glial cells. J Neurosci 1995; 15: 7861–71.
Dani JW, Chernjavsky A, Smith SJ . Neuronal activity triggers calcium waves in hippocampal astrocyte networks. Neuron 1992; 8: 429–40.
Grosche J, Matyash V, Moller T, Verkhratsky A, Reichenbach A, Kettenmann H . Microdomains for neuron-glia interaction: parallel fiber signaling to Bergmann glial cells. Nat Neurosci 1999; 2: 139–43.
Aguado F, Espinosa-Parrilla JF, Carmona MA, Soriano E . Neuronal activity regulates correlated network properties of spontaneous calcium transients in astrocytes in situ. J Neurosci 2002 22.
Bezzi P, Carmignoto G, Pasti L, Vesce S, Rossi D, Rizzini BL, et al. Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature 1998; 391: 281–5.
Bezzi P, Gundersen V, Galbete JL, Seifert G, Steinhauser C, Pilati E, et al. Astrocytes contain a vesicular compartment that is competent for regulated exocytosis of glutamate. Nat Neurosci 2004; 7: 613–20.
Zhang Q, Pangrsic T, Kreft M, Krzan M, Li N, Sul JY . Fusion-related release of glutamate from astrocytes. J Biol Chem 2004; 279: 12724–33.
Hua X, Malarkey EB, Sunjara V, Rosenwald SE, Li WH, Parpura V . Ca2+-dependent glutamate release involves two classes of en-doplasmic reticulum Ca2+ stores in astrocytes. J Neurosci Res 2004; 76: 86–97.
Kreft M, Stenovec M, Rupnik M, Grilc S, Krzan M, Potokar M, et al. Properties of Ca(2+)-dependent exocytosis in cultured astrocytes. Glia 2004; 46: 437–45.
Montana V, Ni Y, Sunjara V, Hua X, Parpura V . Vesicular glutamate transporter-dependent glutamate release from astrocytes. J Neurosci 2004; 24: 2633–42.
Hassinger TD, Atkinson PB, Strecker GJ, Whalen LR, Dudek FE, Kossel AH, et al. Evidence for glutamate-mediated activation of hippocampal neurons by glial calcium waves. J Neurobiol 1995; 28: 159–70.
Sanzgiri RP, Araque A, Haydon PG . Prostaglandin E(2) stimulates glutamate receptor-dependent astrocyte neuromodulation in cultured hippocampal cells. J Neurobiol 1999; 41: 221–9.
Araque A, Sanzgiri RP, Parpura V, Haydon PG . Calcium elevation in astrocytes causes an NMDA receptor-dependent increase in the frequency of miniature synaptic currents in cultured hippocampal neurons. J Neurosci 1998; 18: 6822–9.
Kang N, Xu J, Xu Q, Nedergaard M, Kang J . Astrocytic glutamate release-induced transient depolarization and epileptiform discharges in hippocampal CA1 pyramidal neurons. J Neurophysiol 2005; 94: 4121–30.
Pascual O, Casper KB, Kubera C, Zhang J, Revilla-Sanchez R, Sul JY, et al. Astrocytic purinergic signaling coordinates synaptic networks. Science 2005; 310: 113–6.
Zonta M, Angulo MC, Gobbo S, Rosengarten B, Hossmann KA, Pozzan T, et al. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nat Neurosci 2003; 6: 43–50.
Mulligan SJ, Mac Vicar BA . Calcium transients in astrocyte endfeet cause cerebrovascular constrictions. Nature 2004; 431: 195–9.
Nicholls DG, Ward MW . Mitochondrial membrane potential and neuronal glutamate excitotoxicity: mortality and millivolts. Trends Neurosci 2000; 23: 166–74.
Leist M, Nicotera P . Calcium and neuronal death. Rev Physiol Biochem Pharmacol 1998; 132: 79–125.
Gillessen T, Budd SL, Lipton SA . Excitatory amino acid neurotoxicity. Adv Exp Med Biol 2002; 513: 3–40.
Alberdi E, Sanchez-Gomez MV, Matute C . Calcium and glial cell death. Cell Calcium 2005; 38: 417–25.
Salter MG, Fern R . NMDA receptors are expressed in developing oligodendrocyte processes and mediate injury. Nature 2005; 438: 1167–71.
Karadottir R, Cavelier P, Bergersen LH, Attwell D . NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 2005; 438: 1162–6.
Micu I, Jiang Q, Coderre E, Ridsdale A, Zhang L, Woulfe J, et al. NMDA receptors mediate calcium accumulation in myelin during chemical ischaemia. Nature 2006; 439: 988–92.
Blumenthal I . Periventricular leucomalacia: a review. Eur J Pediatr 2004; 163: 435–42.
Caplan LR . Binswanger's disease – revisited. Neurology 1995; 45: 626–33.
Lyons SA, Kettenmann H . Oligodendrocytes and microglia are selectively vulnerable to combined hypoxia and hypoglycemia injury in vitro. J Cereb Blood Flow Metab 1998 18.
Fern R . Ischemia: astrocytes show their sensitive side. Prog Brain Res 2001; 132: 405–11.
Fern R . Intracellular calcium and cell death during ischemia in neonatal rat white matter astrocytes in situ. J Neurosci 1998 18.
Duffy S, Mac Vicar BA . In vitro ischemia promotes calcium influx and intracellular calcium release in hippocampal astrocytes. J Neurosci 1996; 16: 71–81.
Pellerin L, Magistretti PJ . Neuroenergetics: calling upon astrocytes to satisfy hungry neurons. Neuroscientist 2004; 10: 53–62.
Desagher S, Glowinski J, Premont J . Astrocytes protect neurons from hydrogen peroxide toxicity. J Neurosci 1996; 16: 2553–62.
Kofuji P, Newman EA . Potassium buffering in the central nervous system. Neuroscience 2004; 129: 1045–56.
Budd SL, Lipton SA . Calcium tsunamis: do astrocytes transmit cell death messages via gap junctions during ischemia? Nat Neurosci 1998; 1: 431–2.
Lin JH, Weigel H, Cotrina ML, Liu S, Bueno E, Hansen AJ, et al. Gap-junction-mediated propagation and amplification of cell injury. Nat Neurosci 1998; 1: 494–500.
Cotrina ML, Kang J, Lin JH, Bueno E, Hansen TW, He L, et al. Astrocytic gap junctions remain open during ischemic conditions. J Neurosci 1998; 18: 2520–37.
Leao AAP . Spreading depression of activity in the cerebral cortex. J Neurophysiol 1944; 7: 359–90.
Pietrobon D . Migraine: new molecular mechanisms. Neuroscientist 2005; 11: 373–86.
Walz W . Role of astrocytes in the spreading depression signal between ischemic core and penumbra. Neurosci Biobehav Rev 1997; 21: 135–42.
Nedergaard M, Hansen AJ . Characterization of cortical depolarizations evoked in focal cerebral ischemia. J Cereb Blood Flow Metab 1993; 13: 568–74.
Gehrmann J, Mies G, Bonnekoh P, Banati R, Iijima T, Kreutzberg GW, et al. Microglial reaction in the rat cerebral cortex induced by cortical spreading depression. Brain Pathol 1993; 3: 11–7.
Kraig RP, Dong LM, Thisted R, Jaeger CB . Spreading depression increases immunohistochemical staining of glial fibrillary acidic protein. J Neurosci 1991; 11: 2187–98.
Konnerth A, Heinemann U, Yaari Y . Nonsynaptic epileptogenesis in the mammalian hippocampus in vitro. I. Development of seizurelike activity in low extracellular calcium. J Neurophysiol 1986 56.
Yaari Y, Konnerth A, Heinemann U . Nonsynaptic epileptogenesis in the mammalian hippocampus in vitro. II. Role of extracellular potassium. J Neurophysiol 1986; 56: 4121–30.
Kang N, Xu J, Xu Q, Nedergaard M, Kang J . Astrocytic glutamate release-induced transient depolarization and epileptiform discharges in hippocampal CA1 pyramidal neurons. J Neurophysiol 2005; 94: 4121–30.
Fellin T Hay don PG . Do astrocytes contribute to excitation underlying seizures? Trends Mol Med 2005; 11: 530–3.
Tian GF, Azmi H, Takano T, Xu Q, Peng W, Lin J, et al. An astrocytic basis of epilepsy. Nat Med 2005; 11: 973–81.
Kim JH . Pathology of epilepsy. Exp Mol Pathol 2001; 70: 345–67.
Blumcke I, Thom M, Wiestler OD . Ammon's horn sclerosis: a maldevelopmental disorder associated with temporal lobe epilepsy. Brain Pathol 2002; 12: 199–211.
Alzheimer A . Über eine eigenartige Erkrankung der Hirnrinde. Allg Z Psychiat Psych-Gericht Med 1907; 64: 146–8.
Nagele RG, Wegiel J, Venkataraman V, Imaki H, Wang KC . Contribution of glial cells to the development of amyloid plaques in Alzheimer's disease. Neurobiol Aging 2004; 25: 663–74.
Wyss-Coray T, Loike JD, Brionne TC, Lu E, Anankov R, Yan F, et al. Adult mouse astrocytes degrade amyloid-beta in vitro and in situ. Nat Med 2003 9.
Abramov AY, Canevari L, Duchen MR . Calcium signals induced by amyloid beta peptide and their consequences in neurons and astrocytes in culture. Biochim Biophys Acta 2004; 1742: 81–7.
Abramov AY, Canevari L, Duchen MR . Changes in intracellular calcium and glutathione in astrocytes as the primary mechanism of amyloid neurotoxicity. J Neurosci 2003; 23: 5088–95.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Wellcome Trust, the Alzheimer Research Trust, NIH, Royal Society and INTAS.
Rights and permissions
About this article
Cite this article
Verkhratsky, A. Glial calcium signaling in physiology and pathophysiology. Acta Pharmacol Sin 27, 773–780 (2006). https://doi.org/10.1111/j.1745-7254.2006.00396.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00396.x
Keywords
This article is cited by
-
Chronic Stress Impairs the Structure and Function of Astrocyte Networks in an Animal Model of Depression
Neurochemical Research (2023)
-
The Neuroimmune System and the Cerebellum
The Cerebellum (2023)
-
Gene expression, proteome and calcium signaling alterations in immortalized hippocampal astrocytes from an Alzheimer’s disease mouse model
Cell Death & Disease (2019)
-
Role of transient receptor potential ankyrin 1 channels in Alzheimer’s disease
Journal of Neuroinflammation (2016)
-
Calcium signals in the nucleus accumbens: Activation of astrocytes by ATP and succinate
BMC Neuroscience (2011)