Abstract
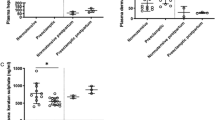
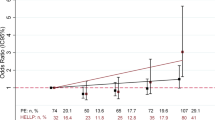
Severe inflammation has been shown to induce a shedding of the endothelial glycocalyx (EGX). Inflammatory cytokines, such as tumor necrosis factor α (TNF-α), impede the thickness of the EGX. While a controlled inflammatory reaction occurs already in normal pregnancy, women with hemolysis, elevated liver enzymes and low platelets (HELLP) syndrome had an exaggerated inflammatory response. This study investigates the shedding of the glycocalyx during normal pregnancy and in women with HELLP syndrome. Glycocalyx components (syndecan 1, heparan sulfate, and hyaluronic acid) were measured in serum of healthy women throughout pregnancy (4 time points, n = 26), in women with HELLP syndrome (n = 17) before delivery and in nonpregnant volunteers (n = 10). Serum concentrations of TNF-α and soluble TNF-α receptors (sTNF-Rs) were assessed once in all 3 groups. Syndecan 1 serum concentrations constantly rose throughout normal pregnancy. Immediately before delivery, a 159-fold increase was measured compared to nonpregnant controls (P < .01). Even higher amounts were observed in patients with HELLP prior to delivery (median 12 252 ng/mL) compared to healthy women matched by gestational age (median 5943 ng/mL; P < .01). Relevantly, increased serum levels of heparan sulfate, hyaluronic acid, and sTNF-Rs were only detected in patients with HELLP (P < .01). These findings suggest that considerable amounts of syndecan 1 are released into maternal blood during uncomplicated pregnancy. The HELLP syndrome is associated with an even more pronounced shedding of glycocalyx components. The maternal vasculature as well as the placenta has to be discussed as a possible origin of circulating glycocalyx components.
Similar content being viewed by others
References
Weinbaum S, Tarbell JM, Damiano ER. The structure and function of the endothelial glycocalyx layer. Annu Rev Biomed Eng. 2007;9:121–167.
Pries AR, Secomb TW, Gaehtgens P. The endothelial surface layer. Pflugers Arch. 2000;440(5):653–666.
Reitsma S, Slaaf DW, Vink H, van Zandvoort MA, oude Egbrink MG. The endothelial glycocalyx: composition, functions, and visualization. Pflugers Arch. 2007;454(3):345–359.
Chappell D, Jacob M, Becker BF, Hofmann-Kiefer K, Conzen P, Rehm M. Expedition glycocalyx. A newly discovered ‘‘Great Barrier Reef’’ [in German]. Anaesthesist. 2008;57(10):959–969.
Klein NJ, Shennan GI, Heyderman RS, Levin M. Alteration in glycosaminoglycan metabolism and surface charge on human umbilical vein endothelial cells induced by cytokines, endotoxin and neutrophils. J Cell Sci. 1992;102(pt 4):821–832.
Jacob M, Bruegger D, Rehm M, et al. The endothelial glycocalyx affords compatibility of Starling’s principle and high cardiac interstitial albumin levels. Cardiovasc Res. 2007;73(3):575–586.
Rehm M, Zahler S, Lotsch M, et al. Endothelial glycocalyx as an additional barrier determining extravasation of 6% hydroxyethyl starch or 5% albumin solutions in the coronary vascular bed. Anesthesiology. 2004;100(5):1211–1223.
Becker BF, Chappell D, Jacob M. Endothelial glycocalyx and coronary vascular permeability: the fringe benefit. Basic Res Cardiol. 2010;105(6):687–701.
Mulivor AW, Lipowsky HH. Inflammation- and ischemiainduced shedding of venular glycocalyx. Am J Physiol Heart Circ Physiol. 2004;286(5):H1672–H1680.
Mulivor AW, Lipowsky HH. Role of glycocalyx in leukocyteendothelial cell adhesion. Am J Physiol Heart Circ Physiol. 2002;283(4):H1282–H1291.
Henry CB, Duling BR. TNF-alpha increases entry of macromolecules into luminal endothelial cell glycocalyx. Am J Physiol Heart Circ Physiol. 2000;279(6):H2815–H2823.
Hofmann-Kiefer KF, Kemming GI, Chappell D, et al. Serum heparan sulfate levels are elevated in endotoxemia. Eur J Med Res. 2009;14(12):526–531.
Jacob M, Chappell D, Hofmann-Kiefer K, Conzen P, Peter K, Rehm M. Determinants of insensible fluid loss: Perspiration, protein shift and endothelial glycocalyx [in German]. Anaesthesist. 2007;56(8):747–758, 760–764.
Nelson A, Berkestedt I, Schmidtchen A, Ljunggren L, Bodelsson M. Increased levels of glycosaminoglycans during septic shock: relation to mortality and the antibacterial actions of plasma. Shock. 2008;30(6):623–627.
Marechal X, Favory R, Joulin O, et al. Endothelial glycocalyx damage during endotoxemia coincides with microcirculatory dysfunction and vascular oxidative stress. Shock. 2008;29(5): 572–576.
Henrich M, Gruss M, Weigand MA. Sepsis-induced degradation of endothelial glycocalix. Scientific World Journal. 2010;10:917–923.
Chappell D, Hofmann-Kiefer K, Jacob M, et al. TNF-alpha induced shedding of the endothelial glycocalyx is prevented by hydrocortisone and antithrombin. Basic Res Cardiol. 2009; 104(1):78–89.
Oragui EE, Nadel S, Kyd P, Levin M. Increased excretion of urinary glycosaminoglycans in meningococcal septicemia and their relationship to proteinuria. Crit Care Med. 2000;28(8): 3002–3008.
Redman CW, Sacks GP, Sargent IL. Preeclampsia: an excessive maternal inflammatory response to pregnancy 1. Am J Obstet Gynecol. 1999;180(2 pt 1):499–506.
Mellembakken JR, Aukrust P, Olafsen MK, Ueland T, Hestdal K, Videm V. Activation of leukocytes during the uteroplacental passage in preeclampsia. Hypertension. 2002;39(1):155–160.
Mellembakken JR, Aukrust P, Hestdal K, Ueland T, Abyholm T, Videm V. Chemokines and leukocyte activation in the fetal circulation during preeclampsia. Hypertension. 2001;38(3):394–398.
Haram K, Svendsen E, Abildgaard U. The HELLP syndrome: clinical issues and management. A Review. BMC Pregnancy Childbirth. 2009;9:8.
Nieuwdorp M, van Haeften TW, Gouverneur MC, et al. Loss of endothelial glycocalyx during acute hyperglycemia coincides with endothelial dysfunction and coagulation activation in vivo. Diabetes. 2006;55(2):480–486.
Vink H, Constantinescu AA, Spaan JA. Oxidized lipoproteins degrade the endothelial surface layer: implications for plateletendothelial cell adhesion. Circulation. 2000;101(13):1500–1502.
Martin JN Jr, Rinehart BK, May WL, Magann EF, Terrone DA, Blake PG. The spectrum of severe preeclampsia: comparative analysis by HELLP (hemolysis, elevated liver enzyme levels, and low platelet count) syndrome classification. Am J Obstet Gynecol. 1999;180(6 pt 1):1373–1384.
O’Hara Padden M. HELLP Syndrome: recognition and perinatal management. Am Fam Physician. 1999;60(3):829–839.
van Runnard Heimel PJ, Franx A, Schobben AF, Huisjes AJ, Derks JB, Bruinse HW. Corticosteroids, pregnancy, and HELLP syndrome: a review. Obstet Gynecol Surv. 2005;60(1):57–70.
Smith HL. Matching with multiple controls to estimate treatment effects in observational studies. Sociol Methodol. 1997;27:325–353.
Gardosi J, Chang A, Kalyan B, Sahota D, Symonds EM. Customised antenatal growth charts. Lancet. 1992;339(8788): 283–287.
Steegers EA, von DP, Duvekot JJ, Pijnenborg R. Pre-eclampsia. Lancet. 2010;376(9741):631–644.
Rehm M, Bruegger D, Christ F, et al. Shedding of the endothelial glycocalyx in patients undergoing major vascular surgery with global and regional ischemia. Circulation. 2007;116(17): 1896–1906.
Kim JM, Lee JA, Cho IS, Ihm CH. Soluble syndecan-1 at diagnosis and during follow up of multiple myeloma: a single institution study. Korean J Hematol. 2010;45(2):115–119.
Anttonen A, Leppa S, Ruotsalainen T, Alfthan H, Mattson K, Joensuu H. Pretreatment serum syndecan-1 levels and outcome in small cell lung cancer patients treated with platinum-based chemotherapy. Lung Cancer. 2003;41(2):171–177.
Parsian H, Rahimipour A, Nouri M, et al. Serum hyaluronic acid and laminin as biomarkers in liver fibrosis. J Gastrointestin Liver Dis. 2010;19(2):169–174.
Puder JJ, Blum CA, Mueller B, De GC, Dye L, Keller U. Menstrual cycle symptoms are associated with changes in low-grade inflammation. Eur J Clin Invest. 2006;36(1):58–64.
Sargent IL, Borzychowski AM, Redman CW. Immunoregulation in normal pregnancy and pre-eclampsia: an overview. Reprod Biomed Online. 2006;13(5):680–686.
Alexopoulou AN, Multhaupt HA, Couchman JR. Syndecans in wound healing, inflammation and vascular biology. Int J Biochem Cell Biol. 2007;39(3):505–528.
Jokimaa V, Inki P, Kujari H, Hirvonen O, Ekholm E, Anttila L. Expression of syndecan-1 in human placenta and decidua. Placenta. 1998;19(2–3):157–163.
Kingdom J, Huppertz B, Seaward G, Kaufmann P. Development of the placental villous tree and its consequences for fetal growth. Eur J Obstet Gynecol Reprod Biol. 2000;92(1):35–43.
Redman CW, Sargent IL. Placental debris, oxidative stress and pre-eclampsia. Placenta. 2000;21(7):597–602.
Knight M, Redman CW, Linton EA, Sargent IL. Shedding of syncytiotrophoblast microvilli into the maternal circulation in preeclamptic pregnancies. Br J Obstet Gynaecol. 1998;105(6): 632–640.
Goswami D, Tannetta DS, Magee LA, et al. Excess syncytiotrophoblast microparticle shedding is a feature of early-onset preeclampsia, but not normotensive intrauterine growth restriction. Placenta. 2006;27(1):56–61.
Gotte M, Echtermeyer F. Syndecan-1 as a regulator of chemokine function. ScientificWorldJournal. 2003;3:1327–1331.
Gotte M. Syndecans in inflammation. FASEB J. 2003;17(6): 575–591.
Jokimaa VI, Kujari HP, Ekholm EM, Inki PL, Anttila L. Placental expression of syndecan 1 is diminished in preeclampsia. Am J Obstet Gynecol. 2000;183(6):1495–1498.
Simon AC, Levenson J, Flaud P. Pulsatile flow and oscillating wall shear stress in the brachial artery of normotensive and hypertensive subjects. Cardiovasc Res. 1990;24(2):129–136.
Simon AC, Levenson J. Abnormal wall shear conditions in the brachial artery of hypertensive patients. J Hypertens. 1990;8(2): 109–114.
Bruegger D, Jacob M, Rehm M, et al. Atrial natriuretic peptide induces shedding of endothelial glycocalyx in coronary vascular bed of guinea pig hearts. Am J Physiol Heart Circ Physiol. 2005;289(5):H1993–H1999.
Borghi C, Cicero AF, Degli ED, et al. Hemodynamic and neurohumoral profile in patients with different types of hypertension in pregnancy. Intern Emerg Med. 2011;6(3):227–234.
Kupferminc MJ, Peaceman AM, Wigton TR, Tamura RK, Rehnberg KA, Socol ML. Immunoreactive tumor necrosis factor-alpha is elevated in maternal plasma but undetected in amniotic fluid in the second trimester. Am J Obstet Gynecol. 1994;171(4):976–979.
Conrad KP, Miles TM, Benyo DF. Circulating levels of immunoreactive cytokines in women with preeclampsia. Am J Reprod Immunol. 1998;40(2):102–111.
Vince GS, Starkey PM, Austgulen R, Kwiatkowski D, Redman CW. Interleukin-6, tumour necrosis factor and soluble tumour necrosis factor receptors in women with pre-eclampsia. Br J Obstet Gynaecol. 1995;102(1):20–25.
Visser W, Beckmann I, Bremer HA, Lim HL, Wallenburg HC. Bioactive tumour necrosis factor alpha in pre-eclamptic patients with and without the HELLP syndrome. Br J Obstet Gynaecol. 1994;101(12):1081–1082.
Schipper EJ, Bolte AC, Schalkwijk CG, Van Geijn HP, Dekker GA. TNF-receptor levels in preeclampsia—results of a longitudinal study in high-risk women. J Matern Fetal Neonatal Med. 2005;18(5):283–287.
van Runnard Heimel PJ, Kavelaars A, Heijnen CJ, et al. HELLP syndrome is associated with an increased inflammatory response, which may be inhibited by administration of prednisolone. Hypertens Pregnancy. 2008;27(3):253–265.
Sibai B, Romero R, Klebanoff MA, et al. Maternal plasma concentrations of the soluble tumor necrosis factor receptor 2 are increased prior to the diagnosis of preeclampsia. Am J Obstet Gynecol. 2009;200(6):630.e1–e8.
Fantuzzi G, Demitri MT, Ghezzi P. Differential effect of glucocorticoids on tumour necrosis factor production in mice: upregulation by early pretreatment with dexamethasone. Clin Exp Immunol. 1994;96(1):166–169.
Reikeras O, Helle A, Krohn CD, Brox JI. Effects of high-dose corticosteroids on post-traumatic inflammatory mediators. Inflamm Res. 2009;58(12):891–897.
Man SF, Xuekui Z, Vessey R, et al. The effects of inhaled and oral corticosteroids on serum inflammatory biomarkers in COPD: an exploratory study. Ther Adv Respir Dis. 2009;3(2):73–80.
Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol. 2000;183(1):S1–S22.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hofmann-Kiefer, K.F., Knabl, J., Martinoff, N. et al. Increased Serum Concentrations of Circulating Glycocalyx Components in HELLP Syndrome Compared to Healthy Pregnancy: An Observational Study. Reprod. Sci. 20, 318–325 (2013). https://doi.org/10.1177/1933719112453508
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719112453508