Abstract
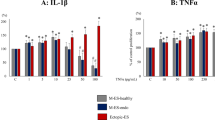
Endometriosis is a multifactorial disease mostly affecting women of reproductive age. An additive effect between inflammation and stress reaction on the growth of endometriosis has been demonstrated. Here we investigated the combined effect between 17β-estradiol (E2) and lipopolysaccharide (LPS) on pelvic inflammation and growth of endometriotic cells. Peritoneal fluid was collected from 46 women with endometriosis and 30 control women during laparoscopy. Peritoneal macrophages (Mφ) and stromal cells from eutopic/ectopic endometrial stromal cells (ESCs) were isolated from 10 women each with and without endometriosis in primary culture. Changes in cytokine secretion (interleukin 6 [IL-6] and tumor necrosis factor α [TNF-α]) by Mφ and proliferation of ESCs in response to single and combined treatment with E2 and LPS were measured by enzyme-linked immunosorbent assay and by bromodeoxyuridine incorporation assay, respectively. A significantly increased secretion of IL-6 and TNF-α in Mφ culture media was found in response to E2 (10−8 mol/L) compared to nontreated Mφ. This effect of E2 was abrogated after pretreatment of cells with ICI 182720 (10−6 mol/L; an estrogen receptor [ER] antagonist). Combined treatment with E2 and LPS (10 ng/mL) additively promoted IL-6 and TNF-α secretion by peritoneal Mφ and growth of eutopic/ectopic ESCs. The additive effects of E2 + LPS on cytokine secretion and growth of ESCs were effectively suppressed after combined blocking of ER and Toll-like receptor 4. An additive effect was observed between E2 and LPS on promoting proinflammatory response in pelvis and growth of endometriosis.
Similar content being viewed by others
References
Attar E, Bulun SE. Aromatase and other steroidogenic genes in endometriosis: translational aspects. Hum Reprod Update. 2006; 12(1):49–56.
Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3):511–519.
Khan KN, Kitajima M, Hiraki H, et al. Immunopathogenesis of pelvic endometriosis: role of hepatocyte growth factor, macrophages and ovarian steroids. Am J Reprod Immunol. 2008;60(5):383–404.
Khan KN, Kitajima M, Hiraki K, et al. Escherichia coli contamination of menstrual blood and effect of bacterial endotoxin on endometriosis. Fertil Steril. 2010;94(7):2860–2863.
Khan KN, Kitajima M, Inoue T, et al. Additive effects of inflammation and stress reaction on Toll-like receptor 4-mediated growth of endometriotic stromal cells. Hum Reprod. 2013;28(10):2794–2803.
Straub RH. The complex role of estrogen in inflammation. Endocr Rev. 2007;28(5):521–574.
Khan KN, Masuzaki H, Fujishita A, et al. Estrogen and progesterone receptor expression in macrophages and regulation of hepatocyte growth factor by ovarian steroids in women with endometriosis. Hum Reprod. 2005;20(7):2004–2013.
Calippe B, Douin-Echinard V, Laffargue M, et al. Chronic estradiol administration in vivo promotes the pro-inflammatory response of macrophages to TLR4 activation: involvement of the phosphatidylinositol 3-kinase pathway. J Immunol. 2008;180(12):7980–7988.
Khan KN, Masuzaki H, Fujishita A, et al. Association of interleukin-6 and estradiol with hepatocyte growth factor in peritoneal fluid of women with endometriosis. Acta Obstet Gynecol Scand. 2002;81(8):764–771.
The American Society for Reproductive Medicine. Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67(5):817–821.
Kamiyama S, Teruya Y, Nohara M, Kanazawa K. Impact of detection of bacterial endotoxin in menstrual effluent on the pregnancy rate in in vitro fertilization and embryo transfer. Fertil Steril. 2004;82(4):788–792.
Babson AL. Olson DR, Palmieri T, Rosss AF, Becker DM, Mulqueen PJ. The IMMULITE assay tube: a new approach to heterogenous ligand assay. Clin Chem. 1991;37(9):1521–1522.
McLaren J, Prentice A, Charnock-Jones DS, et al. Vascular endothelial growth factor is produced by peritoneal fluid macrophages in endometriosis and is regulated by ovarian steroids. J Clin Invest. 1996;98(2):482–489.
King AE, Collins F, Klonisch T, Sallenave JM, Critchley HOD, Saunders PTK. An additive interaction between the NFkB and estrogen receptor signaling pathways in human endometrial epithelial cells. Hum Reprod. 2010;25(2):510–518.
Takagi S. Detection of 5-bromo-2-deoxyuridine (BrdU) incorporation with monoclonal anti-BrdU antibody after deoxyribonuclease treatment. Cytometry. 1993;14(6):640–649.
Halme J, White C, Kauma S, Estes J, Haskill S. Peritoneal macrophages from patients with endometriosis release growth factor activity in vitro. J Clin Endocrinol Metab. 1988;66(5): 1044–1049.
Halme J. Release of tumor necrosis factor-alpha by human peritoneal macrophages in vivo and in vitro. Am J Obstet Gynecol. 1989;161(6 pt 1):1718–1725.
Cakmak H, Guzeloglu-Kayisli O, Kayisli UA, Arici A. Immuneendocrine interactions in endometriosis. Front Biosci. 2009;E1: 429–443.
Nirgianakis K, Bersinger NA, McKinnon B, Kostov P, Imboden S, Mueller MD. Regression of the inflammatory microenvironment of the peritoneal cavity in women with endometriosis by GnRHa treatment. Eur J Obstet Gynecol Reprod Biol. 2013; 170(2):550–554.
Eischen A, Duclos B, Schmitt-Gognel M, et al. Human resident peritoneal macrophages: phenotype and histology. Br J Haematol. 1994;88(4):712–722.
Khan KN, Kitajima M, Imamura T, et al. Toll-like receptor 4 (TLR4)-mediated growth of endometriosis by human heat shock protein 70 (Hsp70). Hum Reprod. 2008;23(10):2210–2219.
Montagna P, Capellino S, Villaggio B, et al. Peritoneal fluid macrophages in endometriosis: correlation between the expression of estrogen receptors and inflammation. Fertil Steril. 2008; 90(1):156–164.
Calippe B, Douin-Echinard V, Delpy L, et al. 17ß-Estradiol promotes TLR4-triggered pro-inflammatory mediator production through direct estrogen receptorα signaling in macrophages in vivo. J Immunol. 2010;185(2):1169–1176.
Mor G, Sapi E, Abrahams VM, et al. Interaction of the estrogen receptors with the Fas ligand promoter in human monocytes. J Immunol. 2003;170(1):114–122.
Kaponis A, Iwabe T, Taniguchi F, et al. The role of NF-kappaB in endometriosis. Front Biosci. 2012;S4:1568–1581.
Kwon JH, Keates S, Simeonidis S, Grall F, Libermann TA, Keates AC. ESE-1, an enterocyte-specific Ets transcription factor, regulates MIP-3alpha gene expression in Caco-2 human colonic epithelial cells. J Biol Chem. 2003;278(2):875–884.
Gruber CJ, Gruber DM, Gruber IM, Wieser F, Huber JC. Anatomy of the estrogen response element. Trends Endocrinol Metab. 2004;15(2):73–78.
Falkenstein E, Tillmann HC, Christ M, Feuring M, Wehling M. Multiple actions of steroid hormones-a focus on rapid, nongenomic effects. Pharmacol Rev. 2000;52(4):513–556.
Rettew JA, Huet YM, Marriott I. Estrogens augment cell surface TLR4 expression on murine macrophages and regulate sepsis susceptibility in vivo. Endocrinology. 2009;150(8):3877–3884.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khan, K.N., Kitajima, M., Inoue, T. et al. 17β-Estradiol and Lipopolysaccharide Additively Promote Pelvic Inflammation and Growth of Endometriosis. Reprod. Sci. 22, 585–594 (2015). https://doi.org/10.1177/1933719114556487
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719114556487