Abstract
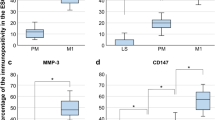
Clusterin (CLU) is expressed in tissues and body fluids and is altered in some pathologies. In endometriosis, a noninvasive test is still lacking, thus, we analyzed CLU in mucus samples of patients. Additionally, we investigated localization of CLU and the putative CLU receptors (apolipoprotein E receptor 2 [ApoER2], megalin, very low-density lipoprotein receptor [VLDLR], and transforming growth factor β receptor type I and II [TβR1/TβR2]). In mucus samples, CLU levels are modestly, but not significantly, higher in cases with endometriosis compared to cases without endometriosis, however, CLU levels are significantly (P =.02) reduced in patients with endometriosis receiving contraception compared to cases with endometriosis without contraception. Analysis of CLU and CLU receptors showed CLU mainly in the uterine epithelial cells in the majority of glands, but also in endothelial cells. Similarly, ApoER2 and TβR1 could also be found preferentially in the endometrial glands. Whereas ApoER2 staining was strong in the vessels, TβR1 was modestly expressed in vessels and muscle cells. In contrast, staining of VLDLR and TbR2 was modest in the glands but stronger in vessels and muscle cells. Megalin staining was faint in the glands. A similar pattern for these proteins could be observed in adenomyosis. We demonstrate for the first time high concentrations of CLU in mucus samples and significantly reduced CLU levels in cases with endometriosis receiving contraception compared to cases with endometriosis without contraception. Furthermore, we identified uterine epithelial and endothelial cells as the main source of CLU and found different preferential CLU receptor complexes on glands, vessels, and smooth muscle cells.
Similar content being viewed by others
References
Fritz, IB, Burdzy, K, Setchell, B, Blaschuk, O Ram rete testis fluid contains a protein (clusterin) which influences cell-cell-interactions in vitro. Biol Reprod. 1983;28(5):1173–1188.
Shannan, B, Seifert, M, Leskov, K, et al. Challenge and promise: roles for clusterin in pathogenesis, progression and therapy of cancer. Cell Death Differ. 2006;13(1):12–19.
Jin, G, Howe, PH Regulation of clusterin gene expression by transforming growth factor β. J Biol Chem. 1997;272(42): 26620–26626.
Jin, G, Howe, PH Transforming growth factor β regulates clusterin gene expression via modulation of transcription factor c-Fos. Eur J Biochem. 1999;263(2):534–542.
Bettuzzi, S, Davalli, P, Davoli, S, et al. Genetic inactivation of ApoJ/clusterin: effects on prostate tumourigenesis and metastatic spread. Oncogene. 2009;28(49):4344–4352.
Chayka, O, Corvetta, D, Dews, M, et al. Clusterin, a haploinsuffi-cient tumor suppressor gene in neuroblastomas. J Natl Cancer Inst. 2009;101(9):663–677.
Bailey, RW, Aronow, B, Harmony, JA, Griswold, MD Heat shock-initiated apoptosis is accelerated and removal of damaged cells is delayed in the testis of clusterin/ApoJ knock-out mice. Biol Reprod. 2002;66(4):1042–1053.
Trougakis, IP, So, A, Jansen, B, Gleave, ME, Gonos, ES Silencing expression of the clusterin/apolipoprotein j gene in human cancer cells using small interfering RNA induces spontaneous apoptosis, reduced growth ability, and cell sensitization to genotoxic and oxidative stress. Cancer Res. 2004;64(4):1834–1842.
Mishima, K, Inoue, H, Nishiyama, T, et al. Transplantation of side population cells restore the function of damaged exocrine glands through clusterin. Stem Cells. 2012;30(9):1925–1937.
Thomas-Tikhonenko, A, Viard-Leveugle, I, Dews, M, et al. Myc-transformed epithelial cells down-regulate clusterin, which inhibits their growth in vitro and carcinogenesis in vivo. Cancer Res. 2004;64(9):3126–3136.
Lenferink, AE, Cantin, C, Nantel, A, et al. Transcriptome profiling of a TGF-β-induced epithelial-to-mesenchymal transition reveals extracellular clusterin as a target for therapeutic antibodies. Onco-gene. 2010;29(6):831–844.
Nuutinen, T, Suuronen, T, Kauppinen, A, Saliminen, A Clusterin: a forgotten player in Alzheimer’s disease. Brain Res Rev. 2009; 61(2):89–104.
McLaughlin, L, Zhu, G, Mistry, M, et al. Apolipoprotein J/clusterin limits the severity of murine autoimmune myocarditis. J Clin Invest. 2000;106(9):1105–1113.
Lee, KB, Jeon, JH, Choi, I, Kwon, OY, et al. Clusterin, a novel modulator of TGF-β signaling, is involved in Smad2/3 stability. Biochem Biophys Res Commun. 2008;366(4):905–909.
Reddy, KB, Karode, MC, Harmony, AK, Howe, PH Interaction of transforming growth factor β receptors with apolipoprotein J/clus-terin. Biochemistry. 1996;35(1):309–314.
Omwandho, CO, Konrad, L, Halis, G, Oehmke, F, Tinneberg, HR Role of TGF-betas in normal human endometrium and endome-triosis. Hum Reprod. 2010;25(1):101–109.
Chegini, N, Zhao, Y, Williams, RS, Flanders, KC Human uterine tissue throughout the menstrual cycle expresses transforming growth factor-β1 (TGFβ1), TGFβ2, TGFβ3, and TGFβ type II receptor messenger ribonucleic acid and protein and contains [125I]TGFb1-binding sites. Endocrinology. 1994; 135(1):439–449.
Zlokovic, BV, Martel, CL, Matsubara, E, et al. Glycoprotein 330/megalin: probable role in receptor-mediated transport of apolipoprotein J alone and in a complex with Alzheimer disease amyloid beta at the blood-brain and blood-cerebrospinal barriers. Proc Natl Acad Sci U S A 1996; 93(9):4229–4234.
Lundgren, S, Carling, T, Hjälm, G, et al. Tissue distribution of human gp330/megalin, a putative Ca2+-sensing protein. J Histo-chem Cytochem. 1997;45(3):383–392.
Nykjaer, A, Dragun, D, Walther, D, et al. An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell. 1999;96(4):507–515.
Hammes, A, Andreassen, TK, Spoelgen, R, et al. Role of endocy-tosis in cellular uptake of sex steroids. Cell. 2005;122(5):751–762.
Trommsdorff, M, Gotthardt, M, Hiesberger, T, et al. Reeler/ disabled-like disruption of neuronal migration in knockout mice lacking the VLDL receptor and ApoE receptor 2. Cell. 1999; 97(6):689–701.
Leeb, C, Eresheim, C, Nimpf, J Clusterin is a ligand for apolipo-protein E receptor 2 (ApoER2) and very low density lipoprotein receptor (VLDLR) and signals via the Reelin-signaling pathway. J Biol Chem. 2014;289(7):4161–4172.
Burk, RF, Hill, KE, Olson, GE, et al. Deletion of apolipoprotein E receptor-2 in mice lowers brain selenium and causes severe neu-rological dysfunction and death when a low-selenium diet is fed. J Neurosci. 2007;27(23):6207–6211.
Frykman, PK, Brown, MS, Yamamoto, T, Goldstein, JL, Herz, J Normal plasma lipoproteins and fertility in gene-targeted mice homozygous for a disruption in the gene encoding very low den-sity lipoprotein receptor. Proc Natl Acad Sci U S A 1995;92(18): 8453–8457.
Argraves, WS, Morales, CR Immunolocalization of cubilin, mega-lin, apolipoprotein, J, and apolipoprotein A-I in the uterus and oviduct. Mol Reprod Dev. 2004;69(4):419–427.
Fuzio, P, Valletti, A, Napoli, A, et al. Regulation of the expression of CLU isoforms in endometrial proliferative diseases. Int J Oncol. 2013;42(6):1929–1944.
Brown, TL, Moulton, BC, Baker, VV, Mira, J, Harmony, JA Expression of apolipoprotein J in the uterus is associated with tissue remodeling. Biol Reprod. 1995;52(5):1038–1049.
Ferrero, S, Gillott, DJ, Remorgida, V, et al. Proteomic analysis of peritoneal fluid in fertile and infertile women with endometriosis. J Reprod Med. 2009;54(1):32–40.
Hackethal, A, Luck, C, von Hobe, AK, et al. A structured ques-tionnaire improves preoperative assessment of endometriosis patients: a retrospective analysis and prospective trial. Arch Gyne-col Obstet. 2011;284(5):1179–1188.
Andersen, CL, Schepeler, T, Thorsen, K, et al. Clusterin expression in normal mucosa and colorectal cancer. Mol Cell Proteomics. 2007;6(6):1039–1048.
Konrad, L, Scheiber, JA, Völck-Badouin, E, et al. Alternative spli-cing of TGF-betas and their high-affinity receptors TβRI, TβRII and TβRIII (Betaglycan) reveal new variants in human prostatic cells. BMC Genomics. 2007;8:318.
Varghese, F, Bukhari, AB, Malhotra, R, De, A IHC Profiler: an open source plugin for the quantitative evaluation and automated scoring of immunohistochemistry images of human tissue sam-ples. Plos One. 2014;9(5):e96801.
Fuhrich, DG, Lessey, BA, Savaris, RF Comparison of HSCORE assessment of endometrial β3 integrin subunit expression with digital HSCORE using computerized image analysis (ImageJ). Anal Quant Cytopathol Histopathol. 2013;35(4):210–216.
Fassbender, A, Burney, RO, Dorien, FO, et al. Update on biomar-kers for the detection of endometriosis. BioMed Res Int. 2015; 2015(3):130854.
de Bruijn, HW, van Beeck Calkoen-Carpay, T, Jager, S, Duk, JM, Aalders, JG, Fleuren, GJ The tumor marker CA 125 is a common constituent of normal cervical mucus. Am J Obstet Gynecol. 1986; 154(5):1088–1091.
Rivera, R, Yacobson, I, Grimes, D The mechanism of action of hormonal contraceptives and intrauterine contraceptive devices. Am J Obstet Gynecol. 1999;181(5 pt 1):1263–1269.
Endrikat, J, Parke, S, Trummer, D, Serrani, M, Duijkers, I, Klipping, C Pituitary, ovarian and additional contraceptive effects of an estradiol-based combined oral contraceptive: results of a rando-mized, open-label study. Contraception. 2013;87(2):227–234.
Grandi, G, Mueller, M, Bersinger, NA, Cagnacci, A, Volpe, A, McKinnon, B Does dienogest influence the inflammatory response of endometriotic cells? A systematic review. Inflamm Res. 2016;65(3):183–192.
Vitiello, D, Pinard, R, Taylor, HS Gene expression profiling reveals putative HOXA10 downstream. Reprod Sci. 2008;15(5): 529–535.
Ulrich, V, Konaniah, ES, Herz, J, et al. Genetic variants of ApoE and ApoER2 differentially modulate endothelial function. Proc Natl Acad Sci U S A 2014;111(37):13493–13498.
Andersen, OM, Yeung, CH, Vorum, H, et al. Essential role of the apolipoprotein E receptor-2 in sperm development. J Biol Chem. 2003;275(26):23989–23995.
Ramesh, S, Morrell, CN, Tarango, C, et al. Antiphospholipid anti-bodies promote leukocyte-endothelial cell adhesion and thrombo-sis in mice by antagonizing eNOS via β2GPI and apoER2. J Clin Invest. 2011;121(1):120–131.
Yakovlev, S, Mikhailenko, I, Cao, C, Zhang, L, Strickland, DK, Medved, L Identification of VLDLR as a novel endothelial cell receptor for fibrin that modulates fibrin-dependent transendothe-lial migration of leukocytes. Blood. 2012;119(2):637–644.
Li, Q, Agno, JE, Edson, MA, Nagaraja, AK, Nagashima, T, Matzuk, MM Transforming growth factor β receptor type 1 is essential for female reproductive tract integrity and function. PLOS Genet. 2011;7(10):e1002320.
Gao, Y, Bayless, KJ, Li, Q TGFBR1 is required for mouse myo-metrial development. Mol Endocrinol. 2014;28(3):380–394.
Ikushima, H, Miyazono, K TGF-β signal transduction spreading to a wider field: a broad variety of mechanisms for context-dependent effects of TGF-β. Cell Tissue Res. 2012; 347(1):37–49.
Lee, S, Hong, SW, Min, BH, et al. Essential role of clusterin in pancreas regeneration. Dev Dyn. 2011;240(3):605–615.
Falgarone, G, Chiocchia, G Chapter 8: Clusterin: a multifacet protein at the crossroad of inflammation and autoimmunity. Adv Cancer Res. 2009;104:139–170.
Vercellini, P, Viganò, P, Somigliana, E, Fedele, L Endometriosis: pathogenesis and treatment. Nat Rev Endocrinol. 2014;10(5): 261–275.
Trougakis, IP The molecular chaperone apolipoprotein J/clusterin as a sensor of oxidative stress: implications in therapeutic approaches–a mini-review. Gerontology. 2013;59(6):514–523.
Carvalho, LF, Samadder, AN, Agarwal, A, Fernandes, LF, Abrão, MS Oxidative stress biomarkers in patients with endometriosis: systematic review. Arch Gynecol Obstet. 2012;286(4):1033–1040.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Konrad, L., Hackethal, A., Oehmke, F. et al. Analysis of Clusterin and Clusterin Receptors in the Endometrium and Clusterin Levels in Cervical Mucus of Endometriosis. Reprod. Sci. 23, 1371–1380 (2016). https://doi.org/10.1177/1933719116641756
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719116641756