Abstract
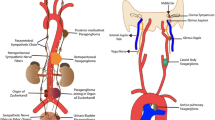
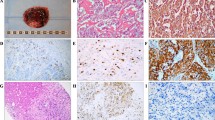
Pheochromocytomas (PCC) are catecholamine-producing tumors that are, by definition, located in the adrenal medulla. Extra-adrenal catecholamine-producing tumors are called paragangliomas (PGL), which should be distinguished from head and neck paragangliomas, which are of parasympathetic origin. As is true for many (neuro)endocrine tumors, but unlike most other epithelial tumors, histopathological analysis does not allow a distinction to be made between PCC and PGL that will follow a benign course and those that have metastasized or will do so, a condition associated with poor prognosis. Therefore, many studies have been undertaken in the past decade, with the aim of providing a marker or a set of markers that allows clinical behavior in PCC and PGL to be predicted. Despite promising results in some areas, such as histopathological scoring systems, the use of the MIB-1 labeling index, and the analysis of telomerase activity, no single test or combination of tests has thus far yielded sufficiently high sensitivity and specificity to result in widespread acceptance in every day clinical practice. The relative rarity of PCC and PGL combined with a frequency of malignancy from as low as 2% up to 25% has hampered the power of past research and can only be overcome by multicenter collaborative efforts. In this article, recent attempts at marker detection, such as those mentioned above, as well as emerging knowledge on the molecular abnormalities in benign and malignant PCC and PGL will be presented.
Similar content being viewed by others
References
van der Harst E, de Herder WW, de Krijger RR, et al. The value of plasma markers for the clinical behaviour of phaeochromocytomas. Eur J Endocrinol 147:85–94, 2002.
de Krijger RR, van der Harst E, van der Ham F, et al. Prognostic value of p53, bcl-2, and c-erb B-2 protein expression in phaeochromocytomas. J Pathol 188:51–55, 1999.
Thompson LD. Pheochromocytoma of the adrenal gland scaled score (PASS) to separate benign from malignant neoplasms: a clinicopathologic and immunophenotypic study of 100 cases. Am J Surg Pathol 26:551–566, 2002.
Kimura N, Watanabe T, Noshiro T, Shizawa S, Miura Y. Histological grading of adrenal and extra-adrenal pheochromocytomas and relationship to prognosis: a clinicopathological analysis of 116 adrenal pheochromocytomas and 30 extra-adrenal sympathetic paragangliomas including 38 malignant tumors. Endocr Pathol 16(1):23–32, 2005.
Kubota Y, Nakada T, Sasagawa I, Yanai H, Itoh K. Elevated levels of telomerase activity in malignant pheochromocytoma. Cancer 82:176–179, 1998.
Boltze C, Mundschenk J, Unger N, et al. Expression profile of the telomeric complex discriminates between benign and malignant pheochromocytoma. J Clin Endocrinol Metab 88:4280–4286, 2003.
Elder EE, Xu D, Hoog A, et al. KI-67 and hTERT expression can aid in the distinction between malignant and benign pheochro-mocytoma and paraganglioma. Mod Pathol 16:246–255, 2003.
Isobe K, Yashiro T, Omura S, et al. Expression of the human telomerase reverse transcriptase in pheochromocytoma and neuroblastoma tissues. Endocr J 51:47–52, 2004.
Clarke MR, Weyant RJ, Watson CG, Carty SE, Prognostic markers in pheochromocytoma. Hum Pathol 29:522–526, 1998.
Nagura S, Katoh R, Kawaoi A, Kobayashi M, Obara T, Omata K. Immunohistochemical estimations of growth activity to predict biological behavior of pheochromocytomas. Mod Pathol 12:1107–1111, 1999.
Gupta D, Shildham V, Holden J, Layfield L. Prognostic value of immunohistochemical expression of topoisomerase alpha II, MIB-1, p53, E-cadherin, retinoblastoma gene protein product, and HER-2/neu in adrenal and extra-adrenal pheochromocytomas. Appl Immuno-histochem Mol Morphol 8:267–274, 2000.
van der Harst E, Bruining HA, Jaap Bonjer H, et al. Proliferative index in phaeochromocytomas: does it predict the occurrence of metastases? J Pathol 191:175–180, 2000.
August C, August K, Schroeder S, et al. CGH and CD 44/MIB-1 immunohistochemistry are helpful to distinguish metastasized from non metastasized sporadic pheochromocytomas. Mod Pathol 17:1119–1128, 2004.
Edstrom E, Mahlamaki E, Nord B, et al. Comparative genomic hybridization reveals frequent losses of chromosomes 1p and 3q in pheochromocytomas and abdominal paragangliomas, suggesting a common genetic etiology. Am J Pathol 156:651–659, 2000.
Dannenberg H, Speel EJ, Zhao J, et al. Losses of chromosomes 1p and 3q are early genetic events in the development of sporadic pheochro-mocytomas. Am J Pathol 157:353–359, 2000.
van't Veer LJ, Paik S, Hayes DF: Gene expression profiling of breast cancer: a new tumor marker. Clin Oncol 23:1631–1635, 2005.
Valk PJ, Delwel R, Lowenberg B. Gene expression profiling in acute myeloid leukemia. Curr Opin Hematol 12:76–81, 2005.
Eisenhofer G, Huynh TT, Pacak K, et al. Distinct gene expression profiles in norepinephrine- and epinephrine-producing hereditary and sporadic pheochromocytomas: activation of hypoxia-driven angiogenic pathways in von Hippel-Lindau syndrome. Endocr Relat Cancer 11:897–911, 2004.
Author information
Authors and Affiliations
Corresponding author
Additional information
Part of this work has been presented at the 20th European Society for Pathology in Paris, France, September, 2005.
Rights and permissions
About this article
Cite this article
de Krijger, R.R., van Nederveen, F.H., Korpershoek, E. et al. New developments in the detection of the clinical behavior of pheochromocytomas and paragangliomas. Endocr Pathol 17, 137–141 (2006). https://doi.org/10.1385/EP:17:2:137
Issue Date:
DOI: https://doi.org/10.1385/EP:17:2:137