Abstract
Background: Stress responses have been studied extensively in animal models, but effects of major life stress on the human brain remain poorly understood. The aim of this study was to determine whether survivors of a major earthquake, who were presumed to have experienced extreme emotional stress during the disaster, demonstrate differences in brain anatomy relative to individuals who have not experienced such stressors.
Methods: Healthy survivors living in an area devastated by a major earthquake and matched healthy controls underwent 3-dimentional high-resolution magnetic resonance imaging (MRI). Survivors were scanned 13–25 days after the earthquake; controls had undergone MRI for other studies not long before the earthquake. We used optimized voxel-based morphometry analysis to identify regional differences of grey matter volume between the survivors and controls.
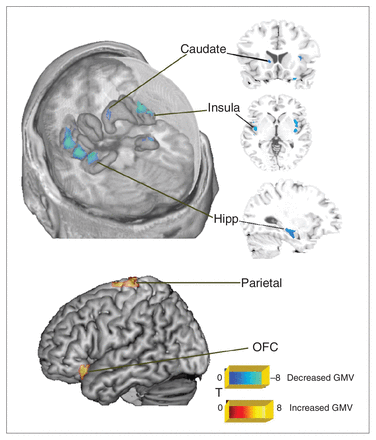
Results: We included 44 survivors (17 female, mean age 37 [standard deviation (SD) 10.6] yr) and 38 controls (14 female, mean age 35.3 [SD 11.2] yr) in our analysis. Compared with controls, the survivors showed significantly lower grey matter volume in the bilateral insula, hippocampus, left caudate and putamen, and greater grey matter volume in the bilateral orbitofrontal cortex and the parietal lobe (all p < 0.05, corrected for multiple comparison).
Limitations: Differences in the variance of survivor and control data could impact study findings.
Conclusion: Acute anatomic alterations could be observed in earthquake survivors in brain regions where functional alterations after stress have been described. Anatomic changes in the present study were observed earlier than previously reported and were seen in prefrontal–limbic, parietal and striatal brain systems. Together with the results of previous functional imaging studies, our observations suggest a complex pattern of human brain response to major life stress affecting brain systems that modulate and respond to heightened affective arousal.
Introduction
Stress responses have been studied in animal models for some time. Human studies demonstrate that stressful life events have both short- and long-term physiologic effects that are mediated by several factors, including genetics, cognitive style and social support.1 However, effects of serious psychological stress on the human brain remain poorly understood. Given the well-established chronic psychological disability that many individuals experience after extreme life stress, there is a need for a better understanding of the neuro-biological effects of stress and an improved ability to identify individuals who most need early mental health interventions.
Previous studies have reported altered brain structure in patients with chronic posttraumatic stress disorder (PTSD), particularly involving the hippocampus, amygdala, anterior cingulate cortex, subcallosal cingulate cortex and medial frontal gyrus.2,3 However, unless response to acute stress is examined, one cannot exclude the potential contributions of increased early life stress, secondary effects related to persistent PTSD or treatment effects on brain changes observed in clinical samples. Thus, studying people from the community shortly after serious psychological stress but before PTSD develops may help advance our understanding of more immediate effects of stress on the human brain.
Ethical concerns limit experimental studies of severe stress response in humans; thus, studying people who survived natural disasters has emerged as a strategy for investigating how people respond to major life stress. To date, few studies have examined the association between life stress and brain structure in adult participants without a history of psycho-pathology or brain disorder.3–6 Moreover, individuals recruited for previous studies had experienced different types of stress-related events, including illness or injury, death of a close friend or relative, unemployment and the ending of important relationships, which might have variable effects on the brain.6 Furthermore, the findings from these studies indicated that grey matter atrophy could only be observed 3 months after stressful life events.6 A report that functional brain alterations have been observed in healthy survivors as early as 25 days after a major earthquake7 raises the question of whether acute neuroanatomical differences could also be detected earlier than 3 months following major emotional stress. To address this issue, we used optimized voxel-based morphometry (VBM) to compare the grey matter volume of physically healthy survivors of the Wenchuan earthquake to that of healthy controls living in the same geographical region who had undergone imaging before the earthquake.
Methods
Participants
We recruited healthy survivors of the Wenchuan earthquake from the local hospital, which serves as the medical centre for southwestern China. The distance between the hospital and the epicentre of the earthquake is 46 miles. Survivors were recruited randomly from the hospital and from communities near the hospital that were the most affected geographic regions, where peak seismic intensity ranged from 9 to 11 on the Mercalli intensity scale. In these regions, thousands of individuals were buried and died under collapsed buildings, and the community remained in fear of intense aftershocks. The survivors underwent scanning within 25 days after the earthquake. To be included in our analysis, survivors must have physically experienced the earthquake, experienced no severe personal medical injury and personally witnessed death or serious injury or the collapse of buildings. All survivors were recruited by poster advertisement in which a free magnetic resonance imaging (MRI) examination of the brain was offered. Each survivor spent about 2 hours in our laboratory for MRI examination and psychological analysis, and no obvious aftershock occurred during the study period.
We recruited healthy controls matched for age, sex, height, weight and years of education randomly from Chengdu, which is located within 50 miles of the earthquake epicenter. They were recruited by poster advertisement, and they had undergone MRI for other studies10,11 shortly before the earthquake.
The following exclusion criteria applied to both groups: the existence of any neurologic or psychiatric disorder, any recent medications known to affect brain function, alcohol or drug abuse, pregnancy or any systemic physical illness, such as hepatitis or diabetes.
An experienced psychiatrist (X.-Q.H.) ensured participants met the inclusion criteria. After that, the researchers (L.C., H.-F.C., S.L. or X.-Q.H.) presented and explained to all participants a standard consent form and the aim, detailed procedures and information about the MRI scanner for this study. All participants were told they could quit the study at any time for any reason.
All participants completed the Structured Clinical Interview for DSM-IV disorders (SCID)12 to rule out a current or past psychiatric disorder, especially anxiety disorders. Two psychiatrists (X.-Q.H. and W.Z.) performed clinical assessments before the MRI examination. Levels of anxiety and depression were evaluated using the Self-Rating Anxiety Scale (SAS)8 and the Self-Rating Depression Scale (SDS).9 Brain MRI scans (i.e., T1- and T2-weighted images) were inspected by an experienced neuroradiologist (S.L.). The 2 groups were scanned on the same research-dedicated 3 T MRI system. The ethics committee of West China Hospital approved our study protocol, and all partcipants provided written informed consent.
Data acquisition
We acquired high-resolution T1-weighted images using a 3 T MRI system (EXCITE; General Electric) with a volumetric 3-dimentional spoiled gradient recall (SPGR) sequence (repetition time 8.5 ms, echo time 3.4 ms, flip angle 12°, slice thickness 1 mm) using an 8-channel phase array head coil. We used a field of view of 240 × 240 mm2, with an acquisition matrix comprising 256 readings of 128 phase encoding steps, producing 156 contiguous coronal slices with slice thickness of 1.0 mm. The final matrix of T1-weighted images was automatically interpolated in-plane to 512 × 512, which yields an in-plane resolution of 0.47 × 0.47 mm2. To assure the quality of the data acquired at different times, a quality assurance scan was acquired, including a spin echo sequence to warm up the scanner and verify the signal-to-noise ratio of images, before each participant was examined.
Image analysis
Image preprocessing and statistical analyses were performed with SPM8 (www.fil.ion.ucl.ac.uk/spm) using the VBM tool-box (VBM8). First, we constructed a whole brain template, and the individual native images of all participants were normalized to the whole brain template and segmented into grey matter, white matter and cerebrospinal fluid (CSF). We then generated a grey matter template from all the grey matter images obtained in step 1. Third, the native images were segmented again into different tissue types and normalized to the grey matter template. Next, the deformation parameters obtained from the preceding step were applied to the original raw images of all participants to create optimally normalized whole brain images, which were recursively segmented into grey matter, white matter and CSF. Next, reobtained grey matter images underwent Jacobian modulation (volume modulation). Finally, we smoothed the grey matter images with a 6 mm full-width at half-maximum Gaussian kernel. We used the images to compare grey matter volume.
Statistical analysis
Sex differences between groups were assessed with a χ2 test, and age, height, weight and years of education were compared using 2-sample t tests. These analyses used a statistical threshold of p < 0.05, and no significant group differences were found.
Voxel-wise comparisons of grey matter volume were performed between groups using 2-sample t tests. Global brain volume was used as a covariate because volume measurements were performed in native space. The significance of group differences in each region was estimated by distributional approximations from the theory of random Gaussian fields. We performed nonstationary correction using the “Threshold and transform spmT-maps” function.13 All t values were evaluated under a false discovery rate (FDR; pFDR < 0.05 threshold).
Region of interest (ROI) analyses were performed by averaging the grey matter volume values of voxels within each a priori ROI, as defined in the WFU pickatlas.14,15 These ROIs included the insula, caudate, hippocampus, parietal cortex and orbitofrontal cortex (OFC), areas that have been found to be functionally abnormal in people with PTSD.2,3,6,7 We conducted ROI analyses in part to identify associations between structural brain abnormalities and clinical symptom severity in exploratory studies. Average grey matter volume values of the ROIs were extracted from the survivor group, and these were correlated with age, with the number of days post-earthquake that scans were performed and with anxiety and depression scores on the SAS and SDS, respectively. Furthermore, we extracted the volumes of the anterior and posterior parts of the hippocampus separately using Freesurfer (http://surfer.nmr.mgh.harvard.edu/, version 4.5.0), and compared them between the survivors and controls, adjusting for total intracranial contents.
Scores on the SAS of 55 or greater are considered to indicate clinically severe anxiety.8 Therefore, we subdivided our survivors into those with severe anxiety (SAS+) and those without clinically severe anxiety (SAS−). We then compared the grey matter volume between the SAS+ and the SAS− groups using 2-sample t tests with global brain volume as a covariate.
Results
Participants
We included 44 survivors (17 female, mean age 37 [standard deviation (SD) 10.6] yr) and 38 controls (14 female, mean age 35.3 [SD 11.2] yr) in our analysis. The demographic and clinical characteristics of participants are summarized in Table 1. Survivors were scanned within 25 (range 13–25 d; mean 21 [SD 3]) days after the earthquake. Among the 44 survivors, 6 had lost 1 or more immediate family members, and the others had at least 1 family member with a severe injury, including bone fractures, head trauma or abdominal/chest injury. Fifteen survivors permanently lost their jobs, and all survivors lost their homes. At the time of the study, 21 survivors lived in their relatives’ homes in Chengdu, 13 survivors lived in the temporary accommodation provided by the local government, and the other 10 survivors lived in the temporary accommodations provided by our hospital. The living quarters of all the participants were within 6.5 miles of our hospital, and all of them could keep in touch with their families by cell phone. Healthy controls were scanned for other studies10,11 shortly (range 7–56, mean 41 [SD 14] d) before the earthquake.
Demographic and clinical characteristics of physically healthy trauma survivors and healthy controls
No participants had ever taken any psychiatric medication. No gross abnormalities suggesting neurologic disease were observed for any participant.
Voxel-based analysis
Compared with controls, the survivors showed significantly lower grey matter volume in the bilateral insula, hippocampus, left putamen and left caudate, and greater grey matter volume in the bilateral OFC and parietal lobe (p < 0.05 corrected for multiple comparisons; Table 2, Fig. 1). Survivors with (SAS+ group: 15 male, 9 female; mean age 37.9 [SD 10.1] yr, mean SAS score 57 [SD 4.6]) and without (SAS− group: 12 male, 8 female; mean age 35.9 [SD 11.2] yr, mean SAS score 38 [SD 6.9]) clinically severe anxiety showed no significant differences in grey matter volume (p > 0.05, corrected for multiple comparisons).
Voxel-based analysis showing regions with greater (red areas) and lower (blue areas) grey matter volume in survivors compared with controls (p < 0.05, corrected). Grey matter volumes (GMV) of the bilateral insula, hippocampus (Hipp), ventral striatum (caudate and putamen), orbitofrontal cortex (OFC) and parietal lobe differed significantly between survivors and controls (all p < 0.05, corrected).
Voxel-based analysis of grey matter volumes in trauma survivors and healthy controls
Region of interest analysis
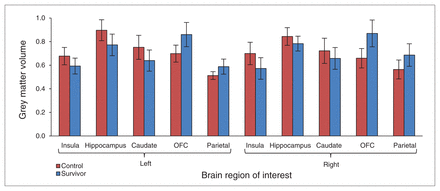
As expected, ROI analysis yielded findings paralleling those in the voxel-based analysis. Grey matter volume was lower in limbic and subcortical regions, including the bilateral insula, hippocampus and caudate, and grey matter volume was greater in the bilateral OFC and parietal lobe (all p < 0.05; Fig. 2) in the survivor group than the control group. Furthermore, the mean (and SD) volume of the anterior part of the bi-lateral hippocampus in the survivor group (left: 1.9 [0.4], right: 2.1 [0.3]) were lower than those of controls (left: 2.1 [0.3], right: 2.2 [0.5]; p < 0.05), whereas the volume of the posterior part of the bilateral hippocampus in the survivor group (left: 1.6 [0.3], right: 1.5 [0.6]) did not differ from those of controls (left: 1.5 [0.4], right: 1.5 [0.5]; p > 0.05). Correlation analyses in the survivor group did not reveal any significant associations between the average grey matter volume values in brain regions in which abnormal volumes were detected and anxiety, depression, age or days from earthquake to the brain scan.
Region of interest analysis (mean and standard deviation) of the mean grey matter volume in the bilateral insula, hippocampus, ventral striatum (caudate), orbitofrontal cortex (OFC) and parietal lobe. The results showed significantly different grey matter volume between survivors and controls in all of these regions (p < 0.05, corrected).
Discussion
Survivors of a severe earthquake who had no history of psychiatric illness showed greater grey matter volume in areas of the association cortex and lower grey matter volume in the insula, hippocampus and ventral striatum than controls. These findings, evident within only 1 month after the earthquake, are consistent with results of animal studies showing that new experience can lead to both the formation and elimination of synapses in the cortex.16 In the context of major life stress, volume reductions in the insula, ventral striatum and hippocampus may reflect disruptions to neuronal integrity or synaptic processes that are a result of high levels of sustained neural activity in affective brain systems.17–19 Such effects may parallel previous findings in patients with a mood disorder showing increased functional activity and lower grey matter volume within affective brain circuitry, which are believed to represent neurotoxic effects of increased neural activity.20–23
Recent evidence supports a role of the OFC as an important control region for modulating affective responses in limbic structures, including the hippocampus, amygdala, ventral striatum and insula.24 Thus, greater grey matter volume in the OFC may be a consequence of increased demands for top–down modulation to reduce sustained hyperactivation in limbic systems. Greater grey matter volume in the parietal lobe may reflect enhanced activity related to the hypervigilant state of many trauma survivors25 who actively scan the environment for perceived threat. In contrast to apparent excitotoxic effects of sustained high levels of neural activity in affective brain circuitry, greater grey matter volume in the parietal cortex and OFC may result from elevated synaptogenesis, dendritic branching, increased dendritic spine density17–19 or neurogenesis.26 Such effects have been seen in studies of rats exposed to new experiences. Another contributing factor might be an increased regional cerebral blood flow27 due to increased neural activity in these regions.7
Though the mechanism for lower volume in the insula, ventral striatum and hippocampus is unclear, several possibilities may conrtribute to this change. First, acute stress is known to elevate levels of corticotropin-releasing hormone and activate the hypothalamic–pituitary–adrenal axis. That process can exert a profound effect on neurogenesis, leading to both a rapid and prolonged decrease in the rate of cell proliferation and an increase in apoptotic cell death, especially in limbic regions, such as the hippocampus.28 These processes may contribute to volumetric differences in limbic circuitry observed in our participants. In addition, heightened sustained excitatory drive from the OFC to local circuit inhibitory neurons29 in brain emotion systems might lead to habituation or other alterations in local inhibitory circuitry that could indirectly enhance excitotoxic processes.28
In fact, the brain regions with altered grey matter volume in survivors belongs to the prefrontal-limbic and striatal systems, which have been recognized to be involved in affective processing30 and decision-making.31 Functional neuroimaging studies have provided direct evidence that prefrontal-limbic and striatal systems play a critical role in anxiety disorders, including the recollection of traumatic memories and the processing of fear and pain,32,33 whereas the striatum and parietal regions are activated when making decisions under time pressure.31 Functional alterations in emotion circuitry have been described previously in individuals who have experienced major life stresses.7 Our observations in the present study indicate that not only functional or physiologic changes, but also robust differences in gross brain anatomy, could be observed within 1 month after a traumatic event. While gross brain anatomy has long been considered to be relatively fixed in adult life outside the context of neurologic disease, several lines of recent research show acute effects of learning34–36 and psychiatric medications37 on brain anatomy. Furthermore, we also noted that the anterior rather than the posterior part of the hippocampus showed lower grey matter volume in survivors. This is consistent with the notion that the anterior part of the hippocampus was more involved in emotional processing while the posterior part was thought to be involved, for example, in spatial navigation.38 The rapid anatomic changes in the limbic cortex in a short time after major life stress, as shown in the present study, have not been previously reported in humans. The severe nature of the emotional stress experienced by participants in the present study may have contributed to our ability to detect anatomic changes earlier than previously reported. The present findings contribute to our understanding of human stress response and provide novel evidence indicating a potential for robust and rapid neuroplasticity in emotional brain systems.
Both animal models and patient studies demonstrate that functional and anatomic changes in prefrontal-limbic and striatal systems play a critical role in stress-related psychiatric disorders.21,31,33,39,40 Our neuroanatomic findings may be of particular clinical relevance for these conditions by showing structural changes in brain areas where stress-related physiologic changes have been previously reported. The present findings suggest that anatomic changes observed previously in patients with chronic stress-related disorders may in fact occur acutely, and perhaps early in the course of these disorders, rather than represent slowly progressive alterations or secondary effects of chronic distress and disability. Although longer term follow-up studies are necessary to document the full time course of anatomic changes seen in stress-related psychiatric disorders and after acute stressful events, our data indicate that substantial structural neuroplasticity in clinically relevant brain circuitry may manifest itself shortly after massive traumatic events. This observation provides further neurobiologic rationale for early mental health interventions after major life stressors to reduce acute stress responses and potentially minimize longer term morbidity.
Although a resting-state fMRI study of earthquake survivors has previously shown functional changes within similar brain regions where anatomic changes were observed in the present study, those functional changes were related to anxiety ratings.7 In contrast, anatomic differences reported here were not related to anxiety or depression ratings. This may reflect a different or lagging manifestation of anatomic alterations that could result from physiologic and affective changes that are manifested earlier, which would suggest a different time course to their clinical relevance. Further, to the extent that different levels of increased neural function may have a different magnitude and time course of effect across brain regions and involve a complex interplay of pathological and compensatory processes, there may be considerable variability across individuals in the association between anatomic changes and immediate psychological distress. It seems most plausible that physiologic changes may more closely parallel emotional distress acutely, whereas anatomic alterations may be more related to cumulative consequences of persistent emotional distress and potentially other secondary effects. Longitudinal research is needed to evaluate these associations.
Limitations
It is important to consider that the use of peak volumetric differences in different brain regions as correlates for the SAS and SDS scores may have led to an underestimation of their association given intersubject variation in such measures.41 Differences in the variance of survivor and control data also could impact study findings. Thus, studies to replicate our observations are needed to confirm and validate our results, especially in individuals who experienced stressors of different types, duration and intensity. Furthermore, other factors, such as physical stress, nutritional deprivation and sleep disturbances, could have contributed to brain changes,42 though the affected regions observed in the present study were more likely to be related to emotion process.
Conclusion
To our knowledge, the present study demonstrated for the first time that individuals who experience severe emotional trauma show stress-induced neuroanatomic alterations shortly (within 25 d) after massively traumatic psychological events. This observation, together with previous reports of functional brain alterations in trauma survivors, suggests that survivors of severe emotional trauma may experience substantial change with respect not only to function, but also to the structural anatomy of prefrontal-limbic, parietal and striatal brain systems. From a public health perspective, the data highlight the need to rapidly intervene to reduce stress levels in people who have experienced major life stress so that acute neurophysiological alterations do not lead to downstream and potentially persistent alterations in psychological and brain function.
Acknowledgements
This study was supported by National Natural Science Foundation (Grant Nos. 81222018,81030027, 81227002 and 81220108013), the Programs for New Century Excellent Talents in University (Grant No. NCET-10-0596) and National Key Technologies R&D Program of China (Program No: 2012BAI01B03) and the von Humboldt Foundation. Q.-Y. Gong acknowledges support from his American CMB Distinguished Professorship Award (Award No. F510000/G16916411), administered by the Institute of International Education. S. Lui is supported by the Distinguished Young Scholars of Sichuan (Award No. 2011JQ0005).
Footnotes
Competing interests: J.A. Sweeney consults for Lilly, Takeda, Roche and Pfizer. As above for Q.-Y. Gong and S. Lui. Otherwise, none declared.
Contributors: S. Lui, X.-Q. Huang and Q.-Y. Gong designed the article. S. Lui, L. Chen and W. Zhang acquired the data, which all authors but X.-Q. Huang analyzed. S. Lui, L. Chen, L. Yao, Y. Xiao, Q.-Z. Wu, X.-Q. Huang, W. Zhang, Y.-Q. Wang and J.A. Sweeney wrote the article. S. Lui, Q.-Z. Wu, J.-R. Zhang, S.-Q. Huang, H.-F. Chen, R.C.K. Chan, J.A. Sweeney and Q.-Y. Gong reviewed the article. All authors approved its publication.
- Received November 30, 2012.
- Revision received February 28, 2013.
- Revision received March 7, 2013.
- Accepted March 7, 2013.