Abstract
Background: Fibrous dysplasia (FD) results from an abnormality in lamellar bone formation and most frequently involves the proximal femur. This can lead to the development of osteoarthritis requiring total hip arthroplasty (THA). Such cases are challenging, and there is a lack of information guiding best management. As such, we devised a study assessing the outcomes and complications in patients with FD undergoing THA with modern implant technology, and we outlined our preferred surgical technique.
Methods: A search of our institutional arthroplasty database was performed to identify patients who underwent THA for FD between January 2001 and July 2018 at Mount Sinai Hospital in Toronto, Canada. Data regarding implants used and the use of allograft material or metal augments or both were obtained. Complications and revision requirements were noted. Radiographic and clinical leg length discrepancies were assessed.
Results: A total of 10 hips in 9 patients who underwent THA for FD were identified. Mean follow-up time was 6.0 years (range 0.5 to 10.3 yr). The majority of patients underwent THA using uncemented femoral and acetabular components with large femoral heads on highly cross-linked polyethylene liners. Most cases (80% of hips) required allograft to the proximal femur. A single complication requiring revision was noted. In 90% of hip surgeries, the patient required transfusion of packed red blood cells. Mean radiographic and clinical leg length discrepancies were 0.9 cm (range −2.4 to 2.4 cm) and 0.9 cm (range −4 to 0 cm), respectively.
Conclusion: Contrary to previous reports, low complication and revision rates were observed with cementless components and routine use of allograft material. The challenging nature of such cases warrants use of an experienced arthroplasty treatment team.
Fibrous dysplasia (FD) is a developmental abnormality caused by failure in the production of normal lamellar bone, and it most frequently involves the proximal femur. The disease is characterized by bone deformities, pain and pathologic fractures. The cause of this benign tumour of bone is unclear, but the condition is believed to result from a mutation in the Gsa subunit of protein G, leading to abnormalities in osteoblast differentiation. Additionally, an increase in osteoclastic bone resorption induced by interleukin-6 is observed.1,2 Some patients may present with endocrine abnormalities such as precocious puberty and cutaneous café au lait spots. Likewise, an association has been observed with multiple endocrine and nonendocrine disorders, such as McCune–Albright and Mazabraud syndromes.1,2 Fibrous dysplasia can be monostotic or polyostotic, and it represents approximately 7% of all benign tumours of bone.1 Twelve percent of patients with FD will develop osteoarthrtitis of the hip and may require total hip arthroplasty (THA) during their lifetime.3
These cases represent a significant technical challenge to orthopedic surgeons because of poor bone quality and deformities resulting from the disease. Unfortunately, there is very little information on best practices to guide management of these cases. We are aware of a single study assessing THAs performed between 1969 and 2001.4 There is, therefore, a need to provide the orthopedic community with an update on patient outcomes and surgical techniques that use modern implant systems and materials to treat FD.
We therefore undertook a retrospective study to assess the outcomes and complications in patients with FD undergoing THA. Additionally, from the available evidence and the results of our study, we aimed to devise treatment recommendations to help guide surgeons in the management of this challenging population of patients undergoing hip arthroplasty.
Methods
We performed a retrospective search of our institutional prospective arthroplasty database to identify patients who underwent THA for FD between January 2001 and July 2018 at Mount Sinai Hospital in Toronto, Canada. For each patient identified in the search, we noted any complications and whether revision surgery was required. The form of FD (monostotic or polyostotic) was documented. We calculated the radiographic hip centre of rotation distance for each patient by measuring the vertical position of the centre of rotation of the femoral head relative to a line drawn parallel to the interteardrop line.5 Postoperative clinical leg length discrepancies (LLDs) were obtained at the time of follow-up visits. Lastly, data regarding use of allograft material or metal augments or both were collected. Mean values and ranges were calculated using SPSS version 25 (IBM). Ethics approval was obtained from the Mount Sinai Hospital Research Ethics Board.
Surgical procedures were performed either through a lateral approach or using an extended trochanteric osteotomy. Blood was made available in the operative suite. Removal of previous hardware was staged, and neck cuts were made in situ if there was concern about intraoperative femoral neck fracture. Trochanteric osteotomy was used to improve visualization of the dysplastic proximal segment for debulking of lesional tissue or for application of a proximal femoral allograft (PFA) or for both. Distal fixation into normal bone was attained whenever possible by a minimum of 2 cortical widths.
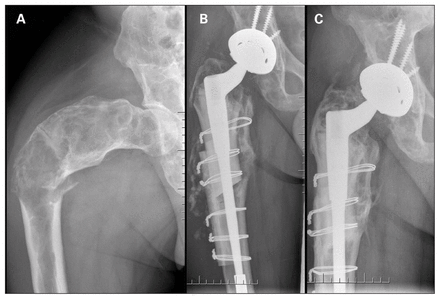
At our centre, regular use of PFA material is preferred when a large segment of the femur is noted to be diseased. When significant deformity is present within the femur, a corrective osteotomy is performed to facilitate later passage of the femoral component. Subsequently, all abnormal fibrous tissue is curetted from the proximal femoral segment. If bone quality is noted to be especially poor, an extended trochanteric osteotomy is performed in preparation for a PFA. The definitive prosthesis is then cemented into the PFA on a back table, and the distal aspect of the stem is inserted into the distal healthy bone segment (Fig. 1). The junction between the allograft and native bone, and corrective osteotomy sites, are routinely reinforced with cortical strut allograft or spanning plate fixation or both (Fig. 2). Definite stability between the PFA and native bone is achieved through the process of creeping substitution.6
Preoperative radiograph demonstrating a shepherd’s crook deformity of the proximal femur with a subtrochanteric pathologic fracture (A), and films at 1 month (B) and 7.5 years (C) after total hip arthroplasty using proximal femoral allograft.
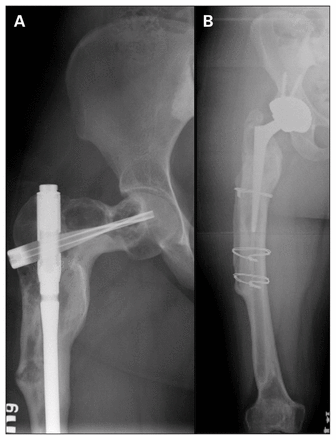
Preoperative (A) and postoperative (B) radiographs of a patient with polyostotic fibrous dysplasia who underwent total hip arthroplasty with use of a cortical strut allograft.
Results
During the study period, 9 patients (10 hips) underwent THA for sequelae relating to FD of the proximal femur. Seven of the patients were men and 2 were women. All patients had their surgery performed between 2008 and 2018. Mean follow-up time was 6.0 years (range 0.5 to 10.3 yr). The average age of patients at the time of surgery was 52.3 years (range 31 to 74 yr). There were 5 monostotic and 4 polyostotic patient cases. All patients with polyostotic FD were noted to have some degree of acetabular involvement that was noted at the time of surgery. None of the patients with monostotic FD had gross involvement of the acetabulum.
Complications
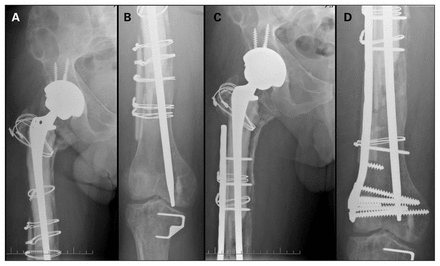
We encountered a single surgical complication requiring early revision surgery (10% of hips, 95% confidence interval 0.3 to 44.5) (Table 1). The patient in question required revision of the femoral component because of subsidence of the cemented implant into the knee joint (Fig. 3). In this case, the femoral implant was placed very distally because of the extensive bony involvement and poor bone quality. Two patients were noted to have had transient sciatic nerve palsy postoperatively, but they recovered fully without further intervention over a period of 4 to 12 weeks. In 9 hip surgeries, the patient had significant blood loss requiring transfusion of packed red blood cells either intraoperatively or in the postoperative period.
Radiographs of a 38-year-old man with stem subsidence into the knee before (A and B) and after (C and D) revision surgery.
Complications
Implants
A variety of surgical implants were used (Table 2). Uncemented stems were employed for the majority of patients (6/10 hips). The acetabular components used were revision porous-metal multihole shells in 9 THA surgeries when concern about acetabular involvement and bone quality was noted. Large-diameter (≥ 36 mm) heads were inserted in 6 of the 10 hips. Head material was either cobalt-chrome (8 hips) or ceramic (2 hips). All liners were composed of highly cross-linked polyethylene. Elevated-rim liners were used in 7 of the 10 hips. Abductors were noted to be deficient or defunctioned in patients with more severe deformity of the proximal segment. This issue was addressed with meticulous abductor repair or advancement or both, and constrained liners were not required.
Characteristics of surgical implants
Allograft requirements
Eighty percent of cases (8 hips) required allograft to the proximal femur (Table 3). Proximal femoral allograft was used in 6 cases (60%), and cortical strut allograft was used in 2 cases (20%) (Fig. 1, Fig. 2). Allograft material was used in 2 of the 5 monostotic hips (40%) and in 4 of the 5 polyostotic cases (80%). No patients required acetabular allograft material, and none required metal augments.
Use of allografts and augments
Radiographic and clinical outcomes
Mean radiographic hip centre of rotation difference and clinical LLDs were small (Table 4). Radiographic measurements demonstrated that the absolute affected hip centre of rotation difference was 0.9 cm (range −2.4 to 2.4 cm). Likewise, clinical measurements demonstrated a mean shortening of the ipsilateral limb of 0.9 cm (range −4 to 0).
Clinical and radiographic leg length discrepancies
Discussion
It is estimated that patients with FD have approximately 7% of all benign bony tumours.1 A significant number of these patients will require THA in their lifetime.3 Unfortunately, there is a lack of literature reporting the results and complications of this surgery in FD patients with modern implant technology and techniques. Furthermore, there is minimal guidance for surgeons treating these challenging cases.
To our knowledge, Sierra and colleagues published the only study on the topic of THA in the FD population.4 They reported on 11 patients treated between 1969 and 2001 and found that although patients had excellent pain relief and function, higher complication rates at the time of surgery and need for revision were concerning. In their cohort, cemented stems were used in 7 hips, and fully or proximally coated cementless stems were used in 5 hips. Unfortunately, further implant details were lacking. Sierra and colleagues found that 7 hips in 6 patients required revision because of aseptic loosening at an average of 12.5 years. Additionally, a much higher proportion of patients with polyostotic compared with monostotic FD required revision (75% and 25%, respectively). Furthermore, more than 40% of patients who underwent THA with a cementless stem required early revision because of loosening. The authors described a technique consisting of bypassing bone defects with cemented long-stemmed femoral components, and they expressed caution about the use of cementless femoral stems. They suggested that the advent of modern conical fluted stems may provide better diaphyseal fixation, which could translate into improved results with cementless technology.
The results of our study contrast with those of Sierra and colleagues in that we did not experience a similar high level of perioperative complications. Furthermore, substantially more uncemented stems were used in our study population, and we did not observe any early loosening of the prostheses with modern implant systems. We speculate that the differences in the results of the 2 studies are secondary to improvements in cementless ongrowth technology and differences in surgical technique. Conical fluted stems were the most commonly used uncemented prostheses in our study, which may have allowed for better diaphyseal fixation distal to the affected segment. Additionally, multihole porous-metal revision shells were used in almost all of our cases. It is routine practice at our centre to adopt a low threshold for using such porous implants when acetabular involvement is suspected or when bone quality and ingrowth potential are of concern. Our cohort used a high volume of allograft material in the proximal femur: 40% for monostotic and 80% for polyostotic cases. Allograft material was not readily available for all patients included in the study by Sierra and colleagues, as noted by the authors, and this may explain some of the differences in the results. This probably holds especially true for patients with polyostotic FD in whom bone stock has been demonstrated to be particularly poor.7 Furthermore, we noted significant abductor deficiency and defunctioning in cases with severe deformity, which was not discussed in previous series. This was addressed by abductor repair or advancement or both, without the need for constrained liners.
Limitations
There are several limitations to our study. We acknowledge that the sample size is small and limited to a single centre where allograft material is readily available in hospital. As such, our results may not be reproducible in other regions with different treatment practices and experience. The aim of this article was, therefore, to focus on treatment principles from our experience and provide guidance rather than fixed guidelines. We also acknowledge the possible effect of variable severity of FD in our patient cohort. Unfortunately, a grading system does not currently exist for FD. This limits the comparability of results among studies. Additionally, in our study there was significant variability in the time since surgery. Although we had several patients with long- and mid-term followup, some patients had undergone surgery more recently. As such, we chose to focus on early failure as our primary outcome measure, because previous reports had noted high rates of early revision with cementless implants.4 It is, however, possible that implant failure requiring revision surgery could develop in these patients over the longer term. Lastly, although we had planned to measure leg lengths both radiographically and clinically, we were limited in our ability to measure true radiographic LLD because of the lack of reproducible bony landmarks in this patient population. Instead, we used the difference in hip centre of rotation distance as a surrogate, which may have affected our results.
Conclusion
This study provides the orthopedic community with an update regarding primary THA performed in patients with FD. Contrary to the findings of previous reports, we observed low complication and revision rates in the early postoperative period with cementless components and routine use of allograft material when bone quality was poor. Improvements in modern implant on growth surfaces and differences in surgical technique are likely to play a role in explaining the differences with earlier studies. An emphasis on obtaining distal cementless fixation i nto healthy bone, while cementing into PFA when bone quality is especially poor, appears to lead to satisfactory results. High rates of transfusion in the perioperative period, however, should alert the treating surgeon to be prepared for elevated levels of blood loss. The challenging nature of these cases warrants involvement of an experienced arthroplasty treatment team to ensure optimal outcomes.
Footnotes
Presented at the Canadian Arthroplasty Society Annual General Meeting, Nov. 22–23, 2018, Toronto, Ont.
Competing interests: O. Safir has served as a paid speaker and consultant for Zimmer Biomet and has been reimbursed by Zimmer Biomet for travel expenses for these activities. A. Gross has received educational consulting fees from Zimmer Biomet. No other competing interests were declared.
Contributors: All authors designed the study. S. Garceau acquired the data, which S. Garceau and P. Kuzyk analyzed. S. Garceau and J. Wolfstadt wrote the article, which all authors critically reviewed. All authors provided final approval of the article for publication.
- Accepted September 17, 2019.