Abstract
Background: There is increasing evidence that psychotherapy can alter the function of the brain of patients with borderline personality disorder (BPD). However, it is not known whether psychotherapy can also modify the brain structure of patients with BPD.
Methods: We used structural MRI data of female patients with BPD before and after participation in 12 weeks of residential dialectical behavioural therapy (DBT) and compared them to data from female patients with BPD who received treatment as usual (TAU). We applied voxel-based morphometry to study voxel-wise changes in grey matter volume over time.
Results: We included 31 patients in the DBT group and 17 in the TAU group. Patients receiving DBT showed an increase of grey matter volume in the anterior cingulate cortex, inferior frontal gyrus and superior temporal gyrus together with an alteration of grey matter volume in the angular gyrus and supramarginal gyrus compared with patients receiving TAU. Furthermore, therapy response correlated with increase of grey matter volume in the angular gyrus.
Limitations: Only women were investigated, and groups differed in size, medication (controlled for) and intensity of the treatment condition.
Conclusion: We found that DBT increased grey matter volume of brain regions that are critically implicated in emotion regulation and higher-order functions, such as mentalizing. The role of the angular gyrus for treatment response may reside in its cross-modal integrative function. These findings enhance our understanding of psychotherapy mechanisms of change and may foster the development of neurobiologically informed therapeutic interventions.
Introduction
Changes in the structure of the adult human brain have long been thought to be limited to aging or pathological conditions. Research in recent decades has challenged this view by providing compelling evidence that learning new behaviour can alter the structure of the adult human brain.1
This learning-dependent structural plasticity has been shown for activities such as juggling,2 musical training3 or acquisition of a great amount of abstract information4 and has recently been expanded to psychotherapy. Two years of cognitive remediation therapy increased grey matter volume in the fusiform gyrus, hippocampus and amygdala5 as well as fractional anisotropy in the genu of the corpus callosum in patients with schizophrenia.6 Ten weeks of cognitive behavioural group therapy reduced grey matter volume in parieto-occipital and prefrontal regions and increased fractional anisotropy in the uncinate and inferior longitudinal fasciculus and structural connectivity in a frontolimbic network in patients with social anxiety disorder.7 In contrast, 16 weeks of brief eclectic psychotherapy showed no effect on the (para-)hippocampus and amygdala in patients with posttraumatic stress disorder.8 However, to our knowledge, no study to date has investigated the effects of psychotherapy on the brain structure of patients with borderline personality disorder (BPD).
Borderline personality disorder is a severe and common mental disorder, and emotion dysregulation has been regarded as its core feature.9–11 Neurobiologically, this is reflected in a prefrontal-limbic imbalance (i.e., a dysfunctional brain pattern in which regulatory areas, especially prefrontal regions, fail to control enhanced reactivity of regions regarded as eliciting affectivity, such as the amygdala).12 Accordingly, a recent meta-analysis reported increased activation of the left amygdala and posterior cingulate cortex together with reduced activation of the bilateral dorsolateral prefrontal cortex13 to negative versus neutral stimuli in patients with BPD compared with healthy controls. Structural neuroimaging studies have corroborated these findings by revealing reduced grey matter volume in prefrontal and limbic areas, including the anterior cingulate cortex (ACC), inferior frontal gyrus, amygdala and hippocampus in patients with BPD compared with healthy controls.13–17
In addition, BPD has become increasingly associated with dysfunction in higher-order brain functions, such as mentalizing.18–20 Mentalizing, or theory of mind, refers to the capacity to make sense of others or ourselves and has been identified as a mechanism of psychotherapeutic change in patients with BPD.21 Consistent with this, functional (see the meta-analysis by Ruocco and colleagues22) and structural17,23 neuroimaging studies reported alterations of brain structures critical for mentalizing, such as the superior temporal gyrus, in patients with BPD compared with healthy controls.
Dialectical behaviour therapy (DBT) is the most intensively studied psychotherapy for BPD and has been shown to be superior to treatment as usual (TAU) and a client-centred therapy control condition.24 Initially developed as an outpatient treatment25 DBT was later adapted to a 12-week residential treatment.26,27 Based on Linehan’s biosocial developmental model of BPD,9 DBT has a special focus on improving emotion regulation.
Up to now, 5 functional neuroimaging studies have investigated how DBT affects neural activity. The first 2 studies28,29 assessed neural correlates of patients with BPD compared with healthy controls during the processing of negative stimuli and reported reduced activity in the ACC, posterior cingulate and insula after 12 weeks of residential DBT,28 and decreased amygdala activation after 1 year of outpatient DBT.29 The other 3 studies30–32 examined patients with BPD before and after 12 weeks of residential DBT in comparison to patients with BPD receiving TAU and a group of healthy controls. They found that DBT changed neural correlates of pain-mediated emotion regulation, marked by normalization of amygdala activity and altered connectivity between the left amygdala and dorsal ACC30 and during emotion regulation with the strategies distraction and reappraisal: after DBT, patients showed decreased activity of the supramarginal gyrus and perigenual ACC31 and decreased activity of the insula and ACC together with increased connectivity of the ACC with prefrontal, temporal and parietal regions.32
To address the question of whether psychotherapy can also alter the brain structure of patients with BPD, we used structural MRI data of patients with BPD before and after 12 weeks of residential DBT and compared them to data of patients who received TAU. We applied a well-established methodology (voxel-based morphometry) that allowed us to study voxel-wise changes in grey matter volume over time. Owing to the absence of prior studies, we did not restrict our analysis to specific brain regions, but instead conducted a whole brain analysis. We hypothesized that 12 weeks of residential DBT would lead to an increase in grey matter volume in brain regions that are explicitly involved in cognitive regulation of emotional processes, such as the rostral and dorsal ACC and the inferior frontal gyrus, and in mentalizing, such as the superior temporal gyrus. In addition, we explored associations between changes in grey matter volume and treatment response.
Methods
Participants
We recruited patients with a current diagnosis of BPD according to DSM-IV to take part in the present study. They were recruited directly at the treatment units (DBT group) or via licensed psychotherapists, the resident’s registration office and advertising on the Internet (TAU group). Two-thirds of participants received 12 weeks of standard residential DBT consisting of individual and group-based skills training (DBT group).26,27 Treatment was administered at 2 specialized inpatient units at the University of Heidelberg in Mannheim or Heidelberg. Therapists were experienced doctoral- or masters-level clinicians and were supervised regularly. The remaining patients with BPD (TAU group) continued to receive the same treatment they were undergoing at study entry (e.g., outpatient psychotherapy, residential crisis intervention, pharmacotherapy, and no treatment). No patient in the TAU group was waiting for DBT treatment. Treatment assignment followed a natural course (i.e., it was based on the preferences of the individual patient).
Both groups were measured after a 12-week interval. None of the participants had significant prior experience with DBT and all were either unmedicated or medication was kept constant during the study period of 12 weeks plus 2 weeks before the first measurement.
General exclusion criteria for all participants were traumatic brain injury; left-handedness; lifetime diagnoses of schizophrenia, bipolar I disorder, or developmental disorders; substance dependence during the last year; illicit substance consumption during the last 2 months; current severe depressive episode; and current benzodiazepine use.
The present study was part of a larger project on the neural correlates of DBT in patients with BPD registered as a clinical trial (German Clinical Trial registration ID DRKS00000778) that included patients with BPD and healthy controls. Following our research question and to reduce the risk of type I errors, we restricted the present analysis to the patients with BPD. The study was conducted according to the Declaration of Helsinki, and the ethics committees of the 2 medical faculties of the University of Heidelberg approved the study. All participants provided written informed consent and were financially compensated for their participation.
Measures
The diagnosis of BPD and comorbid psychiatric disorders were determined in face-to-face interviews using the International Personality Disorder Examination33 and the Structured Clinical Interview for DSM-IV.34 The interviewers had a psychological or medical background and had obtained an advanced academic degree (i.e., master of science or medical doctorate) and were trained in multi-day courses by senior clinicians to use the diagnostic instruments.
Additionally, we used the following measures: the Difficulties in Emotional Regulation Scale (DERS),35 which measures emotion dysregulation; the Zanarini Rating Scale for Borderline Personality Disorder (ZAN-BPD),36 which assesses the change in DSM-IV-TR (identical to DSM-5, section II) BPD symptomatology; and the Borderline Symptom List (BSL),37 which measures the severity of BPD symptomatology. In contrast to the ZAN-BPD, the BSL additionally captures aspects of BPD symptomatology that are based on the revised version of the Diagnostic Interview for BPD38 and the opinions of both clinical experts and of patients with BPD (propensity to experience excessive guilt or shame, different types of problematic behaviour). In case of missing values, we conducted simple mean imputation. No measure showed more than 3% missing values.
MRI data acquisition and preprocessing
All participants underwent structural scanning at the Central Institute for Mental Health in Mannheim, Germany, on a 3 T Magnetom TIM Trio MR scanner (Siemens Medical Solutions) using a T1-weighted 3-dimensional magnetization prepared rapid gradient echo sequence and a 32-channel standard head coil (image matrix 256 × 256, field of view 256 mm, voxel size 1 × 1 × 1 mm3, repetition time 1570 ms, echo time 2.75 ms, flip angle 15°). Experienced radiologists reviewed MRI scans to exclude clinically important abnormalities, such as structural brain damage or signs of atrophy.
Analyses were performed using the Computation Anatomy Toolbox39 for Statistical Parametric Mapping, version 12 (SPM12).40 Preprocessing for the cross-sectional analysis was performed according to the method of Gaser and Kurth,41 using the default settings. Preprocessing for the longitudinal analysis considered the characteristics of intrasubject analysis by an initial intrasubject realignment of the baseline and follow-up image with subsequent calculation of the mean of both realigned images. Then, a diffeomorphic anatomic registration through exponentiated lie algebra (DARTEL) normalization using an implemented DARTEL template was estimated for the segmented mean image. These normalization parameters were applied to the segmentations of the images of both time points. These segmented and normalized images were modulated (i.e., each voxel value was multiplied with the Jacobian determinant derived from the normalization), which allows assessing the actual volume of grey matter.42 We checked the resulting normalized and modulated segments for both time points for preprocessing errors and artifacts41 and smoothed them with a 10 mm Gaussian kernel. Analyses were made with the normalized and modulated grey matter volume segments. Segmentation failed in 1 participant in the DBT group. The results were visualized using the BrainNet Viewer.43
Statistical analysis
We analyzed group differences in number of psychotropic medications/axis I comorbidities using independent t tests and in professional occupation using χ2 tests. We performed 2 × 2 repeated-measurement analyses of variance with the between-subject factor group (DBT, TAU) and the within-subject factor time (t1, t2) to assess differences between the groups in ZAN-BPD, BSL and DERS measurements. Within-group differences between t1 and t2 for the ZAN-BPD, BSL and DERS measurements were analyzed using dependent t tests. We conducted our analyses using SPSS software version 22.0.0 (IBM).
Total intracranial volume (TIV) and number of psychotropic medications were used as nuisance variables for all whole brain analyses. Cross-sectional imaging data were analyzed using a 2-sample t test. In accordance with the method of Gaser and Kurth,41 longitudinal imaging data were analyzed using a flexible factorial design with group (DBT, TAU) as a between-subjects factor, time (t1, t2) as a within-subjects factor and the subject constants. Contrast weights were made according to the method of Gläscher and Gitelman.44 For statistical inference, we used the threshold-free cluster enhancement toolbox, which follows a nonparametric, permutation-based (n = 5000) approach without the need to define an arbitrary cluster-forming threshold.45 Post hoc analyses were conducted by extracting the grey matter volume of the significant clusters via the MarsBaR toolbox.46 We tested the direction of grey matter volume change between t1 and t2 in the DBT group with dependent t tests. Effect sizes of group differences were determined using independent t tests. Location of the peak voxels were determined according to the automated anatomic labelling (AAL) atlas.47 To differentiate between the subregions of the ACC, we performed additional region of interest (ROI) analyses of the rostral and dorsal ACC, as described in the study by Bishop and colleagues.48 We used spheres with an 8 mm radius and the centre Montreal Neurological Institute (MNI) coordinates x, y, z = −2, 44, 20 for the rostral and x, y, z = 4, 14, 36 for the dorsal ACC.
To derive topographically more distinct clusters, we conducted additional analyses. Specifically, we determined the portion of grey matter volume in anatomic ROIs within the significant clusters from the group × time interaction. To this end, we first created anatomic masks (AAL) according to the location of the peak voxel of every cluster from the group × time interaction. Second, we multiplied the statistical image from the group × time interaction thresholded at p < 0.05, family-wise error (FWE)–corrected, with this mask. Finally, we extracted the grey matter volume from this anatomic ROI within the significant clusters and performed follow-up tests as described previously. Associations between the changes in grey matter volume in these anatomic ROIs were tested using bivariate correlations.
To investigate the association between treatment response and changes in brain structure we calculated bivariate correlations between the percentage change of grey matter volume in the anatomic ROI within the significant clusters and treatment response, as measured using the Reliable Change Index (RCI) of the ZAN-BPD.49
We considered a threshold of p < 0.05 corrected for multiple comparisons (FWE) to be significant for all whole brain analyses.
Results
Participant demographic and psychometric data
We included a total of 48 patients with BPD in the present study: 31 patients in the DBT group and 17 patients in the TAU group. Of those in the TAU group, 35.3% received different forms of outpatient psychotherapy, 5.9% received residential crisis intervention, and 58.8% did not report any psychotherapeutic treatment during study participation. No patient in the TAU group was waiting for DBT. In total, 38.7% of patients in the DBT group and 64.7% in the TAU group were unmedicated. The demographic and clinical characteristics of the sample are summarized in Table 1.
Demographic and clinical characteristics of patients with borderline personality disorder (BPD) who received dialectical behavioural therapy (DBT) and patients with BPD who received treatment as usual (TAU)
The DBT and TAU groups did not differ in terms of age and professional occupation (all p > 0.1; Table 1). The DBT group did, however, receive a larger amount of psychotropic medications than the TAU group (1.16 ± 1.27 v. 0.42 ± 0.62, p = 0.027). Number of psychotropic medications was therefore used as nuisance variable in all whole brain analyses. Symptomatology (BSL, ZAN-BPD) and emotion dysregulation (DERS) did not differ between the 2 patient groups at t1 (all p > 0.1; Table 2). Over time, all measures showed larger decreases in the DBT group than in the TAU group (differences in Cohen d between the DBT and TAU groups were 0.49 for BSL, 0.70 for ZAN-BPD and 1.03 for DERS), which led to a significant group × time interaction for emotion dysregulation (F1,46 = 12.16, p = 0.001) and trendwise significant group × time interactions for the measures of BPD symptomatology (BSL: F1,46 = 3.19, p = 0.08; ZAN-BPD: F1,46 = 3.26, p = 0.08).
Psychometric data of patients with borderline personality disorder (BPD) who received dialectical behavioural therapy (DBT) and patients with BPD who received treatment as usual (TAU)
Imaging data
Cross-sectional analysis
As expected, the DBT and TAU groups did not differ in terms of grey matter volume at t1 (p ≥ 0.38, FWE-corrected).
Longitudinal analysis
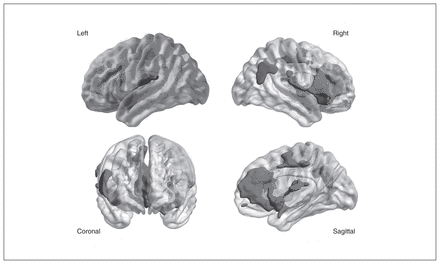
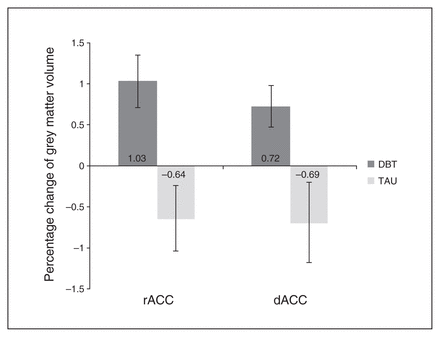
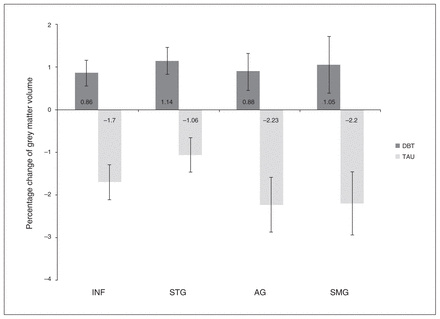
The group × time interaction comparing the DBT and TAU groups over the 12-week period of measurement revealed 5 significant clusters (all p < 0.05, FWE-corrected; Fig. 1 and Table 3). The largest cluster was located in the right ACC (peak voxel x, y, z = 1.5, 39, 7.5). To differentiate between ACC subregions, we conducted an ROI analysis of the rostral and dorsal ACC. Post hoc tests showed an increase in grey matter volume in both subregions in the DBT group (t30 = 3.28, p = 0.018, Bonferroni-corrected for the rostral ACC; t30 = 2.93, p = 0.036, Bonferroni-corrected for the dorsal ACC) as well as a 1.68 percentage change of grey matter volume in the rostral ACC and a 1.42 percentage change in the dorsal ACC compared with the TAU group (d = 0.97 for rostral ACC and d = 0.82 for dorsal ACC; Fig. 2.). Furthermore, the DBT group showed an increase in grey matter volume (t30 = 2.84, p = 0.047, Bonferroni-corrected for the inferior frontal gyrus; t30 = 3.66, p = 0.006, Bonferroni-corrected for the superior temporal gyrus) as well as a 2.57 percentage change of grey matter volume in clusters comprising the right orbital part of the inferior frontal gyrus and a 2.20 percentage change in the left superior temporal gyrus (d = 1.52 for the inferior frontal gyrus and d = 1.30 for the superior temporal gyrus; Fig. 3). The 2 remaining clusters showed no statistically significant direction of change between t1 and t2 for the DBT group (t30 = 1.92, p = 0.38, Bonferroni-corrected for the angular gyrus; t30 = 1.46, p = 0.93, Bonferroni-corrected for the supramarginal gyrus) as well as a 3.11 percentage change in grey matter volume for the angular gyrus and a 3.23 percentage change for the supramarginal gyrus between the DBT and TAU groups (d = 1.23 for the angular gyrus and d = 0.95 for the supramarginal gyrus; Fig. 3).
Significant clusters (p < 0.05, family-wise error [FWE]–corrected) from the group × time interaction comparing patients with borderline personality disorder (BPD) who received residential dialectical behavioural therapy and patients with BPD who received treatment as usual. Average structural brain in left, right, coronal and sagittal view with shaded p values superimposed and thresholded at p < 0.05, FWE-corrected.
Percentage change of grey matter volume in the rostral (rACC) and dorsal (dACC) anterior cingulate cortex for patients with borderline personality disorder (BPD) who received residential dialectical behavioural therapy (DBT) and patients with BPD who received treatment as usual (TAU). Error bars represent the standard error of the mean. The DBT group showed a 1.68 (rACC) and 1.42 (dACC) percentage change in grey matter volume compared with the TAU group.
Percentage change of grey matter volume in clusters comprising the orbital part of the right inferior frontal gyrus (INF), left superior temporal gyrus (STG), right angular gyrus (AG) and right supramarginal gyrus (SMG) for patients with borderline personality disorder (BPD) who received residential dialectical behavioural therapy (DBT) and patients with BPD who received treatment as usual (TAU). Error bars represent the standard error of the mean. The DBT group showed a 2.57 (INF), 2.20 (STG), 3.11 (AG) and 3.23 (SMG) percentage change in grey matter volume compared with the TAU group.
Significant clusters (p < 0.05, family-wise error–corrected) of the group × time interaction comparing patients with borderline personality disorder (BPD) who received dialectical behavioural therapy (DBT) and patients with BPD who received treatment as usual (TAU)
Additional analyses
Additional analyses were conducted to derive topographically more distinct clusters. Specifically, we determined the portion of grey matter volume in anatomic ROIs (ACC, inferior frontal gyrus, superior temporal gyrus, angular gyrus, supramarginal gyrus) within the significant clusters from the group × time interaction. Results were identical in magnitude and significance as for the whole clusters (Appendix 1, Table S1, available at jpn.ca/170132-a1). Furthermore, the grey matter volume changes in these ROIs were all associated with each other (all r values between 0.37 and 0.74, all p < 0.05; Appendix 1, Table S2).
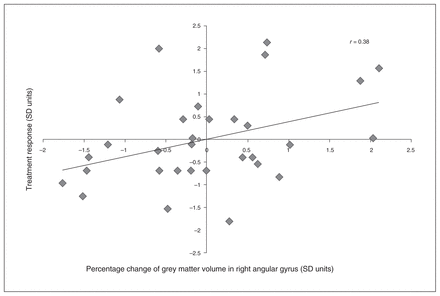
The explorative investigation of associations between treatment response and change of grey matter volume over the period of measurement revealed that treatment response was associated with increased grey matter volume in the right angular gyrus (r = 0.38, p = 0.032; Fig. 4), but in none of the other regions (all p > 0.05).
Association (r = 0.38, p = 0.032) between an increase of grey matter volume (percentage change) in the angular gyrus and treatment response (reliable change index of the Zanarini rating scale) in patients with borderline personality disorder who received residential dialectical behavioural therapy. SD = standard deviation.
Given the high comorbidity of eating disorders in our sample (DBT 35.50%, TAU 29.40%; Table 1) and that weight change is associated with alterations in grey matter volume,50 we tested whether the 2 groups differed in weight change over the period of measurement. Results showed that this was not the case (mean change −0.13 kg in the DBT group v. 0.94 kg in the TAU group, p = 0.19; Appendix 1, Table S3), which makes us confident that the findings were not confounded by differences in weight change between the 2 groups.
Discussion
To the best of our knowledge this is the first study to investigate the effect of psychotherapy on the brain structure of patients with BPD. We hypothesized that DBT would alter brain regions that are explicitly involved in cognitive regulation of emotional information and mentalizing. The results support these notions by showing that patients with BPD receiving 12 weeks of residential DBT experienced an increase in grey matter volume in the ACC and the orbital part of the inferior frontal gyrus. Furthermore, DBT increased the grey matter volume in the superior temporal gyrus and altered the grey matter volume in the angular and supramarginal gyrus. Explorative analyses indicated that therapy response was associated with an increase of grey matter volume in the angular gyrus.
The dorsal ACC is believed to play an important role in attention and executive function, while the rostral ACC is implicated in the assessment and regulation of emotional information, especially in situations that require cognitive control in the presence of high emotional input.51,52 Our results suggest that DBT leads to an increase in grey matter volume in both the dorsal and the rostral ACC, which may shed light on the mechanisms of change associated with psychotherapy. Patients participating in DBT receive and process a vast amount of cognitive information; for example, the emotion regulation protocol exercises the patients’ attentive and executive functions to help them make adequate response selections in service of adaptive emotion regulation.53 In fact, emotion regulation and distress tolerance skills train the patients to actively regulate their emotional responses via cognitive processing. Thus, our reported results of grey matter volume increase in both subdivisions of the ACC in the DBT group are in accordance with the therapeutic targets of DBT and may be regarded as correlates of the effects of therapeutic techniques aiming at keeping or regaining cognitive control over maladaptive emotions.
In addition, the alteration of ACC volume through DBT is in line with the findings of 3 longitudinal functional neuroimaging studies that are part of the current clinical trial. All 3 studies reported alterations in ACC regions: DBT led to altered connectivity of the dorsal ACC and the amygdala during pain-mediated emotion regulation;30 to differential activity in a section of the rostral ACC (the perigenual ACC) during distraction31 as well as an activity decrease; and to increased connectivity with prefrontal, temporal and parietal regions of the ACC.32 The concordance of functional and structural data was also seen for the right supramarginal gyrus, which showed an activity decrease during distraction in the study by Winter and colleagues31 and an alteration of grey matter volume in the present study. Hence, in accordance with classical Hebbian theory54 one could speculate that DBT induces a functional change that is then followed by structural adaptation.
The increase of grey matter volume in the orbital part of the inferior frontal gyrus may also be traced back to the emphasis of DBT on emotion regulation. The inferior frontal gyrus has a core role in suppressing response tendencies.55 A central aspect of the emotion regulation skills is to inhibit emotional responses. For instance, if the behavioural urge accompanying the present emotion is disadvantageous, the patient is taught to counteract it. Similarly, distress tolerance skills help the patient not to follow the emotional drive by using strategies such as distraction. Thus, the increase of grey matter volume in the inferior frontal gyrus may reflect effects of training patients to use different strategies of behavioural inhibition.
The angular, superior temporal and supramarginal gyri are not only anatomically, but also functionally adjacent. All have been implicated in mentalizing.56,57 Mentalizing, or theory of mind, refers to the capacity to make sense of ourselves or others.20 Participating in inpatient DBT may challenge both explicit goal-oriented tasks of mentalizing and less active mentalizing during idle moments. The former is explicitly trained in the interpersonal effectiveness skills module. The latter is likely to occur frequently during the stay in an inpatient unit, where the whole framework is encouraging and animating the patient to self and other referential reasoning. The change in grey matter volume in these brain areas may therefore be regarded as a correlate of enhanced mentalizing — either active or in passing — during DBT.
The revealed associations between the changes in grey matter volume in all brain regions suggest an orchestrated change to brain structure through DBT. Theoretical20 as well as empirical18 work show a tight interplay between affect dysregulation and mentalizing. More specifically, dysfunction in mentalizing has been shown to exert its effect on BPD symptomatology through affective dysregulation.18 Hence, the association between changes in grey matter volume in brain regions primarily implicated in emotion regulation (ACC) and those brain regions with a known role in mentalizing (angular gyrus, superior marginal gyrus, superior temporal gyrus) may reflect a particular sensitivity to psychotherapeutic interventions or even mutual interdependence between these regions.
Results further revealed that treatment response in the DBT group was associated with change in grey matter volume in the angular gyrus. Current theories on the angular gyrus emphasize its cross-integrative function.58 That is, the angular gyrus may be regarded as a cross-modal integrative hub58 that combines and integrates multisensory information to transform perception into recognition, word forms into meaning, scenes and events into experiences, and spatial locations into targets for exploration. Inherent to all these processes is the preparedness of the individual to acquire new knowledge about oneself and the outer world. Consistent with this, the ability to trust others as a source of new knowledge has been regarded as the fundamental characteristic of BPD and termed “epistemic hypervigilance.”59 The role of the angular gyrus in treatment response may thus reside in its integrative function, which in turn allows individuals to embrace information presented within psychotherapy for their own benefits.
Notably, the grey matter volume change in the other brain regions (ACC, inferior frontal gyrus, supramarginal gyrus, superior temporal gyrus) did not correlate significantly with treatment effect. Given the frequently reported delayed emergence of psychotherapy effects, particularly for complex mental disorders such as BPD,60–62 this finding might reflect that changes in these regions impact upon the treatment results in the long run. Thus, it would be of great interest to conduct long-term follow-up investigations to capture potential delayed associations between brain structure and treatment effects.
Interestingly, the TAU group experienced considerable grey matter volume loss in the investigated brain regions. The magnitude of this loss was comparable to that reported in studies of other mental disorders.5,63,64 It is appreciated that severe mental disorders, such as schizophrenia65 or bipolar disorder,66 are accompanied by grey matter volume loss. This raises the question of whether the grey matter volume loss in the TAU group may — at least partly — correspond to the natural course of BPD and that psychotherapy, such as DBT, might exert a neuroprotective effect. However, although the DBT and TAU groups were matched for age and professional occupation and the effect of psychotropic medications was controlled for, we cannot exclude the influence of other confounding factors, such as menstrual cycle.67
There are several reasons why it was unlikely to find alterations of the hippocampus and the amygdala after 12 weeks of DBT. First, previous studies using a comparable psychotherapy and duration of treatment did not report a change in these brain regions.8 Second, DBT has a clear focus on the explicit cognitive regulation of emotions, whereas the amygdala — and presumably the hippocampus — are typically implicated in automatic (implicit) emotional responses.68 Finally, amygdalar and hippocampal regions may instead be altered in the long term. That is, constant and durable input from regulatory regions may be necessary to structurally alter these highly conserved evolutionary regions.
Using neurobiological methods to help us better understand or improve psychotherapeutic techniques has received considerable interest.69–71 Findings such as those reported here contribute to these endeavours, as they help us to better understand the mechanisms of change associated with psychotherapy. Additionally, and in line with current conceptualizations of psychopathology that classify mental disorders on the basis of behavioural dimensions and neurobiological measures,72 they may aid in the development of neurobiologically informed therapeutic interventions.
Limitations
This study has some limitations. First, the TAU group was small (n = 17), which we tried to address by using a rather conservative significance threshold of p < 0.05, FWE-corrected. However, replication in independent samples is required to confirm the results. Second, patients were allowed to be medicated. Importantly, medication was kept constant during the study period of 12 weeks plus 2 weeks before the first measurement, but the DBT group received a larger number of psychotropic medications than the TAU group. Yet, influence on our results may have been limited as we included the number of psychotropic medications as a control variable in our analysis. Third, we measured only women. Particularly, in view of the known sex differences on brain structure, 73 this limits the transferability of our findings to men. Fourth, approximately one-third of our patient control group received different forms of outpatient psychotherapy, which reduces the capacity to rigorously control for pure time effects. Fifth, there was no randomized treatment assignment, which limits the ability to draw causal conclusions. Sixth, the biological nature of the reported changes in brain structure remains elusive.1 Future studies combining different imaging modalities and animal studies are needed to better understand the exact processes underlying the signal changes found with MRI morphometry. Seventh, we used an FWE-corrected significance level in all imaging analyses. However, we did not apply an additional correction (e.g., Bonferroni) during the explorative investigation of the association between treatment response and change in grey matter volume. Finally, our study design doesn’t support any conclusion regarding the DBT-specificity of the results. Using a control group with a comparable intense treatment condition could address this question.
Conclusion
Our study provides initial insight into the effects of psychotherapy on the brain structure of patients with BPD. More specifically, DBT increased the grey matter volume of brain regions that are critically implicated in emotion regulation and higher-order brain functions, such as mentalizing. These findings enhance our understanding of the mechanisms of change associated with psychotherapy and may foster the development of neurobiologically informed therapeutic interventions.
Acknowledgements
This study was supported by grants from the German Research Foundation (DFG) awarded to S. Herpertz (HE 2660/7-2) and C. Schmahl (SCHM 1526/8-2).
Footnotes
↵* These authors contributed equally to this work.
Competing interests: None declared.
Contributors: S. Herpertz and C. Schmahl designed the study. R. Schmitt, D. Winter and I. Niedfeld acquired the data, which F. Mancke, I. Niedtfeld, S. Herpertz and C. Schmahl analyzed. F. Mancke wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received July 13, 2017.
- Revision received September 19, 2017.
- Accepted September 19, 2017.