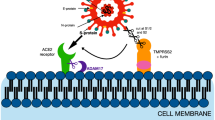
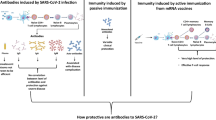
Abstract
Although the coronavirus disease 2019 (COVID-19) epidemic is still ongoing, vaccination rates are rising slowly and related treatments and drugs are being developed. At the same time, there is increasing evidence of preexisting immunity against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in humans, mainly consisting of preexisting antibodies and immune cells (including T cells and B cells). The presence of these antibodies is mainly due to the seasonal prevalence of four common coronavirus types, especially OC43 and HKU1. The accumulated relevant evidence has suggested that the target of antibodies is mainly the S2 subunit of S protein, followed by evolutionary conservative regions such as the nucleocapsid (N) protein. Additionally, preexisting memory T and B cells are also present in the population. Preexisting antibodies can help the body protect against SARS-CoV-2 infection, reduce the severity of COVID-19, and rapidly increase the immune response post-infection. These multiple effects can directly affect disease progression and even the likelihood of death in certain individuals. Besides the positive effects, preexisting immunity may also have negative consequences, such as antibody-dependent enhancement (ADE) and original antigenic sin (OAS), the prevalence of which needs to be further established. In the future, more research should be focused on evaluating the role of preexisting immunity in COVID-19 outcomes, adopting appropriate policies and strategies for fighting the pandemic, and vaccine development that considers preexisting immunity.
摘要
新型冠状病毒肺炎(COVID-19)疫情仍在持续,疫苗接种率正在缓慢上升,相关治疗方法和药物也在研发中。越来越多的证据表明,人类群体中已存在针对严重急性呼吸综合征冠状病毒2型(SARS-CoV-2)的免疫力,包括抗体和免疫细胞(T细胞和B细胞)。这些抗体的存在主要是由于四种常见冠状病毒类型(尤其OC43和HKU1)的季节性流行所致。预存抗体的靶点主要是S蛋白的S2亚基,其次是核衣壳(N)等蛋白进化保守区。此外,人群中也存在预存的记忆T细胞和B细胞。预存抗体可以帮助身体抵御SARS-CoV-2感染,降低COVID-19的严重程度并迅速增加感染后的免疫反应。这些多重影响可以直接影响某些个体的疾病进展,甚至死亡的风险性。除了积极作用外,预存免疫也可能产生消极后果,例如抗体依赖性增强(ADE)和原始抗原原罪(OAS),其流行程度需要进一步确定。未来,更多的研究应侧重于评估已有免疫力在COVID-19中的作用,采取适当的政策和策略来抗击新冠大流行,同时也要考虑已有免疫力的疫苗开发。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anderson EM, Goodwin EC, Verma A, et al., 2021. Seasonal human coronavirus antibodies are boosted upon SARS-CoV-2 infection but not associated with protection. Cell, 184(7):1858–1864.e10. https://doi.org/10.1016/j.cell.2021.02.010
Bonifacius A, Tischer-Zimmermann S, Dragon AC, et al., 2021. COVID-19 immune signatures reveal stable antiviral T cell function despite declining humoral responses. Immunity, 54(2):340–354.e6. https://doi.org/10.1016/j.immuni.2021.01.008
Braun J, Loyal L, Frentsch M, et al., 2020. SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19. Nature, 587(7833):270–274. https://doi.org/10.1038/s41586-020-2598-9
Cao WC, Liu W, Zhang PH, et al., 2007. Disappearance of antibodies to SARS-associated coronavirus after recovery. N Engl J Med, 357(11):1162–1163. https://doi.org/10.1056/NEJMc070348
Dai LP, Gao GF, 2021. Viral targets for vaccines against COVID-19. Nat Rev Immunol, 21(2):73–82. https://doi.org/10.1038/s41577-020-00480-0
de Assis RR, Jain A, Nakajima R, et al., 2021. Analysis of SARS-CoV-2 antibodies in COVID-19 convalescent blood using a coronavirus antigen microarray. Nat Commun, 12:6. https://doi.org/10.1038/s41467-020-20095-2
de Vries RD, 2020. SARS-CoV-2-specific T-cells in unexposed humans: presence of cross-reactive memory cells does not equal protective immunity. Signal Transduct Tar Ther, 5:224. https://doi.org/10.1038/s41392-020-00338-w
Dong ES, Du HR, Gardner L, 2020. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis, 20(5):533–534. https://doi.org/10.1016/S1473-3099(20)30120-1
Doshi P, 2020. Covid-19: do many people have pre-existing immunity? BMJ, 370:m3563. https://doi.org/10.1136/bmj.m3563
Feng BH, Zhang D, Wang Q, et al., 2021. Effects of angiotensin II receptor blocker usage on viral load, antibody dynamics, and transcriptional characteristics among COVID-19 patients with hypertension. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 22(4):330–340. https://doi.org/10.1631/jzus.B2000730
Fierz W, Walz B, 2020. Antibody dependent enhancement due to original antigenic sin and the development of SARS. Front Immunol, 11:1120. https://doi.org/10.3389/fimmu.2020.01120
Greenbaum JA, Kotturi MF, Kim Y, et al., 2009. Pre-existing immunity against swine-origin H1N1 influenza viruses in the general human population. Proc Natl Acad Sci USA, 106(48):20365–20370. https://doi.org/10.1073/pnas.0911580106
Grifoni A, Weiskopf D, Ramirez SI, et al., 2020. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell, 181(7):1489–1501.e15. https://doi.org/10.1016/j.cell.2020.05.015
Huang CL, Wang YM, Li XW, et al., 2020. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet, 395(10223):497–506. https://doi.org/10.1016/S0140-6736(20)30183-5
Huang Y, Yang C, Xu XF, et al., 2020. Structural and functional properties of SARS-CoV-2 spike protein: potential antivirus drug development for COVID-19. Acta Pharmacol Sin, 41(9):1141–1149. https://doi.org/10.1038/s41401-020-0485-4
Jackson CB, Farzan M, Chen B, et al., 2022. Mechanisms of SARS-CoV-2 entry into cells. Nat Rev Mol Cell Biol, 23(1):3–20. https://doi.org/10.1038/s41580-021-00418-x
Jia LQ, Weng SF, Wu J, et al., 2022. Pre-existing antibodies targeting a linear epitope on SARS-CoV-2 S2 cross-reacted with commensal gut bacteria and shaped vaccine induced immunity. medRxiv, prepint. https://doi.org/10.1101/2021.07.13.21260404
Kaplonek P, Wang CQ, Bartsch Y, et al., 2021. Early cross-coronavirus reactive signatures of protective humoral immunity against COVID-19. bioRxiv, prepint. https://doi.org/10.1101/2021.05.11.443609
King AMQ, Adams MJ, Carstens EB, et al., 2011. Virus Taxonomy. Ninth Report of the International Committee on Taxonomy of Viruses. Elsevier, St. Louis, p.770–783.
Kissler SM, Tedijanto C, Goldstein E, et al., 2020. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science, 368(6493):860–868. https://doi.org/10.1126/science.abb5793
Knoops K, Kikkert M, van den Worm SHE, et al., 2008. SARS-coronavirus replication is supported by a reticulovesicular network of modified endoplasmic reticulum. PLoS Biol, 6(9):e226. https://doi.org/10.1371/journal.pbio.0060226
Kupferschmidt K, 2021. New coronavirus variants could cause more reinfections, require updated vaccines. Science COVID-19 Report. https://www.science.org/content/article/new-coronavirus-variants-could-cause-more-reinfections-require-updated-vaccines [accessed on Jan. 15, 2021].
Lapp SA, Edara VV, Lu A, et al., 2021. Original antigenic sin responses to Betacoronavirus spike proteins are observed in a mouse model, but are not apparent in children following SARS-CoV-2 infection. PLoS ONE, 16(8):e0256482. https://doi.org/10.1371/journal.pone.0256482
Lauring AS, Malani PN, 2021. Variants of SARS-CoV-2. JAMA, 326(9):880–880. https://doi.org/10.1001/jama.2021.14181
le Bert N, Tan AT, Kunasegaran K, et al., 2020. SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls. Nature, 584(7821):457–462. https://doi.org/10.1038/s41586-020-2550-z
Lee WS, Wheatley AK, Kent SJ, et al., 2020. Antibody-dependent enhancement and SARS-CoV-2 vaccines and therapies. Nat Microbiol, 5(10):1185–1191. https://doi.org/10.1038/s41564-020-00789-5
Lefkowitz EJ, Dempsey DM, Hendrickson RC, et al., 2018. Virus taxonomy: the database of the International Committee on Taxonomy of Viruses (ICTV). Nucl Acids Res, 46(D1):D708–D717. https://doi.org/10.1093/nar/gkx932
Majdoubi A, Michalski C, O’Connell SE, et al., 2021. A majority of uninfected adults show preexisting antibody reactivity against SARS-CoV-2. JCI Insight, 6(8):e146316. https://doi.org/10.1172/jci.insight.146316
Millet JK, Jaimes JA, Whittaker GR, 2021. Molecular diversity of coronavirus host cell entry receptors. FEMS Microbiol Rev, 45(3):fuaa057. https://doi.org/10.1093/femsre/fuaa057
Mveang Nzoghe A, Essone PN, Leboueny M, et al., 2021. Evidence and implications of pre-existing humoral cross-reactive immunity to SARS-CoV-2. Immun, Inflamm Dis, 9(1):128–133. https://doi.org/10.1002/iid3.367
Netea MG, Joosten LAB, Latz E, et al., 2016. Trained immunity: a program of innate immune memory in health and disease. Science, 352(6284):aaf1098. https://doi.org/10.1126/science.aaf1098
Ng KW, Faulkner N, Cornish GH, et al., 2020. Preexisting and de novo humoral immunity to SARS-CoV-2 in humans. Science, 370(6522):1339–1343. https://doi.org/10.1126/science.abe1107
Ng OW, Chia A, Tan AT, et al., 2016. Memory T cell responses targeting the SARS coronavirus persist up to 11 years post-infection. Vaccine, 34(17):2008–2014. https://doi.org/10.1016/j.vaccine.2016.02.063
Nguyen-Contant P, Embong AK, Kanagaiah P, et al., 2020. S protein-reactive IgG and memory B cell production after human SARS-CoV-2 infection includes broad reactivity to the S2 subunit. mBio, 11(5):e01991–20. https://doi.org/10.1128/mBio.01991-20
Ortega N, Ribes M, Vidal M, et al., 2021. Seven-month kinetics of SARS-CoV-2 antibodies and role of pre-existing antibodies to human coronaviruses. Nat Commun, 12: 4740. https://doi.org/10.1038/s41467-021-24979-9
Schulien I, Kemming J, Oberhardt V, et al., 2021. Characterization of pre-existing and induced SARS-CoV-2-specific CD8+ T cells. Nat Med, 27(1):78–85. https://doi.org/10.1038/s41591-020-01143-2
Sette A, Crotty S, 2020. Pre-existing immunity to SARS-CoV-2: the knowns and unknowns. Nat Rev Immunol, 20(8): 457–458. https://doi.org/10.1038/s41577-020-0389-z
Shi Y, Wang G, Cai XP, et al., 2020. An overview of COVID-19. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(5): 343–360. https://doi.org/10.1631/jzus.B2000083
Song G, He WT, Callaghan S, et al., 2021. Cross-reactive serum and memory B-cell responses to spike protein in SARS-CoV-2 and endemic coronavirus infection. Nat Commun, 12:2938. https://doi.org/10.1038/s41467-021-23074-3
Sridhar S, Begom S, Bermingham A, et al., 2013. Cellular immune correlates of protection against symptomatic pandemic influenza. Nat Med, 19(10):1305–1312. https://doi.org/10.1038/nm.3350
Sui ZW, Dai XH, Lu QB, et al., 2021. Viral dynamics and antibody responses in people with asymptomatic SARS-CoV-2 infection. Signal Transduct Tar Ther, 6:181. https://doi.org/10.1038/s41392-021-00596-2
Vashishtha VM 2021. Is ‘original antigenic sin’ complicating Indian vaccination drive against Covid-19? Hum Vacc Immunother, 17(10):3314–3315. https://doi.org/10.1080/21645515.2021.1945904
Weiskopf D, Schmitz KS, Raadsen MP, et al., 2020. Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome. Sci Immunol, 5(48):eabd2071. https://doi.org/10.1126/sciimmunol.abd2071
Wilkinson TM, Li CKF, Chui CSC, et al., 2012. Preexisting influenza-specific CD4+ T cells correlate with disease protection against influenza challenge in humans. Nat Med, 18(2):274–280. https://doi.org/10.1038/nm.2612
Wu F, Zhao S, Yu B, et al., 2020. A new coronavirus associated with human respiratory disease in China. Nature, 579(7798):265–269. https://doi.org/10.1038/s41586-020-2008-3
Zhou P, Yang XL, Wang XG, et al., 2020. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 579(7798):270–273. https://doi.org/10.1038/s41586-020-2012-7
Acknowledgments
This research was supported by the National Science and Technology Major Project of China (No. 2018ZX10302206-003), the Zhejiang Provincial Natural Science Foundation of China (No. LY16H160057), the Health Commission of Zhejiang Province (No. 2016KYA162), and the Zhejiang Provincial Science and Technology Project of Traditional Chinese Medicine of China (No. 2015ZA026).
Author information
Authors and Affiliations
Contributions
Gang WANG, Ze XIANG, Wei WANG, and Zhi CHEN were responsible for data collection and article writing. Zhi CHEN was responsible for conceptualization and manuscript revision. All authors have read and approved the final manuscript.
Corresponding author
Additional information
Compliance with ethics guidelines
Gang WANG, Ze XIANG, Wei WANG, and Zhi CHEN declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Wang, G., Xiang, Z., Wang, W. et al. Seasonal coronaviruses and SARS-CoV-2: effects of preexisting immunity during the COVID-19 pandemic. J. Zhejiang Univ. Sci. B 23, 451–460 (2022). https://doi.org/10.1631/jzus.B2200049
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200049
Key words
- Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
- Coronavirus disease 2019 (COVID-19)
- Preexisting immunity
- Seasonal coronaviruses
- Vaccine