Summary
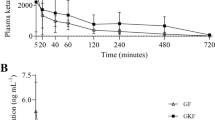
The effects of anaesthesia and surgery on the pharmacokinetics of ketobemidone were studied in 12 patients. Plasma ketobemidone concentrations were assayed with a mass-fragmentographic method. The peroperative Vdarea was 5.9±2.6L/kg and the terminal half-life was 3.9 ± 1.7h. In the postoperative period Vdarea decreased to 3.7 ±0.4L/kg and the terminal half-life to 2.1 ±0.4h. Plasma clearance (Clp) did not change significantly, peroperative Clp being 18±4.3ml/min/kg and postoperative Clp being 22 ±7.5ml/min/kg. The pharmacokinetics of ketobemidone were not influenced by the addition of a spasmolytic agent in the commercial combination ketobemidone preparation ‘Ketogin’.
Postoperative pain was relieved in 15 patients by patient-controlled intravenous administration of ketobemidone by means of a programmable drug injector. The mean ketobemidone consumption was 2.3 ± 0.8mg/h, which produced a mean plasma concentration of 28 ± 11ng/ml. Pseudosteady-state plasma concentrations of ketobemidone were established with a mean minimum effective concentration (MEC) of 25 ± 11ng/ml. Ketobemidone ‘plain’ and ‘Ketogin’ did not differ significantly in these respects. Analgesia was considered by all patients to be satisfactory.
Similar content being viewed by others
References
Anderson, P.; Arnér, S.; Bondesson, U.; Boréus, L-O. and Hartvig, P.: Clinical pharmacokinetics of ketobemidone. Its bioavailability after rectal administration. European Journal of Clinical Pharmacology 19: 217–223 (1981).
Bondesson, U. and Hartvig, P.: Mass fragmentographic method for the determination of ketobemidone in plasma. Journal of Chromatography 179: 207–212 (1979).
Bondesson, U.; Arnér, S.; Anderson, P.; Boréus, L-O. and Hartvig, P.: Clinical pharmacokinetics and oral bioavailability of ketobemidone. European Journal of Clinical Pharmacology 17: 45–50 (1980).
Boxenbaum, H.-G.; Riegelman, S. and Elashoff, R.M.: Statistical estimations in pharmacokinetics. Journal of Pharmacokinetics and Biopharmaceutics 2: 123–148 (1974).
Dahlström, B.; Tamsen, A.; Paalzow, L. and Hartvig, P.: Patient-controlled analgesic therapy, part IV: Pharmacokinetics and analgesic plasma concentrations of morphine. Clinical Pharmacokinetics 7: 266–279 (1982).
Eddy, N.B.; Halbach, H. and Braenden, O.J.: Synthetic substances with morphine-like effect. 17. Pethidine derivatives. (C) Ketobemidone (l-methyl-4(m-hydroxyphenyl)-4-piperidyl ethyl ketone). Bulletin of the World Health Organization 17: 815–824 (1957).
Epstein, R.M.; Wheeler, H.O., Frumin, M.J.; Habif, D.V.; Papper, E.M. and Bradley, S.E.: The effect of hypercapnia on estimated hepatic blood flow, circulating splanchnic blood volume, and hepatic sulphobromophthalein clearance during general anaesthesia in man. Journal of Clinical Investigation 40: 592–598 (1961).
Kehlet, H. and Binder, C.: Alterations in distribution volume and biological half-life of cortisol during major surgery. Journal of Clinical Endocrinology and Metabolism 36: 330–334 (1973).
Mather, L.E. and Meffin, P.J.: Clinical pharmacokinetics pethidine. Clinical Pharmacokinetics 3: 352–368 (1978).
Metzler, C.M.; Elfring, G.L. and McEven, A.J.: A user’s manual for NONLIN and associated programs. Research Biostatistics. The Upjohn Company, Kalamazoo, Michigan (1974).
Peltola, P. and Soisalo, P.: The analgesic properties and addiction liability of ketobemidone and morphine. Archives of Internal Medicine 101: 741–746 (1958).
Petersen, P.V.: Studies on a new spasmolytical compound 1,1-diphenyl-3-dimethylaminobutene-a (A29), related to methadone, and to the combined use of this compound and a potent analgesic, ketobemidone (A21). Acta Pharmacologica et Toxicologica 7: 51–64 (1951).
Statistical Analysis System (SAS Institute Cary, North Carolina, 1979).
Scott, C.C.; Robbins, E.B. and Chen, K.K.: Comparison of some new analgesic compounds. Science 104(2712): 587–588 (1946).
Stanski, D.R.; Greenblatt, D. and Lowenstein, E.: Kinetics of intravenous and intramuscular morphine. Clinical Pharmacology and Therapeutics 24: 52–59 (1978).
Stjernström, H.; Jorfeldt, L. and Wiklund, L.: Influence of abdominal surgical trauma and intraoperative infusion of glucose on splanchnic glucose metabolism in man. Clinical Physiology 1: 511–523 (1981).
Swartz, R.D. and Sidell, F.R.: Effects of heat and exercise on the elimination of pralidoxime in man. Clinical Pharmacology and Therapeutics 14: 83–89 (1973).
Swartz, R.D.; Sidell, F.R. and Cucinell, S.A.: Effects of physical stress on the disposition of drugs eliminated by the liver in man. Journal of Pharmacology and Experimental Therapeutics 188: 1–7 (1974).
Tamsen, A.; Hartvig, P.; Fagerlund, C.; Dahlström, B. and Bondesson, U.: Patient-controlled analgesic therapy: Clinical experience. Acta Anaesthesiologica Scandinavica, Supplementum. In press (1982).
Tamsen, A.; Hartvig, P.; Fagerlund, C. and Dahlström, B.: Patient-controlled analgesic therapy, part I: Pharmacokinetics of pethidine in the per- and postoperative periods. Clinical Pharmacokinetics 7: 149–163 (1982a).
Tamsen, A.; Hartvig, P.; Fagerlund, C. and Dahlström, B.: Patient-controlled analgesic therapy, part II: Individual analgesic demand and analgesic plasma concentrations of pethidine in postoperative pain. Clinical Pharmacokinetics 7: 164–175 (1982b).
Wagner, J.G.: Fundamentals of Clinical Pharmacokinetics, p.287 (Drug Intelligence Publications, Hamilton, Illinois 1975).
Wiklund, L.: Postoperative hepatic blood flow and its relation to systemic circulation and blood gases during splanchnic blockade and fentanyl analgesia. Acta Anaesthesiologica Scandinavica 19 (Suppl. 58): 5–19 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tamsen, A., Bondesson, U., Dahlström, B. et al. Patient-controlled Analgesic Therapy, Part III: Pharmacokinetics and Analgesic Plasma Concentrations of Ketobemidone. Clin Pharmacokinet 7, 252–265 (1982). https://doi.org/10.2165/00003088-198207030-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-198207030-00005