Summary
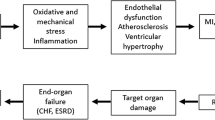
The effects of cardiovascular drugs on endothelium and vascular smooth muscle function are important for the prevention of cardiovascular disease. Changes in endothelial function are an early event in most forms of cardiovascular disease and, later in the disease process, vascular smooth muscle cells are functionally altered and begin to migrate to and proliferate in the intima. Calcium antagonists and angiotensin converting enzyme (ACE) inhibitors are widely used in patients with cardiovascular disease and are thought to have vascular protective effects. ACE, an enzyme located in the endothelial cell membrane, activates angiotensin I and angiotensin II, and deactivates bradykinin. Bradykinin activates endothelial bradykinin (B2) receptors, which results in the formation of nitric oxide and prostacyclin. Hence, ACE inhibitors not only prevent the formation of angiotensin II, but also increase the local levels of bradykinin and in turn nitric oxide and prostacyclin. These compounds are vasodilators and potent inhibitors of platelet function, and therefore may mediate important protective effects of ACE inhibitors. Furthermore, nitric oxide may have antiproliferative effects in vascular smooth muscle cells.
Calcium antagonists do not appear to affect the release of endothelium-derived relaxing factors or any other endothelial product. However, they facilitate endothelium-dependent relaxation and reduce the contracting effects of endothelin-1 at the level of smooth muscle. Indeed, in some blood vessels, e.g. the large coronary arteries and the human forearm circulation, verapamil and nifedipine antagonise endothelin-induced contractions. In addition, calcium antagonists inhibit the effects of platelet-derived growth factor and may have antiproliferative effects in vascular smooth muscle cells. In conditions involving progressive dysfunction of the endothelium, vascular deposition of platelets increases the local levels of platelet-derived growth factor, and the antiproliferative effects of calcium antagonists may thus be particularly important.
Similar content being viewed by others
References
Arai H, Hori S, Aramori I, Ohkubo H, Nakanishi S. Cloning and expression of a cDNA encoding and endothelin receptor. Nature 348: 730–732, 1990
Auch-Schwelk W, Bossaller C, Claus M, Graf K, Gräfe M, et al. Endothelium-dependent relaxations to angiotensin-converting enzyme inhibitors in isolated coronary arteries stimulated with bradykinin. Journal of Cardiovascular Pharmacology 20 (Suppl. 9): S62–S67, 1992
Bossaller C, Auch-Schwelk W, Weber F, Götze S, Gräfe M, et al. Endothelium-dependent relaxations are augmented in rats chronically treated with the angiotensin-converting enzyme inhibitor enalapril. Journal of Cardiovascular Pharmacology 20 (Suppl. 9): 91–95, 1992
Busse R, Lamontagne D. Endothelium-derived bradykinin is responsible for the increase in calcium produced by angiotensin-converting enzyme inhibitors in human endothelial cells. Naunyn Schmiedeberg’s Archives of Pharmacology 344: 126–129, 1991
Bredt DS, Hwang PM, Glatt CE, Lowenstein C, Reed RR, et al. Cloned and expressed nitric oxide synthase structurally resembles cytochrome P-450 reductase Nature 351: 714, 1991
Caldwell PRB, Seegal BC, Hsu KC, Das M, Sooffer RL. Angiotensin converting enzyme: vascular endothelial cell location. Science 191: 1050–1051, 1976
Cauvin C, Tejerina M, Hwang O, Kai-Yamamoto M, van Breemen C. The effects of Ca2+-antagonists on isolated rat and rabbit mesenteric resistance vessels. What determines the sen-sitivyt of agonist-activated vessels to Ca2+-antagonists? Annals of the New York Academy of Sciences S22: 338–350, 1988
Creager MA, Roddy M-A, Coleman SM, Dzau VJ. The effect of ACE inhibition on endothelium-dependent vasodilation in hypertension Journal of Vascular Research 29: 97, 1992
Dohi Y, Criscione L, Pfeiffer K, Lüscher TF. Normalization of endothelial dysfunction of hypertensive mesenteric resistance arteries by chronic therapy with benazapril or nifedipine. Abstract. Journal of the American College of Cardiology 19 (Suppl. 5): 226A, 1992a
Dohi Y, Boulanger CM, Hahn AWA, Bühler FR, Lüscher TF. Endothelin stimulated by angiotensin II augments vascular contractility of hypertensive resistance arteries. Hypertension 19: 131–137, 1992b
Dohi Y, Hahn AWA, Boulanger CM, Lüscher TF. Vascular renin angiotensin system and endothelial function: effect of ACE inhibitors. In MacGregor et al. (Eds) Current advance in ACE-inhibition II, pp. 226–229, Churchill Livingstone, Edinburgh, 1991
Dohi Y, Lüscher TF. Aging differentially affects direct and indirect actions of endothelin-1 in perfused mesenteric arteries of the rat. British Journal of Pharmacology 100: 889–893, 1990
Dzau VJ. Short- and long-term determinants of cardiovascular function and therapy: contributions of circulating and tissue renin-angiotensin systems. Journal of Cardiovascular Pharmacology 14 (Suppl. 4): 1–5, 1989
Erne P, Bolli P, Bürgisser E, Bühler FR. Correlation of platelet calcium with blood pressure: effect of antihypertensive therapy New England Journal of Medicine 310: 1084, 1984
Garg UC, Hassid A. Nitric oxide-generating vasodilators and 8-bromo-cyclic guanosine monophosphate inhibit mitogenesis and proliferation of cultured rat vascular smooth muscle cells. Journal of Clinical Investigation 83: 1774–1777, 1989
Godfraind T, Mennig D, Morel N, Wibo M. Effect of endothelin-1 on calcium channel gating by agonists in vascular smooth muscle. Journal of Cardiovascular Pharmacology 13 (Suppl. 5): 112–117, 1989
Goto K, Kasuya Z, Matsuki N, et al. Endothelin activates the dihydropyridine-sensitive, voltage-dependent calcium channel in vascular smooth muscle. Proceedings of the National Academy of Sciences of the United States of America 86: 3915–3918, 1989
Johns A, Freay AD, Adams DJ, Lategan TW, Ryan US, et al. The role of calcium in the activation of endothelial cells. Journal of Cardiovascular Pharmacology 12 (Suppl. 5): 119–123, 1988
Kiowski W, Lüscher TF, Linder L, Bühler FR. Endothelin-1-induced vasoconstriction in man: reversal by calcium channel blockade but not by nitrovasodilators or endothelium-derived relaxing factor. Circulation 83: 469–475, 1991
Lückhoff A, Pohl U, Mülsch A, Busse R. Differential role of extra- and intracellular calcium in the release of EDRF and prostacyclin from cultured endothelial cells. British Journal of Pharmacology 95: 189–196, 1988
Lüscher TF. Angiotensin, ACE inhibitors and endothelial control of vasomotor tone. Basic Research in Cardiology 88 (Suppl. 1): 15–24, 1993
Lüscher TF, Vanhoutte PM. The endothelium: modulator of cardiovascular function, pp. 1–215, CRC Press, Boca Raton, 1990
Lüscher TF, Boulanger CM, Hahn AHA, Yang Z. Molecular and cellular biology of endothelin (Part I). Journal of Hypertension 11: 7–11, 1993a
Lüscher TF, Boulanger CM, Hahn AHA, Yang Z. Molecular and cellular biology of endothelin (Part II). Journal of Hypertension 11: 121–126, 1993b
Masaki T, Godfraind T, Lüscher TF, Rubanyi GM, Vanhoutte PM, et al. IUPHAR Committee on the classification of endothelin receptors. Pharmacology Review, in press, 1993
Miller VM, Komori K, Burnett JC, Vanhoutte PM. Differential sensitivity to endothelin in canine arteries and veins. American Journal of Physiology 257: 1127–1131, 1989
Rapoport RM, Draznin MB, Murad F. Endothelium-dependent relaxation in rat aorta may be mediated through cyclic GMP-dependent protein phosphorylation. Nature 306: 174–176, 1983
Resink TJ, Grigorian GY, Moldabaeva AK, Danilov SM, Bühler FR. Histamine-induced phosphoinositide metabolism in cultured human umbilical vein endothelial cells. Association with thromboxane and prostacyclin release. Biochemical and Biophysical Research Communications 144: 438–446, 1987
Resink TJ, Scott-Burden T, Bühler FR. Endothelin stimulates phospholipase C in cultured vascular smooth muscle cells. Biochemical and Biophysical Research Communications 157: 1360–1368, 1988
Ross R. The pathogenesis of atherosclerosis an update. New England Journal of Medicine 314: 488–500, 1986
Rubanyi GM, Vanhoutte PM. Calcium and activation of release of endothelium-derived relaxing factor. Annals of the New York Academy of Sciences 522: 226–231, 1988
Sakurai T, Yanagisawa M, Takuwa Y, et al. Cloning of a cDNA encoding a non-isopeptide-selective subtype of the endothelin receptor. Nature 348: 732–735, 1990
Tabuchi Y, Nakamaru M, Rakugi H, Nagano M, Ogihara N. Endothelin enhances adrenergic vasoconstriction in perfused rat mesenteric arteries. Biochemical and Biophysical Research Communications 159: 1304–1308, 1989
Timmermans PBMWM, Benfield P, Chiu AT, Herblin WF, Wong PC, et al. Angiotensin II receptors and functional correlates. American Journal of Hypertension 5 (Suppl.): 221S–235S, 1992
Vane J. Endothelins come home to roost. Nature 348: 673–675, 1990
Vanhoutte PM. The expert committee of the World Health Organization on classification of calcium antagonists: the viewpoint of the raporteur. American Journal of Cardiology 59 (Suppl. 2): 3A–8A, 1987a
Vanhoutte PM. The end of the quest? Nature 327: 459–460, 1987b
Wallnöfer A, Weir S, Rüegg U, Cauvin C. The mechanism of action of endothelin-1 as compared with other agonists in vascular smooth muscle. Journal of Cardiovascular Pharmacology 13 (Suppl. 5): 23–31, 1989
Wiemer G, Scholkens BA, Becker RH, Busse R. Ramiprilat enhances endothelial autocoid formation by inhibiting breakdown of endothelial derived bradykinin. Hypertension 18: 558–563, 1991
Williams LT. Signal transduction by the platelet-derived growth factor receptor. Science 243: 1564–1570, 1989
Yanagisawa M, Kurihara H, Kimura S, et al. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332: 411–415, 1988
Yang Z, Arnet U, von Segesser L, Siebenmann R, Turina M, et al. Different effects of angiotensin-converting enzyme inhibition in human arteries and veins. Journal of Cardiovascular Pharmacology 22 (Suppl. 5): S17–S22, 1993a
Yang Z, von Segesser L, Bauer E, Stulz P, Turina M, et al. Different mobilization of calcium in endothelin-1-induced contractions in human arteries and veins: effects of calcium antagonists. Journal of Cardiovascular Pharmacology 16: 654–660, 1990b
Yang Z, Richard V, von Segesser L, Bauer E, Stulz P, et al. Threshold concentrations of endothelin-1 potentiate contractions to norepinephrine and serotonin in human arteries: a new mechanism of vasospasm? Circulation 82: 188–195, 1990a
Yang Z, Noll G, Lüscher TF. Calcium antagonists differently inhibit proliferation of human coronary artery smooth muscle cells in response to pulsatile stretch and platelet-derived growth factor. Circulation 88: 832–836, 1993b
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lüscher, T.F., Yang, Z. Calcium Antagonists and ACE Inhibitors. Drugs 46 (Suppl 2), 121–132 (1993). https://doi.org/10.2165/00003495-199300462-00021
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199300462-00021