Abstract
Scleroderma, also known as progressive systemic sclerosis (SSc), is a multisystem autoimmune disorder characterized by inflammation and fibrosis involving the skin as well as internal organs such as the vasculature, esophagus, and the respiratory tract. Pulmonary involvement consists most often of interstitial fibrosis and pulmonary vascular disease leading to pulmonary arterial hypertension (PAH). Bronchiectasis is an uncommon pulmonary manifestation of systemic sclerosis. Pulmonary hemorrhage with acute renal failure and diffuse alveolar hemorrhage in the absence of a history of renal involvement or penicillamine intake have rarely been reported in patients with systemic sclerosis.
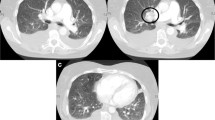
On high resolution CT, evidence of interstitial disease is seen in approximately 90% of patients, the main findings being a fine reticular pattern involving the subpleural regions of the lower lobe. Other common findings include ground-glass opacities, honeycombing, and parenchymal micronodules. The most distinctive pulmonary histologic findings in patients with scleroderma are the vascular changes found in PAH in the absence of significant interstitial fibrosis.
There is no strong evidence that any drug alters the course of the two main types of lung disease in systemic sclerosis. This apparent failure of therapy may reflect the fact that pulmonary involvement is usually identified at an established or late stage. It has been suggested that, for fibrosing alveolitis, corticosteroids are most effective if given in combination with cyclophosphamide. In some patients with SSc, PAH has been considered as a major cause of morbidity and mortality. Centrally infused prostacyclin (epoprostenol) and its subcutaneously infused analog treprostinil improve hemodynamics, as well as the quality of life and survival in these patients. Iloprost has also shown a positive effect on PAH in SSc patients. More recently, bosentan, an endothelin receptor antagonist, has proved effective in controlling PAH after 6 months’ treatment. Sildenafil has been used as a selective pulmonary vasodilator in SSc patients with isolated PAH. This drug decreased mean pulmonary artery pressure and pulmonary vascular resistance, and increased cardiac output, with much improvement of the physical condition of the patients. Lung transplant can be considered as a last option.
Clinicians must be aware of the possibility of lung disease in patients with SSc so that it can be treated as early as possible.

Similar content being viewed by others
References
Steen V, Owens G, Fino G, et al. Pulmonary involvement in systemic sclerosis (scleroderma). Arthritis Rheum 1985; 28: 759–67
Steen V. Predictors of end stage lung disease in systemic sclerosis. Ann Rheum Dis 2003; 62: 97–9
Weaver AL, Divertie MB, Titus JL. Pulmonary scleroderma. Dis Chest 1968; 54: 490–8
Rossi GA, Bitterman PB, Rennard SI, et al. Evidence for chronic inflammation as a component of the interstitial lung disease associated with progressive systemic sclerosis. Am Rev Respir Dis 1985; 124: 612–7
Harrison NK, Myers AR, Corrin B, et al. Structural features of interstitial lung disease in systemic sclerosis. Am Rev Respir Dis 1991; 144: 706–13
Morgan C, Knight C, Lunt M, et al. Predictors of end stage lung disease in a cohort of patients with scleroderma. Ann Rheum Dis 2003; 62: 146–50
Scully R, Mark E, McNeely W, et al. Case records of the Massachusetts General Hospital. N Engl J Med 1989; 320: 1333–40
Gindzienska-Sieskiewicz E, Kowal-Bielecka O, Kita J, et al. The occurrence of pulmonary hypertension in patients with systemic sclerosis hospitalized in the department of rheumatology and internal medical University of Bialystok in years 2003–2004. Rocz Akad Med Bialymst 2005; 50: 297–300
Salerni R, Rodnan GP, Leon DF, et al. Pulmonary hypertension in the CREST syndrome variant of progressive systemic sclerosis (scleroderma). Ann Intern Med 1977; 68: 394–9
White B, Moore WC, Wigley FM, et al. Cyclophosphamide is associated with pulmonary function and survival benefit in patients with scleroderma and alveolitis. Ann Intern Med 2000; 132: 947–54
Jacobsen S, Halberg P, Ullman S, et al. A longitudinal study of pulmonary function in Danish patients with systemic sclerosis. Clin Rheumatol 1997; 16: 384–90
Highland KB, Silver RM. Clinical aspects of lung involvement: lessons from idiopathic pulmonary fibrosis and the scleroderma lung study. Curr Rheumatol Rep 2005; 7(2): 135–41
Du Bois RM. The genetic predisposition to interstitial lung disease. Chest 2002; 121: 14–20
Dziankowska-Bartkowiak B, Waszczykowska E, Luczynska M, et al. Serum levels of tissue inhibitor of metalloproteinases 2 in systemic sclerosis: a preliminary study. Med Sci Monit 2002; 8(2): 108–12
Mattila L, Airola K, Ahonen M, et al. Activation of tissue inhibitor of metalloproteinases-3 (TIMP-3) mRNA expression in scleroderma skin fibroblasts. J Invest Dermatol 1998; 110: 416–21
Beon M, Harley RA, Wessels A, et al. Myofibroblast induction and microvascular alteration in scleroderma lung fibrosis. Clin Exp Rheumatol 2004; 22(6): 733–42
Wallaert B, Hatron PY, Grosbois JM, et al. Subclinical pulmonary involvement in connective tissue diseases assessed by bronchoalveolar lavage: relationship between alveolitis and subsequent changes in lung function. Am Rev Respir Dis 1986; 133: 574–80
Rudd RM, Haslam PL, Turner-Warwick M. Cryptogenic fibrosing alveolitis: relationship of pulmonary physiology and bronchoalveolar lavage to response to treatment and prognosis. Am Rev Respir Dis 1982; 124: 1–9
Steen VD, Graham G, Conte C, et al. Isolated diffusing capacity reduction in systemic sclerosis. Arthritis Rheum 1992; 35: 765–70
Alton E, Turner-Warwick M. Lung involvement in scleroderma. In: Jayson MIV, Black CM, eds. Systemic sclerosis: scleroderma. London: Wiley, 1988: 181–205
Watters LC, King TE, Schwarz MI, et al. A clinical, radiological, physiological scoring system for the longitudinal assessment of patients with idiopathic pulmonary fibrosis. Am Rev Respir Dis 1986; 133: 97–103
Eaton T, Young P, Milne D, et al. Six minute walk, maximal exercise tests: reproducibility in fibrotic interstitial pneumonia. Am J Resp Crit Care Med 2005; 171: 1150–7
Witt Ch, Dörner T, Hiepe F, et al. Diagnosis of alveolitis in interstitial lung manifestation in connective tissue diseases: importance of late inspiratory crackles, 67 gallium scan and bronchoalveolar lavage. Lupus 1996; 5: 606–12
Wells AU, Hansell DM, Rubens MB, et al. Fibrosing alveolitis in systemic sclerosis. Arthritis Rheum 1997; 40: 1229–36
Witt C, Borges AC, John M, et al. Pulmonary involvement in diffuse cutaneous systemic sclerosis: bronchoalveolar fluid granulocytosis predicts progression of fibrosing alveolitis. Ann Rheum Dis 1999; 58: 635–40
Silver RM. Scleroderma: clinical problems. The lungs. Rheum Dis Clin North Am 1996; 22: 825–40
Silver RM, Scott Miller K, Kinsella MB, et al. Evaluation and management of scleroderma lung disease using bronchoalveolar lavage. Am J Med 1990; 88: 470–6
Behr J, Vogelmeier C, Beinert T, et al. Bronchoalveolar lavage for evaluation and management of scleroderma disease of the lung. Am J Respir Crit Care Med 1996; 154: 400–6
Clements PJ, Goldin JG, Kleerup EC, et al. Regional differences in bronchoalveolar lavage and thoracic high resolution computed tomography results in dyspneic patients with systemic sclerosis. Arthritis Rheum. 2004; 50(6): 1909–17
Silver RM, Warrick JH, Kinsella MB, et al. Cyclophosphamide and low-dose prednisone therapy in patients with systemic sclerosis (scleroderma) with interstitial lung disease. J Rheumatol 1993; 20(5): 838–44
Akesson A, Scheja A, Lundin A, et al. Improved pulmonary function in systemic sclerosis after treatment with cyclophosphamide. Arthritis Rheum 1994; 37: 729–35
Varai G, Earle L, Jimenez SA, et al. A pilot study of intermittent intravenous cyclophosphamide for the treatment of systemic sclerosis associated lung disease. J Rheumatol 1998; 25: 1325–9
Davas EM, Peppas C, Maragou M, et al. Intravenous cyclophosphamide pulse therapy for the treatment of lung disease associated with scleroderma. Clin Rheumatol 1999; 18: 455–61
Pakas I, Ioannidis JP, Malagari K, et al. Cyclophosphamide with low or high dose prednisolone for systemic sclerosis lung disease. J Rheumatol 2002; 29: 298–304
Giacomelli R, Valentini G, Salsano F, et al. Cyclophosphamide pulse regimen in the treatment of alveolitis in systemic sclerosis. J Rheumatol 2002; 29: 731–6
Griffiths B, Miles S, Hilary M, et al. Systemic sclerosis and interstitial lung disease: a pilot study using pulse intravenous methylprednisolone and cyclophosphamide to assess the effect on high resolution computed tomography scan and lung function. J Rheumatol 2002; 29: 2371–8
Schnabel A, Reuter M, Gross WL. Intravenous pulse cyclophosphamide in the treatment of interstitial lung disease due to collagen vascular diseases. Arthritis Rheum 1998; 41: 1215–20
Kowal-Bielecka O, Kowal K, Rojewska J, et al. Cyclophosphamide reduces neutrophilic alveolitis in patients with scleroderma lung disease: a retrospective analysis of serial bronchoalveolar lavage investigations. Ann Rheum Dis 2005; 64: 1343–6
Airo P, Danieli E, Parrinello G, et al. Intravenous cyclophosphamide therapy for systemic sclerosis: a single-center experience and review of the literature with pooled analysis of lung function test results. Clin Exp Rheumatol 2004; 22(5): 551–2
Beretta L, Caronni M, Raimondi M, et al. Oral cyclophosphamide improves pulmonary function in scleroderma patients with fibrosing alveolitis: experience in one centre. Clin Rheumatol. Epub 2006 Apr 14
Steen VD, Medsger TA, Rodnan GP. D-Penicillamine therapy in progressive systemic sclerosis (scleroderma). Ann Intern Med 1982; 97: 652–9
Calguneri M, Apras S, Ozbalkan Z, et al. The efficacy of oral cyclophosphamide plus prednisolone in early diffuse systemic sclerosis. Clin Rheumatol 2003; 22: 289–94
De Clerck LS, Dequeker J, Francx L, et al. D-Penicillamine therapy and interstitial lung disease in scleroderma: a long term followup study. Arthritis Rheum 1987; 30: 643–50
Akesson A, Blom-Bulow B, Scheja A, et al. Long-term evaluation of penicillamine or cyclofenil in systemic sclerosis: results from a two-year randomized study. Scand J Rheumatol 1992; 21(5): 238–44
Clements PJ, Lachenbruch PA, Sterz M, et al. Cyclosporine in systemic sclerosis: results of a forty eight week open safety study in ten patients. Arthritis Rheum 1993; 36(1): 75–83
Pope Je, Bellamy N, Seibold JR, et al. A randomized, controlled trial of methotrexate versus placebo in early diffuse scleroderma. Arthritis Rheum 2001; 44(6): 1351–8
Sontag SJ, O’Connell S, Khandelwal S, et al. Most asthmatics have gastroesophageal reflux with or without bronchodilator therapy. Gastroenterology 1990; 99: 613–20
Mansfield LE. Gastroesophageal reflux and diseases of the respiratory tract: A review. J Asthma 1989; 26: 271–8
Mansfield LE, Hameister HH, Spaulding HS, et al. The role of the vagus nerve in airway narrowing caused by intraesophageal hydrochloric acid provocation and esophageal distention. Ann Allergy 1981; 47: 431–4
Tuchman DN, Boyle JT, Pack AI, et al. Comparison of airway responses following tracheal or esophageal acidification in the cat. Gastroenterology 1984; 87: 872–81
Mansfield LE, Stein MR. Gastroesophageal reflux and asthma: a possible reflex mechanism. Ann Allergy 1978; 41: 224–6
Pack AI. Acid: a nocturnal bronchoconstrictor [editorial]? Am Rev Respir Dis 1990; 141: 1391–2
Larrain A, Carrasco E, Galleguillos F, et al. Medical and surgical treatment of nonallergic asthma associated with gastroesophageal reflux. Chest 1991; 99: 1330–5
Perrin-Fayolle M, Gormand F, Braillon G, et al. Long-term results of surgical treatment for gastroesophageal reflux in asthmatic patients. Chest 1989; 96: 40–5
Johnson DA, Drane WE, Curran J, et al. Pulmonary disease in progressive systemic sclerosis: a complication of gastroesophageal reflux and occult aspiration? Arch Intern Med 1989; 149: 589–93
Moran TJ. Experimental aspiration pneumonia. Arch Pathol 1955; 60: 122–9
Chernow B, Johnson LF, Janowitz WR, et al. Pulmonary aspiration as a consequence of gastroesophageal reflux: a diagnostic approach. Dig Dis Sci 1979; 24: 839–44
Troshinsky MB, Kane GC, Varga J, et al. Pulmonary function and gastroesophageal reflux in systemic sclerosis. Ann Intern Med 1994; 121: 6–19
Henry DA. Pulmonary cysts in progressive systemic sclerosis (scleroderma). Rev Interam Radiol 1980; 5(4): 113–6
Bar J, Ehrenfeld M, Rozenman J, et al. Pulmonary-renal syndrome in systemic sclerosis. Semin Arthritis Rheum 2001; 30(6): 403–10
Yamada T, Nakajima H, Tanaka E, et al. Case of systemic sclerosis presenting with alveolar hemorrhage and positive anti-neutrophil cytoplasmic myeloperoxidase antibody (MPO-ANCA) without pathological renal involvement. Ryumachi 2003; 43(4): 690–5
Lavie F, Rozenberg S, Coutaux A, et al. Bronchiectasis in a patient with CREST syndrome. Joint Bone Spine 2002; 69(5): 515–8
Andonopoulos AP, Yarmenitis S, Georgiou P, et al. Bronchiectasis in systemic sclerosis: a study using high resolution computed tomography. Clin Exp Rheumatol 2001; 19(2): 187–90
Stupi AM, Steen VD, Owens GR, et al. Pulmonary hypertension in the CREST syndrome variant of systemic sclerosis. Arthritis Rheum 1986; 29: 515–23
Galei N, Torbicki A, Robyn B, et al. Guidelines on diagnosis and treatment of pulmonary arterial hypertension. Europ Heart J 2006; 25(24): 2243–78
Steen VD, Owens GR, Fino GJ, et al. Isolated diffusing capacity reduction in systemic sclerosis. Arthritis Rheum 1992; 28: 759–67
Kharitonov SA, Cailes JB, Black CM, et al. Decreased nitric oxide in the exhaled air of patients with systemic sclerosis with pulmonary hypertension. Thorax 1997; 52: 1051–5
Ihn H, Sato S, Fujimoto M, et al. Measurement of anticardiolipin antibodies by ELISA using β2-glycoprotein I (β2-GPI) in systemic sclerosis. Clin Exp Immunol 1996; 37: 1188–92
Jinnin H, Ihn K, Yamane Y, et al. Plasma plasmin-2-plasmin inhibitor complex levels are increased in systemic sclerosis patients with pulmonary hypertension. Rheumatology 2003; 42: 240–3
Kon OM, Daniil Z, Black CM, et al. Clearance of inhaled technetium-99m-DTPA as a clinical index of pulmonary vascular disease in systemic sclerosis. Eur RespirJ 1999; 13(1): 133–6
Yamane K, Ihn H, Asano Y, et al. Clinical and laboratory features of scleroderma patients with pulmonary hypertension. Rheumatology 2000; 39: 1269–71
Rich S, Kauffman E, Levy PS. The effect of high doses of calcium channel blockers on survival in primary pulmonary hypertension. N Engl J Med 1992; 327: 76–81
Seibold JR, Molony RR, Turkevich D, et al. Acute hemodynamic effects of ketanserin in pulmonary hypertension secondary to systemic sclerosis. J Rheumatol 1987; 14: 519–24
Barst RJ, Rubin LJ, McGoon MD, et al. Survival in primary pulmonary hypertension with long term continuous intravenous prostacyclin. Ann Intern Med 1994; 121: 409–15
Shapiro SM, Oudiz RJ, Cao T, et al. Primary pulmonary hypertension: improved long-term effects and survival with continuous intravenous epoprostenol infusion. J Am Coll Cardiol 1997 Aug; 30(2): 343–9
Higenbottam T, Butt AY, McMahon A, et al. Long-term intravenous prostaglandin (epoprostenol or iloprost) for treatment of severe pulmonary hypertension. Heart 1998 Aug; 80(2): 151–5
Cao TS, Yuan LJ, Duan YY, et al. Clinical study on long-term treatment of primary pulmonary hypertension with continuous intravenous epoprostenol infusion [in Chinese]. Zhonghua Nei Ke Za Zhi 2003; 42(2): 106–9
Sitbon O, Humbert M, Nunes H, et al. Long-term intravenous epoprostenol infusion in primary pulmonary hypertension: prognostic factors and survival. J Am Coll Cardiol 2002 Aug 21; 40(4): 780–8
Barst RJ, Rubin LJ, Long WA, et al. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension: the Primary Pulmonary Hypertension Study Group. N Engl J Med 1996; 334: 296–302
McLaughlin VV, Genthner DE, Panella MM, et al. Reduction in pulmonary vascular resistance with long-term epoprostenol (prostacyclin) therapy in primary pulmonary hypertension. N Engl J Med 1998; 338: 273–7
Humbert M, Sanchez O, Fartoukh M, et al. Short-term and long-term epoprostenol (prostacyclin) therapy in pulmonary hypertension secondary to connective tissue diseases: results of a pilot study. Eur Respir J 1999; 13(6): 1351–6
United Therapeutics Corporation. Remodulin® (treprostinil sodium) injection: product information. Research Triangle Park (NC): United Therapeutics Corporation, 2002
Oudiz RJ, Schilz RJ, Barst RJ, et al. Treprostinil, a prostacyclin analogue, in pulmonary arterial hypertension associated with connective tissue disease. Chest 2004; 126: 420–7
Tapson VF, Gomberg-Maitland M, McLaughlin VV, et al. Safety and efficacy of IV treprostinil for pulmonary arterial hypertension: a prospective, multicenter open-label, 12 week trial. Chest 2006; 129(3): 683–8
Bettoni L, Geri A, Airo P, et al. Systemic sclerosis therapy with iloprost: a prospective observational study of 30 patients treated for a median of 3 years. Clin Rheumatol 2002; 21(3): 244–50
Baker SE, Hockman RH. Inhaled iloprost in pulmonary arterial hypertension. Ann Pharmacother 2005; 39: 1265–74
Humbert M, Cabane J. Successful treatment of systemic sclerosis digital ulcers and pulmonary arterial hypertension with endothelin receptor antagonist bosentan. Rheumatology 2003; 42: 191–3
Yamane K. Endothelin and collagen vascular disease: a review with special reference to Raynaud’s phenomenon and systemic sclerosis. Intern Med 1994; 33: 579–82
Badesch DB, Tapson VF, McGoon MD, et al. Continuous intravenous epoprostenol for pulmonary hypertension due to the scleroderma spectrum of disease: a randomized, controlled trial. Ann Intern Med 2000; 132: 425–34
Oldfield V, Lyseng-Williamson KA. Bosentan: a review of its use in pulmonary arterial hypertension and systemic sclerosis. Am J Cardiovasc Drugs 2006; 6(3): 189–208
Rubin LJ, Badesch DB, Barst RJ, et al. Bosentan therapy for pulmonary arterial hypertension. N Engl J Med 2002; 346: 896–903
Hayakawa I, Shirasaki F, Hirano T, et al. Successful treatment with sildenafil in systemic sclerosis patients with isolated pulmonary arterial hypertension: two case reports. Rheumatol Int 2006; 26(3): 270–3
Singh TP, Rohit M, Grover A, et al. A randomized, placebo-controlled, double-blind, crossover study to evaluate the efficacy of oral sildenafil therapy in severe pulmonary artery hypertension. Am Heart J 2006; 151(4): 851–5
Aizawa K, Hanaoka T, Kasai H, et al. Long-term vardenafil therapy improves hemodynamics in patients with pulmonary hypertension. Hypertens Res 2006; 29(2): 123–8
Beyer S, Speich R, Fischler M, et al. Long-term experience with oral or inhaled vasodilator combination therapy in patients with pulmonary hypertension. Swiss Med Wkly 2006; 136: 114–8
Massad MG, Powell CR, Kpodonu J, et al. Outcomes of lung transplantation in patients with scleroderma. World J Surg 2005; 29(11): 1510–5
Acknowledgment
There are no sources of funding or conflicts of interest directly relevant to the preparation or content of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shahin, A.A. Pulmonary Involvement in Systemic Sclerosis. Treat Respir Med 5, 429–436 (2006). https://doi.org/10.2165/00151829-200605060-00008
Published:
Issue Date:
DOI: https://doi.org/10.2165/00151829-200605060-00008