Abstract
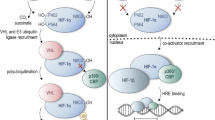
Reduced oxygen supply that does not satisfy tissue and cellular demand (hypoxia) regularly occurs both in health and disease. Hence, the capacity for cellular oxygen sensing is of vital importance for each cell to be able to alter its energy metabolism and promote adaptation to hypoxia. The hypoxia-inducible factor (HIF) prolyl hydroxylases 1–3 (PHD1–3) and the asparagine hydroxylase factor-inhibiting HIF (FIH) are the primary cellular oxygen sensors, which confer cellular oxygen-dependent sensitivity upon HIF as well as other hypoxia-sensitive pathways, such as nuclear factor κB (NF-κB). Studying these enzymes allows us to understand the oxygen-dependent regulation of cellular processes and has led to the development of several putative novel therapeutics, which are currently in clinical trials for the treatment of anemia associated with kidney disease. Pharmacologic inhibition and genetic knockdown are commonly established techniques in protein biochemistry and are used to investigate the activity and function of proteins. Here, we describe specific protocols for the knockdown and inhibition of the HIF prolyl hydroxylases 1–3 (PHD1–3) and the asparagine hydroxylase factor-inhibiting HIF (FIH) using RNA interference (RNAi) and hydroxylase inhibitors, respectively. These techniques are essential tools for the analysis of the function of the HIF hydroxylases, allowing the investigation and discovery of novel functions and substrates of these enzymes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kaelin WG Jr, Ratcliffe PJ (2008) Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol Cell 30(4):393–402
Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ, Kriegsheim A, Hebestreit HF, Mukherji M, Schofield CJ, Maxwell PH, Pugh CW, Ratcliffe PJ (2001) Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 292(5516):468–472
Berra E, Benizri E, Ginouves A, Volmat V, Roux D, Pouyssegur J (2003) HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1alpha in normoxia. EMBO J 22(16):4082–4090
Appelhoff RJ, Tian YM, Raval RR, Turley H, Harris AL, Pugh CW, Ratcliffe PJ, Gleadle JM (2004) Differential function of the prolyl hydroxylases PHD1, PHD2, and PHD3 in the regulation of hypoxia-inducible factor. J Biol Chem 279(37):38458–38465
Cockman ME, Lancaster DE, Stolze IP, Hewitson KS, McDonough MA, Coleman ML, Coles CH, Yu X, Hay RT, Ley SC, Pugh CW, Oldham NJ, Masson N, Schofield CJ, Ratcliffe PJ (2006) Posttranslational hydroxylation of ankyrin repeats in IkappaB proteins by the hypoxia-inducible factor (HIF) asparaginyl hydroxylase, factor inhibiting HIF (FIH). Proc Natl Acad Sci U S A 103(40):14767–14772
Cummins EP, Berra E, Comerford KM, Ginouves A, Fitzgerald KT, Seeballuck F, Godson C, Nielsen JE, Moynagh P, Pouyssegur J, Taylor CT (2006) Prolyl hydroxylase-1 negatively regulates IkappaB kinase-beta, giving insight into hypoxia-induced NFkappaB activity. Proc Natl Acad Sci U S A 103(48):18154–18159
Stiehl DP, Wirthner R, Koditz J, Spielmann P, Camenisch G, Wenger RH (2006) Increased prolyl 4-hydroxylase domain proteins compensate for decreased oxygen levels. Evidence for an autoregulatory oxygen-sensing system. J Biol Chem 281(33):23482–23491
Scholz CC, Cavadas MA, Tambuwala MM, Hams E, Rodriguez J, von Kriegsheim A, Cotter P, Bruning U, Fallon PG, Cheong A, Cummins EP, Taylor CT (2013) Regulation of IL-1beta-induced NF-kappaB by hydroxylases links key hypoxic and inflammatory signaling pathways. Proc Natl Acad Sci U S A 110(46):18490–18495
Scholz CC, Rodriguez J, Pickel C, Burr S, Fabrizio JA, Nolan KA, Spielmann P, Cavadas MA, Crifo B, Halligan DN, Nathan JA, Peet DJ, Wenger RH, Von Kriegsheim A, Cummins EP, Taylor CT (2016) FIH regulates cellular metabolism through hydroxylation of the deubiquitinase OTUB1. PLoS Biol 14(1):e1002347
Carthew RW, Sontheimer EJ (2009) Origins and mechanisms of miRNAs and siRNAs. Cell 136(4):642–655
Bagnall J, Leedale J, Taylor SE, Spiller DG, White MR, Sharkey KJ, Bearon RN, See V (2014) Tight control of hypoxia-inducible factor-alpha transient dynamics is essential for cell survival in hypoxia. J Biol Chem 289(9):5549–5564
Scholz CC, Taylor CT (2013) Hydroxylase-dependent regulation of the NF-kappaB pathway. Biol Chem 394(4):479–493
Fu J, Taubman MB (2010) Prolyl hydroxylase EGLN3 regulates skeletal myoblast differentiation through an NF-{kappa}B-dependent pathway. J Biol Chem 285(12):8927–8935
Wenger RH, Kurtcuoglu V, Scholz CC, Marti HH, Hoogewijs D (2015) Frequently asked questions in hypoxia research. Hypoxia 3:35–43
Bruning U, Cerone L, Neufeld Z, Fitzpatrick SF, Cheong A, Scholz CC, Simpson DA, Leonard MO, Tambuwala MM, Cummins EP, Taylor CT (2011) MicroRNA-155 promotes resolution of hypoxia-inducible factor 1alpha activity during prolonged hypoxia. Mol Cell Biol 31(19):4087–4096
Stiehl DP, Bordoli MR, Abreu-Rodriguez I, Wollenick K, Schraml P, Gradin K, Poellinger L, Kristiansen G, Wenger RH (2012) Non-canonical HIF-2alpha function drives autonomous breast cancer cell growth via an AREG-EGFR/ErbB4 autocrine loop. Oncogene 31(18):2283–2297
Cavadas MA, Mesnieres M, Crifo B, Manresa MC, Selfridge AC, Scholz CC, Cummins EP, Cheong A, Taylor CT (2015) REST mediates resolution of HIF-dependent gene expression in prolonged hypoxia. Sci Rep 5:17851
Coelen RJ, Jose DG, May JT (1983) The effect of hexadimethrine bromide (polybrene) on the infection of the primate retroviruses SSV 1/SSAV 1 and BaEV. Arch Virol 75(4):307–311
Jain IH, Zazzeron L, Goli R, Alexa K, Schatzman-Bone S, Dhillon H, Goldberger O, Peng J, Shalem O, Sanjana NE, Zhang F, Goessling W, Zapol WM, Mootha VK (2016) Hypoxia as a therapy for mitochondrial disease. Science 352(6281):54–61
Barrett TD, Palomino HL, Brondstetter TI, Kanelakis KC, Wu X, Haug PV, Yan W, Young A, Hua H, Hart JC, Tran DT, Venkatesan H, Rosen MD, Peltier HM, Sepassi K, Rizzolio MC, Bembenek SD, Mirzadegan T, Rabinowitz MH, Shankley NP (2011) Pharmacological characterization of 1-(5-chloro-6-(trifluoromethoxy)-1H-benzoimidazol-2-yl)-1H-pyrazole-4-carboxylic acid (JNJ-42041935), a potent and selective hypoxia-inducible factor prolyl hydroxylase inhibitor. Mol Pharmacol 79(6):910–920
Acknowledgments
This work was supported by ERACoSysMed (15/ERA-CSM/3267) and Science Foundation Ireland (11/PI/1005) awarded to Cormac T. Taylor, and by a Forschungskredit of the University of Zurich (FK-15-046) awarded to Carsten C. Scholz.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Pickel, C., Taylor, C.T., Scholz, C.C. (2018). Genetic Knockdown and Pharmacologic Inhibition of Hypoxia-Inducible Factor (HIF) Hydroxylases. In: Huang, L. (eds) Hypoxia. Methods in Molecular Biology, vol 1742. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7665-2_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7665-2_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7664-5
Online ISBN: 978-1-4939-7665-2
eBook Packages: Springer Protocols